 |
 |
 |
| |
Pharmacokinetic-pharmacodynamic (PK-PD) analyses of TMC435 in treatment-naïve hepatitis C (HCV)-infected patients in the OPERA-1 study
|
| |
| |
Reported by Jules Levin
EASL Apr 14-18 2010 Vienna Austria
V. Sekar1, P. Vis2, O. Lenz2, P. Meyvisch2, M. Peeters2, G. De Smedt2
1Tibotec Inc, USA; 2Tibotec BVBA, Belgium
AUTHOR CONCLUSIONS
In treatment-naive HCV-infected patients, following 28 days of treatment with TMC435 in combination with P/R, a dose-proportional increase in TMC435 exposure was observed from 25 to 75mg QD, with a more than dose-proportional increase in TMC435 exposure observed from 75 to 200mg QD.
There was no clear PK-PD relationship between TMC435 exposure and antiviral activity with TMC435 doses of 75mg QD or higher.
A trend towards mild increases in serum bilirubin and ALP was observed with higher TMC435 exposure, with increases mainly observed at the 200mg dose.
However, no clear PK-PD relationship was observed for other liver parameters.
The PK-PD findings reported here support the TMC435 75 and 150mg QD doses selected for further evaluation in treatment-naive patients as part of the ongoing
Phase IIb trial, PILLAR.

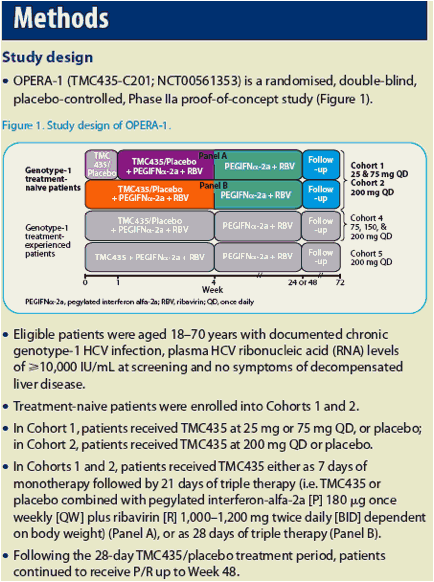


Table 1. Patient demographics and baseline characteristics of patients included in Cohorts 1 and 2 of OPERA-1.
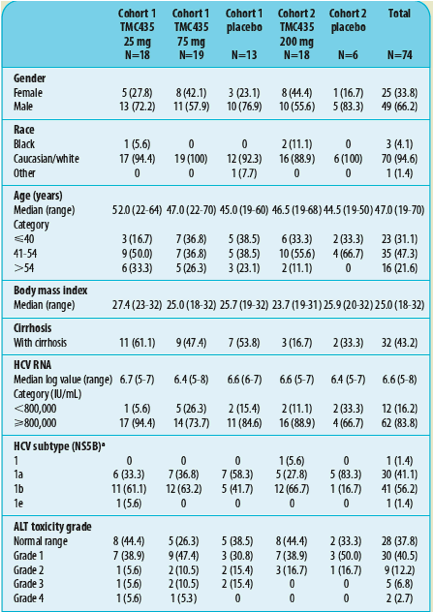
a For 1 subject in the placebo group of Cohort 1 the HCV subtype (NS5B) was unknown
ALT, alanine aminotransferase; HCV, hepatitis C virus; RNA, ribonucleic acid

