| |
Association of estimated glomerular filtration rate and albuminuria with all-cause and cardiovascular mortality in general population cohorts: a collaborative meta-analysis
|
| |
| |
Chronic Kidney Disease Prognosis Consortium
The Lancet, Early Online Publication, 18 May 2010
"This meta-analysis, which included more than 100 000 individuals with ACR measurements and 1·1 million participants with dipstick measurements from 21 general population cohorts, showed that eGFR and albuminuria were associated with all-cause mortality and cardiovascular mortality independently of each other and traditional cardiovascular risk factors."
"Thus, eGFR less than 60 mL/min/1·73 m2 and ACR 1·1 mg/mmol (10 mg/g) or more are independent predictors of all-cause mortality and cardiovascular mortality in the general population......ACR higher than 3·4 mg/mmol was associated with more than two-fold mortality risk within all eGFR categories but the lowest (see table 2, abnormal proteinuria - albumin/creatinine ratio - increased mortality risk even if eGFR was normal).....These findings provide a quantitative basis for use of these two kidney measures for risk assessment and definition and staging of chronic kidney disease......The risk became significant around eGFR 60 mL/min/1·73 m2 and was two times higher around eGFR 30-45 mL/min/1·73 m2 compared with optimum eGFR levels (90-104 mL/min/1·73 m2) independently of albuminuria and potential confounders.....The association between albuminuria and mortality was linear on the log-log scale, with a two times higher risk at ACR of approximately 11·3 mg/mmol (within the range usually described as microalbuminuria 3·4-33·8 mg/mmol)2 compared with an optimum ACR level (0·6 mg/mmol), independently of eGFR and conventional risk factors. Risk of mortality was significant at an ACR of 1·1 mg/mmol compared with 0·6 mg/mmol. Furthermore, mortality gradient conferred by ACR greater than 3·4 mg/mmol was more than two-fold within all but the lowest eGFR category......Our findings showed that even a trace urine protein on dipstick test was associated with an increased risk of mortality, consistent with the fact that 60% of individuals with trace protein on dipstick have microalbuminuria.44 These findings suggest that the dipstick test is useful for risk stratification despite being a less precise measure of albuminuria......Studies with a higher proportion of black people tended to have higher HRs for all-cause mortality. Although not significant, this finding is consistent with previous studies showing that lower eGFR is more strongly associated with adverse outcomes in black people than in white people"
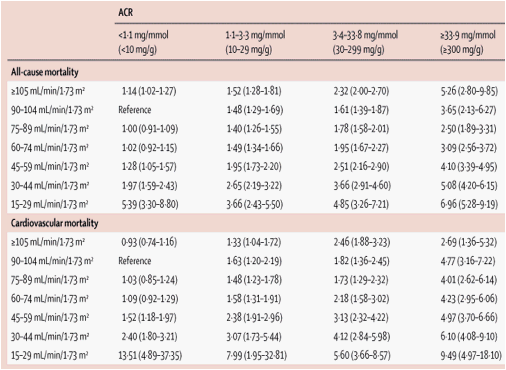
Table 2
Pooled estimates of adjusted hazard ratios (95% CI) for all-cause and cardiovascular mortality according to categories of eGFR (listed in the left column) and ACR
Summary
Background
Substantial controversy surrounds the use of estimated glomerular filtration rate (eGFR) and albuminuria to define chronic kidney disease and assign its stages. We undertook a meta-analysis to assess the independent and combined associations of eGFR and albuminuria with mortality.
Methods
In this collaborative meta-analysis of general population cohorts, we pooled standardised data for all-cause and cardiovascular mortality from studies containing at least 1000 participants and baseline information about eGFR and urine albumin concentrations. Cox proportional hazards models were used to estimate hazard ratios (HRs) for all-cause and cardiovascular mortality associated with eGFR and albuminuria, adjusted for potential confounders.
