 |
 |
 |
| |
CROI (2010 Feb 16-19) Selected Neurocognitive Abstracts
|
| |
| |
Persistence and Progression of HIV-associated Neurocognitive ...
July 19th-22nd 2009. Capetown, South Africa, Back · grey_arrow_rt.gif ... Antiretroviral Therapy (CART) and the Role of Comorbidities: The CHARTER Study ...
www.natap.org/2009/IAS/IAS_81.htm
Prevalence and Correlates of Minor Neurocognitive Disorders in Asymptomatic HIV-infected Outpatients: (51.5%) were classified as cognitively impaired.
N Ciccarelli, M Fabbiani, S Di Giambenedetto, I Fanti, M Colafigli, L Bracciale, E Tamburrini, R Cauda, Andrea De Luca*, and M C Silveri
Catholic Univ of the Sacred Heart, Rome, Italy
Background: Despite the availability of potent antiretroviral regimens (cART), minor cognitive disorders are increasingly recognized in HIV-infected patients. However, their prevalence, characteristics and clinical and treatment-related correlates need to be better investigated.
Methods: We performed a cross-sectional single cohort study, consecutively enrolling asymptomatic HIV+ subjects during routine outpatient visits. All patients underwent Mini Mental State Examination and an extensive neuropsychological battery exploring memory, attention, and executive abilities. The Zung Depression Scale was also administered. Each patient was considered cognitively impaired if he scored below the cut-off in >3 tests, on the basis of results obtained in matched healthy HIV-negative subjects. Central nervous system penetration-effectiveness (CPE) rank was calculated for each cART regimen according to CHARTER group. Factors associated with cognitive impairment were investigated by linear or logistic regression analysis.
Results: There were 136 patients enrolled, with the following characteristics: 59.6% males, median age 47 years (IQR 40 to 54), 15.4% non-Italian born, 27.2% with past AIDS-defining events, 77.2% with HIV-RNA<50 copies/mL, median CD4 cells count 509/μL (IQR 394 to 703). Overall, 119 (87.5%) subjects were on cART, 68% of whom with a stable (>1 year) regimen. CPE rank was >1.5 in 55.5% of subjects on therapy. The overall median of pathological tests was 3 (IQR 1 to 4). Seventy patients (51.5%) were classified as cognitively impaired. At multivariate analysis, CD4 cells count nadir (OR 0.73 per 100 cells increase, P =0.018) was significantly associated to a lower risk of cognitive impairment, while efavirenz (EFV) use was associated to increased risk (OR 5.37, P =0.004). No association was observed between Zung Depression score and cognitive performance. Considering any single task, only performance in procedural learning was significantly predicted by EFV use (P =0.030). Higher CD4 cells count nadir was a good predictor of better performance in memory (P =0.039). In this cross-sectional study, no association between CPE rank and cognitive impairment was observed, even after excluding patients receiving EFV.
Conclusions: High prevalence of minor cognitive disorders was observed in apparently asymptomatic HIV+ individuals. Mild cognitive impairment was associated to lower nadir CD4 count and EFV use. Neuropsychological investigation is important in the long-term management of HIV+ patients.
CNS Toxicity of Antiretroviral Drugs
J Liner, R Meeker, and Kevin Robertson
Univ of North Carolina at Chapel Hill, US
Background: Potent combination therapies have been shown to improve cognition, although recent studies have shown that cognitive manifestations of HIV infection persist in some on treatment. It is known that there are lower drug concentrations in the central nervous system (CNS) than in plasma. Some are working on methods to deliver antiretrovirals to the CNS, but there is limited data on the potential toxicity of antiretrovirals to neurons.
Methods: In cultured rat cortical neurons we challenged cells for 1 week with doses of 16 antiretrovirals at concentrations ranging from 0.01 to 300 μg/mL to determine single drug toxicities. In addition, 6 combinations of antiretrovirals currently in the Department of Health and Human Services (DHHS) guidelines were tested at therapeutic concentrations. We assessed neuronal pathology and cell death (MAP-2 staining), calcium signaling in response to glutamate, and mitochondrial membrane potential (TMRM) using fluorescent microscopy.