Table 2. Pharmacokinetic proGle of TMC435 in treatment-naive patients in Cohorts 1 and 2 of OPERA-1 who received 7 days of TMC435 monotherapy followed by 21 days of triple therapy (TMC435+P/R) (A) or 28 days of triple therapy (TCM435+P/R) (B).
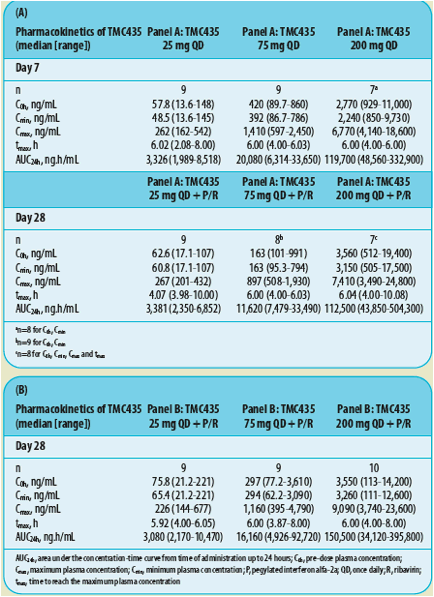
AUC24h, area under the concentration-time curve from time of administration up to 24 hours; C0h, pre-dose plasma concentration; Cmax, maximum plasma concentration; Cmin, minimum plasma concentration; P, pegylated interferon alfa-2a; QD, once daily; R, ribavirin; tmax, time to reach the maximum plasma concentration

Figure 2. Change in log10 plasma HCV RNA from baseline to Day 28 as a function of TMC435 exposure (deGned as AUC24h).
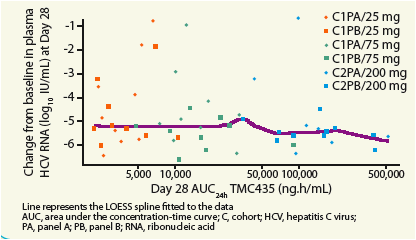

Table 3. Simulated ratios of the medians of TMC435 pharmacokinetic parameters
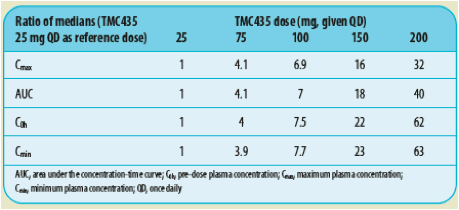

Figure 3. Change from baseline to Day 28 in (A) total, (B) direct and (C) indirect bilirubin; (D) alkaline phosphotase (ALP); (E) aspartate aminotransferase (AST) and (F) alanine aminotransferase (ALT) levels as a function of TMC435 exposure (defned as AUC24h).
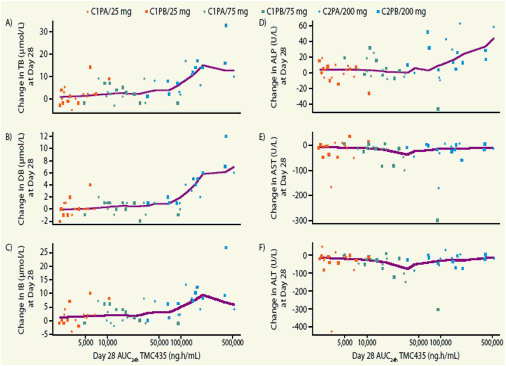
Lines represent the LOESS spline !tted to the data
AUC24h, area under the concentration-time curve from time of administration up to 24 hours; C, cohort; DB, direct bilirubin; IB, indirect bilirubin; PA, panel A; PB, panel B; TB, total bilirubin
References
1. Lin TI et al. Antimicrob Agents Chemother 2009; 53: 1377-1385.
2. Reesink HW et al. Gastroenterology 2010; 138: 913-921.
3. Reesink HW et al. Poster presented at the 60th American Association for the Study of Liver Diseases (AASLD) meeting, Boston, MA, USA, 30 October-3 November, 2009.
4. Marcellin P et al. Poster presented at the 44th Annual Meeting of European Association for the Study of the Liver (EASL), Copenhagen, Denmark, 22-26 April, 2009.
5. Manns M et al. Presented at the 44th Annual Meeting of European Association for the Study of the Liver (EASL), Copenhagen, Denmark, 22-26 April, 2009.
|
| |
|
 |
 |
|
|