Findings
The analysis included 105 872 participants (730 577 person-years) from 14 studies with urine albumin-to-creatinine ratio (ACR) measurements and 1 128 310 participants (4 732 110 person-years) from seven studies with urine protein dipstick measurements. In studies with ACR measurements, risk of mortality was unrelated to eGFR between 75 mL/min/1·73 m2 and 105 mL/min/1·73 m2 and increased at lower eGFRs. Compared with eGFR 95 mL/min/1·73 m2, adjusted HRs for all-cause mortality were 1·18 (95% CI 1·05-1·32) for eGFR 60 mL/min/1·73 m2, 1·57 (1·39-1·78) for 45 mL/min/1·73 m2, and 3·14 (2·39-4·13) for 15 mL/min/1·73 m2. ACR was associated with risk of mortality linearly on the log-log scale without threshold effects. Compared with ACR 0·6 mg/mmol, adjusted HRs for all-cause mortality were 1·20 (1·15-1·26) for ACR 1·1 mg/mmol, 1·63 (1·50-1·77) for 3·4 mg/mmol, and 2·22 (1·97-2·51) for 33·9 mg/mmol. eGFR and ACR were multiplicatively associated with risk of mortality without evidence of interaction. Similar findings were recorded for cardiovascular mortality and in studies with dipstick measurements.
Interpretation
eGFR less than 60 mL/min/1·73 m2 and ACR 1·1 mg/mmol (10 mg/g) or more are independent predictors of mortality risk in the general population. This study provides quantitative data for use of both kidney measures for risk assessment and definition and staging of chronic kidney disease.
Funding
Kidney Disease: Improving Global Outcomes (KDIGO), US National Kidney Foundation, and Dutch Kidney Foundation.
Introduction
Chronic kidney disease is recognised as a major global public health problem.1, 2 The disease affects 10-16% of the adult population in Asia, Australia, Europe, and the USA,3-6 and increases the risk of all-cause mortality, cardiovascular disease, and progression to kidney failure, even after accounting for traditional risk factors such as hypertension and diabetes mellitus.1, 7
The 2002 Kidney Disease Outcomes Quality Initiative (KDOQI) guidelines2 define chronic kidney disease as persistent kidney damage, usually marked by albuminuria or reduced glomerular filtration rate (GFR), and assign disease stages on the basis of GFR. The KDOQI guidelines have encouraged research into the prognostic effect of chronic kidney disease and have contributed to increased awareness of it.8, 9 However, substantial controversy surrounds the use of GFR and albuminuria to define and assign stages to chronic kidney disease. Some investigators have proposed a lower GFR threshold (eg, GFR <45 mL/min/1·73 m2vs <60 mL/min/1·73 m2) or age-specific or sex-specific GFR thresholds to define chronic kidney disease, use of higher urine albumin-to-creatinine ratio (ACR) as a marker of kidney damage (ACR ≥33·9 mg/mmol [≥300 mg/g] vs ≥3·4 mg/mmol [≥30 mg/g]), combining chronic kidney disease stages 1 and 2 (GFR 60 mL/min/1·73 m2 or more), or adding albuminuria within each stage determined by GFR.2,8-11
Several studies have reported the association between estimated GFR (eGFR) or albuminuria and clinical outcomes in the general population. However, most of these studies investigated only one measure at a time, did not investigate age-specific associations, and analysed broad eGFR categories (eg, <60 mL/min/1·73 m2 or ≥60 mL/min/1·73 m2). A few studies have dealt with some, but not all, of these issues.3,12-16 Thus, there is a need for a comprehensive examination that adjusts for each measure, tests for their interactions on risk with narrow categories, and examines the association separately in younger and older individuals.
Kidney Disease: Improving Global Outcomes (KDIGO)17 has taken the lead in establishing the Chronic Kidney Disease Prognosis Consortium. The Consortium is tasked with compiling and meta-analysing the best available data to provide a more comprehensive assessment of the independent and combined associations of eGFR and albuminuria with mortality and kidney outcomes. The results of these assessments will have clear implications for improving the definition and staging of chronic kidney disease. The Consortium currently consists of 45 cohorts, which arise from general, high-risk, or chronic kidney disease populations. Separate reports will describe mortality and kidney outcomes by population type. This report describes the findings from a collaborative meta-analysis of the 21 general population cohorts for all-cause and cardiovascular mortality.