Results: Analyses of MAP-2 staining demonstrated median toxic doses ranging from 81 to 64,500 ng/mL. Median toxic doses of didanosine (DDI), zidovudine (AZT), emtricitabine (FTC), and tenofovir (TDF) in particular were in the range of reported plasma concentrations. Four agents showed some signs of damage in the range of cerebrospinal fluid (CSF) concentrations. A maximum loss of 32% to 52% of neuron density was achieved at concentrations above 1 μg/mL. After treatment of cultures with a combination of EFV/FTC/TDF for 2 days, a glutamate challenge induced a 70% increase in the acute calcium response in the treated culture when compared to controls followed by a delayed destabilization of intercellular calcium. Individual agents did not elicit the same level of destabilization as combinations. With the exception of DDI, reduced TMRM staining in ART-treated neurons was seen indicating a loss of the mitochondrial membrane potential relative to controls.
Conclusions: Protecting the brain so that patients can be productive over their lifetime has become a priority. The median toxic doses for several antiretrovirals were well within the plasma therapeutic concentration range and led to a considerable loss of neurons. The ability of certain antiretrovirals and antiretroviral combinations to perturb neuronal calcium homeostasis and affect the mitochondrial membrane potential under conditions that produce little cell death also indicates the early presence of neuronal dysfunction. There may be a balance between initiating treatment to stop neuronal loss, due to HIV versus potential treatment side effects. These findings highlight potential toxicity of antiretrovirals in the CNS that should be considered.
Neuropsychological Performance Is Better in HIV-infected Subjects Treated with Neuroactive HAART
Fabrizio Starace, M de Stefano, A D’Abrosca, M Gargiulo, and A Chirianni
Cotugno Hosp, Naples, Italy
Background: The association between central nervous system (CNS) penetration effectiveness of ARV drugs and cognitive performance is still unclear. For example, in ACTG 736 study an inverse relationship was found between ART with good CNS penetration and neuropsychological performance. Here we report results from an exploratory case-control study carried out in virally suppressed HIV+ individuals.
Methods: Trained research psychologists—blind to ART regimens—administered the Cotugno Mini Battery (a brief neuropsychological battery including Verbal Memory Span, Auditory Verbal Learning, Trial 1-5, Verbal Fluency, Color Trails 2, Symbol Digit Modalities Test and Timed Gait) to subjects admitted to care. For the purpose of this analysis, cases were included if they showed stable plasma viral suppression and had ART regimen unchanged for at least 6 months before assessment. Exclusion criteria were age >60, formal educational <8 years, depressive symptomatology (MADRS score >15), other major neurological or psychiatric disorders, current use of illicit drugs. CNS penetration effectiveness score was calculated according to Italian National Guidelines for HIV Therapy. Subjects with higher CNS penetration effectiveness rank were compared with those with lower CNS penetration effectiveness rank (cut-off = 2.0) in terms of performance on each neuropsychological test.
Results: We included 45 subjects in the analysis (80% male; median age 42 years; median years of education 11); 30 patients were on ART regimen with a high CNS penetration effectiveness score (≥2). As compared to those with a low CNS penetration effectiveness score, they showed a better performance on Verbal Memory Span (5.6±1.3 vs. 4.8±1.1; p <0.05), Verbal Fluency (14.7±9.9 vs 9.8±4.6; P <0.05), Symbol Digit Modalities Test (44.4±16.4 vs 34.6±11.4; P <0.05). No significant difference was found on other tests.
Conclusions: In this sample of virally suppressed HIV+ individuals in stable HAART and not exposed to other risk factors, neurocognitive performance was better in subjects taking neuroactive ART drugs on measures of working memory, verbal fluency, and executive functions. Findings suggest the need for routine neurocognitive screening to guide the optimal management of milder but potentially relevant neuropsychological deficits. Future studies should longitudinally evaluate the course of these deficits to help identify possible interventions aiming to reduce the incidence of neurocognitive impairment in patients who achieved effective systemic viral suppression.
---
HIV-associated Neurocognitive Disorder in Acute and Early HIV Infection
David Moore*, S Letendre, R Deutsch, S Little, D Smith, D Franklin, D Rosario, R Heaton, I Grant, and the CHARTER Group
Univ of California, San Diego, US
Background: HIV-associated neurocognitive disorder (HAND) persists despite potent antiretroviral therapy (ART). Most research focuses on chronic HIV infection but events during acute and early HIV infection (AEH) may set the stage for development and persistence of HAND.
