| |
Successful prevention of hepatitis C virus
(HCV) liver graft reinfection by silibinin mono-therapy
|
| |
| |
Jnl of Hepatology June 2010
U.P. Neumann1, M. Biermer2, D. Eurich1, P. Neuhaus1, T. Berg
1 Klinik fur Allgemein-, Viszeral und Transplantationschirurgie, Charite Universitatsmedizin Berlin, Campus Virchow-Klinikum, Germany
2 Medizinische Klinik mit Schwerpunkt Hepatologie und Gastroenterologie, Charite - Universitatsmedizin Berlin, Campus Virchow-Klinikum, Germany
3 Klinik und Poliklinik fur Gastroenterologie und Rheumatologie Sektion Hepatologie Universitatsklinikum Leipzig, Germany
To the Editor:
Therapeutic measurements to control hepatitis C reinfection after orthotopic liver transplantation (OLT) still pose many problems and remain unsatisfactory [1]. While hepatitis C viremia decreases strongly during the anhepatic phase of OLT, initiation of viral replication does occur within hours or days [2], [3]. At this early stage after OLT, i.e. once chronic HCV reinfection is established, the virologic response is often markedly reduced. Although specifically targeted antiviral therapeutic regimes for hepatitis C (STAT-C) are currently extensively studied, none of them have been tried in the post-OLT period treatment.
However, as recently described by Ferenci et al. [4], high dose intravenous silibinin (Legalon SIL) can express potent antiviral activity. This effect was shown to be driven by direct inhibition of the viral RNA polymerase NS5B [5], [6].
In this letter, we can report the first successful prevention of HCV reinfection after OLT by the administration of silibinin (1400mg/d). This drug was applied immediately after OLT by daily infusions for 14days.
At the time of OLT, the 57-year-old male patient exhibited a MELD Score of 23, Child-Pugh stage C liver cirrhosis, and a 25mm hepatocellular carcinoma in the left lobe. HCV infection (genotype 3a) was first diagnosed in 1997 and three interferon-based treatment regimens - at last PegInterferon alpha and Ribavirin were given for 48weeks 6years before OLT - failed to induce a sustained virologic response (SVR). The anhepatic phase during OLT surgical procedures lasted 61min and postoperative care transpired without complication after a short phase of renal insufficiency and haemodialysis treatment on day 1 and 2. Immunosuppressive therapy included methyl-prednisolone, mycophenolatmofetil, and belatacept. Aminotransferase levels reached normal values within 12 postoperative days. Bilirubin levels rose to a maximum of 9.5mg/dl 7days after OLT and showed a protracted mild elevation until 4weeks later. The patient was discharged from the hospital 21days after OLT.
The levels of HCV RNA 3months prior to OLT were rather low (17.800IU/ml), and further declined significantly during the anhepatic phase. Silibinin infusions were started 8h after OLT. At this time HCV RNA levels measured 182IU/ml, and dropped again to 127IU/ml after 48h (RNA levels measured directly after haemodialysis are not available). Already from day 3 onwards, HCV RNA levels were below <15IU/ml and became undetectable at day 9. During follow-up HCV RNA remained negative when examined at day 14, 21, 66, 84, and 168 (Fig. 1).
Fig. 1. Effect of intravenous silibinin administration on the course of hepatitis C viremia (upper panel) and aminotransferase (ALT) and bilirubin levels (lower panel) after liver transplantation. HCV RNA levels were measured by real-time PCR assay (Cobas AmpliPrep Taqman, Roche Diagnostics, Germany); ALT and bilirubin levels are depicted as relative values of the upper limit of normal (ULN).
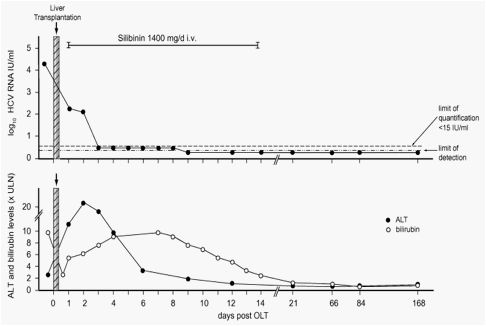
This is the first report of the successful suppression of early HCV reinfection after OLT with a 14day course of silibinin mono-therapy. This new treatment regimen thus induced an SVR, as after 6months of follow-up, RNA for HCV was undetectable. Accordingly, liver histology 6months after OLT did not show any cellular inflammation. Low pre-transplant HCV RNA levels may be taken as a favourable prognostic factor for the unexpected therapeutic efficiency of silibinin. Applying silibinin during the anhepatic phase or even in the days before transplant may expand the number of patients benefiting from this approach. This report may stimulate further trials and the concept of interferon-free HCV clearance induced by direct antivirals.
|
|
| |
| |
|
|
|