Discussion
This meta-analysis, which included more than 100 000 individuals with ACR measurements and 1·1 million participants with dipstick measurements from 21 general population cohorts, showed that eGFR and albuminuria were associated with all-cause mortality and cardiovascular mortality independently of each other and traditional cardiovascular risk factors. The consistency in both continuous and categorical models for eGFR and ACR shows that our findings are robust. With 21 studies from 14 countries in Asia, Europe, North America, and Oceania, our study shows the range of association as well as summary estimates around the world.
We noted an exponential increase in risk of mortality at low eGFRs. The risk became significant around eGFR 60 mL/min/1·73 m2 and was two times higher around eGFR 30-45 mL/min/1·73 m2 compared with optimum eGFR levels (90-104 mL/min/1·73 m2) independently of albuminuria and potential confounders. However, there was some variation across outcomes, models, and age groups tested. By contrast with previous claims,11 these findings suggest that mild to moderate reduction in eGFR is associated with adverse clinical outcomes.
Risk of mortality was fairly constant at eGFR 75-105 mL/min/1·73 m2 and, as previously reported,20, 37 was higher at eGFR more than 105 mL/min/1·73 m2, especially for all-cause mortality. However, these findings should be interpreted with caution. The MDRD Study equation is known to underestimate measured GFR at levels of 60 mL/min/1·73 m2 or more in healthy individuals,38 and to overestimate measured GFR in individuals with reduced muscle mass caused by ill health. This overestimation potentially contributes to the U-shaped association of GFR with mortality. The fact that such a U-shaped curve was less apparent for cardiovascular mortality than for all-cause mortality supports this explanation for the U-shaped association, as does the more linear association in studies in which GFR was estimated from cystatin C.37, 39 Thus, further studies that use a more accurate creatinine-based equation40 or equations incorporating other biomarkers such as cystatin C41 might provide better estimates for clinical risk at GFR 60 mL/min/1·73 m2 or more.
The association between albuminuria and mortality was linear on the log-log scale, with a two times higher risk at ACR of approximately 11·3 mg/mmol (within the range usually described as microalbuminuria 3·4-33·8 mg/mmol)2 compared with an optimum ACR level (0·6 mg/mmol), independently of eGFR and conventional risk factors. Risk of mortality was significant at an ACR of 1·1 mg/mmol compared with 0·6 mg/mmol. Furthermore, mortality gradient conferred by ACR greater than 3·4 mg/mmol was more than two-fold within all but the lowest eGFR category. This gradient is greater than the risk between adjacent stages of chronic kidney disease based on eGFR, suggesting that albuminuria provides additional prognostic information beyond eGFR alone.
Clinical and laboratory guidelines recommend ACR as the preferred measure of albuminuria for the definition of chronic kidney disease and for assignment of disease stages.2, 10, 42 The urine dipstick test is often used for initial screening because it is less expensive and can be done at the point of care.2, 43 Our findings showed that even a trace urine protein on dipstick test was associated with an increased risk of mortality, consistent with the fact that 60% of individuals with trace protein on dipstick have microalbuminuria.44 These findings suggest that the dipstick test is useful for risk stratification despite being a less precise measure of albuminuria.
The HRs for low eGFRs (ie, <60 mL/min/1·73 m2) with all-cause and cardiovascular mortality tended to be higher in younger than in older populations. Similar findings have been reported for traditional cardiovascular risk factors.45 However, the shape of the eGFR-mortality associations was similar in participants younger and older than 65 years. Additionally, tests for interaction for eGFR and age in most studies were not significant. The HRs for eight-fold higher ACRs were quantitatively similar in participants younger and older than 65 years.
We recorded significant heterogeneity for the association between low eGFRs and all-cause mortality, but not for cardiovascular mortality. Studies with a higher proportion of black people tended to have higher HRs for all-cause mortality. Although not significant, this finding is consistent with previous studies showing that lower eGFR is more strongly associated with adverse outcomes in black people than in white people.46 For ACR, none of the baseline variables tested were significantly associated with the variation in mortality risk. Although the reasons for significant quantitative heterogeneity will need further investigation, the patterns of risk association were qualitatively similar across a wide range of studies (webappendix p 21).