Methods: To determine prevalence of HAND in AEH, we examined 77 participants within <1 year of estimated date of infection (mean duration 20 weeks), with comprehensive neuropsychological (NP) testing and compared them to 52 HIV seronegative participants (HIV-) of similar background. To assess potential mechanisms of HAND in AEH, we compared biomarker levels in AEH participants to those in 83 participants with chronic HIV infection, defined as ≥1 year of infection (mean 7.0 years) selected to be similar on co-morbidity factors. The majority of AEH participants were ART naïve (77%) as compared to 33% of chronic HIV infection participants. We calculated a global NP deficit score (GDS), and determined NP impairment from this score. We evaluated mean comparisons with ANOVAs and calculated Cohen’s d effect sizes. Using linear regression, we examined possible predictors of NP functioning: stage of infection, plasma viral load, peak plasma viral load, CD4 count, nadir CD4 count, a panel of 11 serum biomarkers, current ART status, and HCV co-infection.
Results: AEH participants had an NP impairment rate of 26% as compared to 29% of chronic HIV infection and 13% of HIV- participants (P =0.02). There was a medium effect between HIV- and AEH (d=0.40), a slightly larger effect between HIV- and chronic HIV infection (d=0.52), and a minimal effect between AEH and chronic HIV infection (d=0.14). There were trends toward elevated levels of MCP-1 (P =0.07) and IP-10 (P =0.09) in AEH as compared to chronic HIV infection. In univariable analyses of all participants, worse NP functioning (P <0.10) was associated with higher plasma viral load, lower nadir CD4+ cell counts, AIDS diagnosis, current ART use, and higher IL-6 levels (all p-values <0.10). Using all participants, a model examining main effects and interactions of candidate predictors revealed that ART use (P =0.02) and higher levels of IL-6 (P =0.03) were associated with worse NP functioning (R2=.08, P =0.005); disease stage (AEH vs chronic HIV infection) was not.
Conclusions: Even in the earliest stages of HIV infection, NP impairment rates are double those in HIV- persons. Regardless of chronicity, those who were on ART (associated with worse HIV disease) and those with elevated serum levels of IL-6 were more likely to have worse NP performance. Because most of the AEH participants were untreated, it remains to be determined if earlier ART may lessen the burden of HAND.
Neurocognitive Performance during Primary HIV-1 Infection
Julia Peterson*1, E Lee1, F Hecht1, R Price1, K Robertson2, and S Spudich1
1Univ of California, San Francisco, US and 2Univ of North Carolina at Chapel Hill, US
Background: Indicators of neurological damage are associated with decreased performance on neuropsychological (NP) testing during chronic HIV-1 infection. Whether there is impairment of neurocognitive functioning during primary HIV-1 infection (PHI) is largely unknown. We aimed to investigate cognitive function in acute and early infection with the evaluation of NP testing performance.
Methods: Study participants were enrolled within 1 year of HIV exposure, confirmed by serologic evidence of recent infection. Analysis consisted of blood and CSF biomarkers and NP exams administered at baseline, 6 weeks, and each 6 months thereafter. In a subset of subjects, longitudinal assessments continued after antiretroviral therapy (ART) was initiated for reasons independent of this protocol. Assessment of impairment was based on the NPZ-4 (timed gait, grooved pegboard, finger tap (non-dominant) and digit symbol tests). SAS General Linear Models and mixed procedures were used for repeated measures analysis.
Results: There were 37 male subjects with PHI enrolled for a median of 103 days post-estimated infection date. Median baseline characteristics included 37 years of age, 16 years of education, and a CD4 count of 539 cells/uL. Of the participants enrolled, 40.5% reported drug use within the previous month and all were ART naïve at baseline except 3 who initiated ART <30 days before the visit. At baseline, 24/37 (65%) of subjects had evidence of impairment on one or more NPZ-4 tests (>1 SD below the mean). Sixteen (43%) had one impaired test, 6 (16%) had 2 tests impaired, and 2 (5%) had 3 impaired tests. Subjects who initiated treatment had lower mean NPZ scores (ART = -0.468 vs no ART =.02, F (1, 47) = 3.98, P =0.05). In the 22 subjects followed longitudinally without ART, there was not a significant change in mean NPZ4 over a total of 73 follow up visits (visit 1 =0.00, visit 6 = -0.83, F (5, 43) = .54, P = ns). In the limited sample of 13 subjects who started therapy, there was no change in NPZ4 detected over 44 total visits (not shown).
Conclusions: In a cohort evaluated a median of 3.5 months after initial HIV infection, NP testing was impaired in at least one domain at baseline in a majority of individuals. Whether the NP impairment in PHI reflects premorbid factors or an effect of HIV in the nervous system requires further study. In longitudinal follow-up, we detected no decline in neurocognitive performance over time in participants during this early period of HIV infection.
from Jules: I don't think earlier HAART can eliminate HIV-associated neurocognitive impairment, as you know HIV gets into the CNS & the brain very quickly, but earlier HAART may reduce the impact of HIV in causing neuurocognitive impairment.
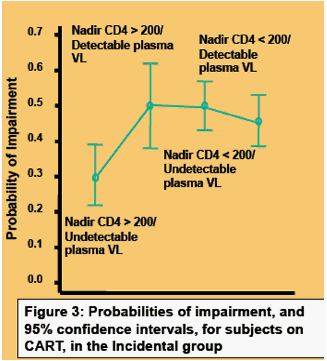
Higher CD4 Nadir Is Associated with Reduced Rates of HIV-associated Neurocognitive Disorders in the CHARTER Study: Potential Implications for Early Treatment Initiation
Ronald Ellis1, R Heaton1, S Letendre1, J Badiee1, J Munoz-Moreno1, F Vaida1, D Clifford2, B Gelman3, D Simpson4, I Grant1, and the CHARTER Group
1Univ of California, San Diego, US; 2Washington Univ in St Louis, MO, US; 3Univ of Texas Med Branch at Galveston, US; and 4Mt Sinai Sch of Med, New York, NY, US
Background: Indirect evidence suggests that early initiation of ART may prevent HIV-associated neurocognitive disorders (HAND). Prevention is important since once HAND occurs, many individuals remain impaired despite effective viral suppression on ART. To determine whether earlier ART initiation protects against HAND, we evaluated the relationship between nadir CD4+ cell counts, a surrogate for worst immunosuppression prior to ART, and risk of HAND.
Methods: Comprehensive medical and neuropsychological evaluations were performed on 1526 subjects in a multicenter cohort study, CNS HIV ART Effects Research (CHARTER). HAND was diagnosed by comprehensive neuropsychological and neuromedical testing according to published criteria. Co-morbid conditions contributing to neuropsychological impairment were evaluated by a single, experienced neuropsychological rater. Nadir CD4 was by self-report or observation.
Results: The median (IQR) nadir CD4 was 172 (48 to 297). A total of 1080 subjects (71%) took ART; 589 of these (55%) had undetectable plasma viral loads; 799 (52.4%) were neuropsychologically impaire; and 692 (45.3%) had significant co-morbid conditions. HAND diagnosis was associated with substantial disability, as measured by a structured self-report instrument. Higher CD4 nadirs were associated with lower rates of neuropsychological impairment in the entire cohort, and in the subsets with minimal co-morbidities and on ART. Among 579 subjects on ART with minimal confounds, the odds ratios (95%CI) and corresponding rates of HAND at CD4 nadir levels of 200 to 349 (n = 127) and ≥350 (n = 51) compared to 0 to 199 (reference, n = 401, of whom 47% had HAND, were 0.72 (0.47 to 1.10; 39%) and 0.57 (0.30 to 1.10; 33%) (trend c = 4.9, P = 0.03). No threshold effect was seen; higher CD4 nadir conferred lower HAND risk at all levels. The association remained significant after adjusting for other predictors including plasma viral load, age, sex, ethnicity, and duration of HIV infection.
Conclusions: These findings confirm prior reports that higher nadir CD4 is associated with a reduced risk of HAND. Results held true both for the cohort as a whole and for a subgroup of special interest comprising individuals currently taking ART and having no significant confounds. Because HAND is a common source of disability in HIV+ individuals on ART, these data suggest that earlier treatment initiation may protect patients from HAND.
CSF Escape Is Uncommon in HIV-1-infected Patients on Stable ART
Arvid Eden1, R Price2, L Hagberg1, and M Gisslen1
1The Sahlgrenska Academy at Univ of Gothenburg, Sahlgrenska Univ Hosp, Sweden and 2Univ of California, San Francisco and San Francisco Gen Hosp, US
Background: ART effectively reduces HIV-1 RNA in plasma, as well as in cerebrospinal fluid (CSF). Occasionally, patients exhibit CSF escape, or elevated RNA levels in CSF while effectively suppressed in plasma. Here, we examine CSF escape in patients effectively treated with ART in relation to treatment regimens.