The findings in this analysis are consistent with the current KDOQI thresholds (ie, eGFR <60 mL/min/1·73 m2 and ≥ACR 3·4 mg/mmol) as indicative of increased mortality risk. They also suggest inclusion of albuminuria stages in assignment of chronic kidney disease stages, independent of GFR stages. Nevertheless, the final decisions about which levels of GFR and albuminuria are used for the definition and staging of chronic kidney disease should incorporate a wide range of considerations, including disease prevalence, incidence of targeted outcomes, risk classification, and cost-effectiveness associated with management directed at each kidney measure.47-49 This approach is particularly the case for ACR, since we noted a continuous association with mortality.
This study does have some limitations. The distributed data analysis consortium we used provides superior uniformity than does a review of the literature, but falls short of having a uniform study protocol and centralised laboratories across all cohorts. Measurements of creatinine and albuminuria were not standardised in all studies. Some studies measured creatinine and albuminuria in fresh samples whereas other studies used frozen samples. Analyses required complete data for both eGFR and albuminuria. We could only test interactions between eGFR, albuminuria, and age in each cohort. Most of the participants who were black were from studies in the USA. Although some of the participating studies supported the proportional hazards assumption for mortality with eGFR or albuminuria as predictors,3, 21, 46, 50 we cannot confirm the validity of this assumption for every study. Thus, the pooled HRs we obtained estimate the average HR over follow-up time.
Thus, eGFR less than 60 mL/min/1·73 m2 and ACR 1·1 mg/mmol (10 mg/g) or more are independent predictors of all-cause mortality and cardiovascular mortality in the general population. These findings provide a quantitative basis for use of these two kidney measures for risk assessment and definition and staging of chronic kidney disease.
Results
Figure 1 shows the flow diagram of study selection for the analysis. Of the 21 studies included in the analysis, nine were from North America, six from Europe, five from Asia, and one from Australia. Characteristics of included studies are shown in table 1. Median follow-up time was 7·9 years (range 2·1-11·6). Overall, 1 234 182 participants (ACR studies, 105 872 participants; dipstick studies, 1 128 310 participants) were followed up for 5 462 687 person-years (ACR studies, 730 577 person-years; dipstick studies, 4 732 110 person-years). The median age of participants was 61 years (47-78) in studies with ACR measurements and 62 years (42-81) in studies with dipstick measurements. The distribution of participants and outcomes according to the categories of eGFR and albuminuria is shown in webappendix pp 1-5. During follow-up, there were 45 584 deaths. 15 of the 21 studies reported data for 9637 deaths from cardiovascular disease.
Pooled estimates of adjusted HRs for all-cause mortality and cardiovascular mortality according to categories of eGFR and ACR for the 14 studies with ACR data are shown in figure 2. Risk of mortality was fairly constant between eGFR 75 mL/min/1·73 m2 and 105 mL/min/1·73 m2 and increased at lower eGFRs. Compared with eGFR 95 mL/min/1·73 m2, HRs for all-cause mortality were 1·18 (95% CI 1·05-1·32) for eGFR 60 mL/min/1·73 m2, 1·57 (1·39-1·78) for 45 mL/min/1·73 m2, and 3·14 (2·39-4·13) for 15 mL/min/1·73 m2. Similar findings were seen for cardiovascular mortality (figure 2). A U-shaped association with higher risk of mortality at eGFR more than 105 mL/min/1·73 m2 was seen for all-cause mortality but was less evident for cardiovascular mortality. The U-shape was more pronounced in the studies with dipstick measurements than in the studies with ACR measurements, with the risk becoming significant at eGFR less than 45 mL/min/1·73 m2 for all-cause mortality and at eGFR 60 mL/min/1·73 m2 for cardiovascular mortality (webappendix p 18).
The relations of ACR to the risks of all-cause mortality and cardiovascular mortality were monotonic on the log-log scale. Compared with ACR 0·6 mg/mmol, HRs for all-cause mortality were 1·20 (95% CI 1·15-1·26) for ACR 1·1 mg/mmol, 1·63 (1·50-1·77) for 3·4 mg/mmol, and 2·22 (1·97-2·51) for 33·9 mg/mmol (figure 2). Similar findings were seen for cardiovascular mortality (figure 2).