Methods: There were 63 patients from 2 centers treated with ART regimens of either efavirenz, lopinavir/ritonavir or atazanavir/ritonavir in combination with 2 NRTI’s consisting of lamivudine or emtricitabine with either tenofovir, abacavir, or zidovudine for at least 6 months, with undetectable (<50 copies/mL) HIV-1 RNA in plasma were included in the analysis. HIV-1 RNA was analysed with Roche Amplicore v.1.5 or Cobas Taqman v.1.0. Fisher’s exact test was used for statistical analysis.
Results: Of 63 identified patients, 7 (11 %) had signs of CSF escape with detectable HIV-1 RNA levels in CSF (range: 46 to 213 copies/mL) despite having undetectable levels in blood. Five of 24 (21 %) efavirenz-treated and 2/13 (15 %) atazanavir-treated patients had detectable RNA in CSF. Of 26 lopinavir-treated patients, none had detectable CSF-RNA, with significantly lower frequency of CSF escape compared to efavirenz-treated patients (P =0.02). Other differences between treatment regimens did not reach statistical significance; 3/17 (18 %) and 4/27 (15 %) of patients treated with abacavir and tenofovir had detectable CSF RNA, respectively. Nineteen patients received a NRTI backbone containing zidovudine, none of which had detectable CSF RNA.
Conclusions: CSF escape, measured as detectable CSF HIV-1 RNA, remains an uncommon finding in patients with successful suppression of plasma viral load. However, there may be cause for consideration of specific ART regimen regarding CNS efficacy, especially in patients with HIV-1 related neurological disease. In our material, CSF escape was most common in patients treated with efavirenz in combination with either abacavir or tenofovir. Of note, no patients treated with the PI lopinavir/ritonavir, or the NRTI zidovudine, had detectable CSF HIV-1 RNA, while retaining undetectable plasma viral load.
cART Alters Changes in Cerebral Function Testing after 48 Weeks in Treatment-naïve, HIV-1-infected Subjects Commencing cART
A Winston1, C Duncombe2, P Li3, J Gill4, S Kerr2,5, Rebekah Puls5, K Petoumenos5, S Taylor-Robinson1, S Emery5, D Cooper5, and the Altair Study Group
1Imperial Coll London, UK; 2HIVNAT Res Collaboration, Bangkok, Thailand; 3Queen Elizabeth Hosp, Kowloon, Hong Kong; 4Calgary Regional Hlth Authority, Canada; and 5Univ of New South Wales, Sydney, Australia
Background: Neurocognitive (NC) impairment remains prevalent, despite combination antiretroviral therapy (cART). Differences between changes in cerebral function tests and alternative cART have not previously been prospectively assessed.
Methods: HIV-infected therapy-naïve individuals, randomly allocated to commence cART within the ALTAIR study (TDF/FTC plus either EFV (arm 1), ATV/RTV (arm 2) or AZT/ABC (arm 3)) were eligible. Cerebral function tests included computerised NC testing (CogState™) and assessment of cerebral metabolites using cerebral proton-MRS in 3 anatomical voxels (right frontal white (FWM), grey matter and basal ganglia (RBG)) at baseline and after 48 weeks on study. N-acetyl-aspartate/creatine (NAA/Cr) ratios were calculated. Differences between changes in NC function and NAA/Cr ratios over 48 weeks and study arms (arm 1 vs 2 and 1 vs 3) were assessed by linear regression modelling.
Results: In this study, 30 subjects completed study procedures (9, 9, and 12 subjects in arms 1, 2, and 3, respectively). Mean CD4+ counts (SD, cells/mL) were 218 (87) and 342 (145) at baseline and at week 48, respectively. Plasma HIV RNA was <50 copies/mL in 28/30 subjects at week 48. Over 48 weeks, greater improvements in identification reaction time (IRT, P =0.04) and executive function (P =0.02) were observed in arm 3 vs 1 (+0.03, –0.30, –0.50 log10 msec change IRT, in arms 1, 2, and 3, respectively). Increases in NAA/Cr were observed in all voxels (maximum 38% in RBG) over 48 weeks of study. In a multivariate model, statistically significantly greater increases in NAA/Cr were observed in arm 1 vs 2 (P =0.03) in FWM (30%, -7%, 0% change in NAA/Cr, in arms 1, 2, and 3, respectively).
Conclusions: This is the first study to prospectively describe different changes in cerebral function testing parameters between different cART. Greater improvements in neuronal recovery markers (NAA/Cr ratio) were observed in recipients of TDF/FTC plus EFV (arm 1) and greater improvements in NC function testing observed in recipients of TDF/FTC plus ABC/AZT (arm 3).
|
| |
|
 |
 |
|
|