The HRs for low eGFRs seemed to be higher in participants younger than 65 years than in those older than 65 years, particularly for all-cause mortality at eGFR between 30 mL/min/1·73 m2 and 60 mL/min/1·73 m2 (webappendix p 19). For example, the HRs associated with eGFR of 45 mL/min/1·73 m2 compared with 95 mL/min/1·73 m2 were 2·14 (95% CI 1·56-2·92) in participants younger than 65 years and 1·60 (1·46-1·75) in those older than 65 years. However, the shapes of the associations between eGFR and risk of all-cause mortality and cardiovascular mortality were largely similar across the two age groups (webappendix p 19). The tests for interaction between eGFR and age were not significant in most studies (webappendix p 6). Similar findings were seen for studies with dipstick measurements (webappendix p 20). By contrast, HRs per eight-fold higher ACR (eg, 4·8 mg/mmol vs 0·6 mg/mmol) were quantitatively similar in participants younger than 65 years and in those older than 65 years (1·49, 95% CI 1·40-1·59, vs 1·52, 1·45-1·61, for all-cause mortality and 1·65, 1·47-1·85, vs 1·53, 1·40-1·67, for cardiovascular mortality, respectively).
Table 2 shows pooled HRs for all-cause mortality and cardiovascular mortality for the 28 categories of eGFR and albuminuria in studies with ACR measurements. eGFR lower than 60 mL/min/1·73 m2 showed a similar association with risk of mortality across all levels of ACR and vice versa, suggesting multiplicative independent risk factors for mortality (parallel lines in figure 3). ACR higher than 3·4 mg/mmol was associated with more than two-fold mortality risk within all eGFR categories but the lowest (table 3). Compared with negative dipstick results, trace urine protein on dipstick was significantly associated with increased risk for all-cause mortality and cardiovascular mortality at eGFR 90-104 mL/min/1·73 m2 with similar association at other eGFR levels. Although HRs tended to be higher in participants younger than 65 years than in participants older than 65 years (particularly for all-cause mortality), eGFR and albuminuria were multiplicatively associated with risk of mortality in both these age groups (webappendix pp 8-11).
Although general population cohorts are not the ideal populations for studying severely reduced GFR, the risk of mortality associated with raised albuminuria seemed to be weaker in participants with eGFR less than 30 mL/min/1·73 m2 than in participants with eGFR more than 30 mL/min/1·73 m2 (table 2 and table 3). However, this difference was very subtle for all-cause mortality and 95% CIs were wide for cardiovascular mortality (figure 3). Indeed, most studies did not report significant interaction between eGFR and ACR (webappendix p 7).
The results from individual cohorts are shown in webappendix pp 12-17 and p 21. We noted moderate but significant heterogeneity in the HRs of eGFR in the studies with ACR measurements of eGFR 60 mL/min/1·73 m2 or less for all-cause mortality (I2 35-56%) but not for cardiovascular mortality. In the dipstick studies, we found large heterogeneity for all-cause mortality across the entire eGFR range (I2 ≥92%). We recorded mild heterogeneity in the HRs of ACR in the entire range tested for all-cause mortality (I2 33-40%).
We undertook meta-regression analysis for the studies with ACR measurements with variables listed in table 1 to seek potential sources of heterogeneity at eGFR 45 mL/min/1·73 m2 (vs 95 mL/min/1·73 m2) and ACR 3·4 mg/mmol (vs 0·6 mg/mmol) for all-cause mortality. These two levels were selected because of their clinical relevance. Only the proportion of black people was borderline significant (p=0·064) and positively associated with variation of log-HR at eGFR 45 mL/min/1·73 m2 (webappendix p 22). For ACR 3·4 mg/mmol, none of the variables tested was significantly associated with log-HR for all-cause mortality across studies (data not shown). We undertook an Egger test for these two points (eGFR 45 mL/min/1·73 m2 and ACR 3·4 mg/mmol), with results showing no underlying publication bias (p=0·078 and p=0·264, respectively).
Methods
Search strategy and selection criteria
We searched PubMed on Aug 6, 2009, for studies published between 1966 and July, 2009, using the following combination of terms: (eGFR OR GFR OR glomerular filtration rate OR kidney function OR renal function) AND (albuminuria OR albumin to creatinine ratio OR ACR OR urinary albumin concentration OR UAC OR dipstick) AND (mortality OR ESRD OR end stage renal disease OR progressive chronic kidney disease OR acute kidney injury) AND (adult[MeSH]) AND (Humans[MeSH]). No language restrictions were applied. The search was done independently by two investigators (PEdJ and RTG). Any study that was judged relevant on the basis of its title was retrieved in abstract form, and if relevant, in full-text form. Disagreement about eligibility was resolved by discussion.
We extended our search by reviewing references from retrieved articles. Further studies and unpublished data were sought by discussion between collaborators, nephrologists, and cardiovascular disease epidemiologists. Additionally, studies were encouraged to join the Consortium through a published position statement of KDOQI and KDIGO17 and the KDIGO website.
To be included in the meta-analysis for this report, the study had to have at least 1000 participants selected from a general population, information at baseline about eGFR and urine albumin concentrations, and either of our two study outcomes, all-cause mortality or cardiovascular mortality, with a minimum of 50 events. As recommended in clinical guidelines,2, 10 we preferred urine ACR as the measure of albuminuria. However, we also accepted urine albumin excretion as well as a qualitative measurement by use of a dipstick test, since a positive result is mainly caused by increased albumin excretion and in some settings dipstick screening is widely used.2 Studies that selected participants on the basis of cardiovascular disease or risk factors for cardiovascular disease were excluded.
Study variables
We used the Modification of Diet in Renal Disease (MDRD) Study equation to estimate GFR from age, sex, ethnic origin, and serum creatinine concentration.18 History of cardiovascular disease was defined as previous myocardial infarction, coronary revascularisation, heart failure, or stroke. Hypertension was defined as systolic blood pressure 140 mm Hg or more, diastolic blood pressure 90 mm Hg or more, or use of antihypertensive medication. Hypercholesterolaemia was defined as total cholesterol concentration 5·0 mmol/L or more in patients with a history of cardiovascular disease and as 6·0 mmol/L or more in patients without history of cardiovascular disease. Diabetes mellitus was defined as fasting glucose concentration 7·0 mmol/L or more, non-fasting glucose concentration 11·1 mmol/L or more, or use of glucose-lowering drugs or self-reported diabetes. Smoking was dichotomised to current smokers versus former or non-smokers. Cardiovascular mortality was defined as death due to myocardial infarction, heart failure, or stroke, or sudden cardiac death. We selected cardiovascular mortality as a specific cause of death since it is the leading cause of death in individuals with chronic kidney disease.7
Statistical analysis
To increase uniformity and reduce bias, investigators from each study analysed their data in accordance with an a priori analytical plan using standard computer programs. The distributed data analysis overview and analytical notes for individual studies are described in webappendix p 24. All analyses were done with Stata version 10 or 11, SAS version 9, or R version 2.9.2. Cox proportional hazards models were used to estimate the hazard ratios (HRs) of all-cause mortality and cardiovascular mortality associated with eGFR and albuminuria, adjusted for age, sex, ethnic origin (black vs non-black), history of cardiovascular disease, systolic blood pressure (continuous), diabetes, concentration of serum total cholesterol (continuous), and smoking.
First, we assessed the independent association between eGFR and albuminuria as continuous variables and risk of mortality. We modelled eGFR and ACR using linear splines with knots at 45 mL/min/1·73 m2, 60 mL/min/1·73 m2, 75 mL/min/1·73 m2, 90 mL/min/1·73 m2, and 105 mL/min/1·73 m2, and at 1·1 mg/mmol, 3·4 mg/mmol, and 33·9 mg/mmol (to convert to mg/g divide by 0·113), respectively. eGFR 95 mL/min/1·73 m2 and ACR 0·6 mg/mmol were treated as reference points. Subgroup analyses divided participants into groups split at age 65 years. Interaction between eGFR and age was assessed by likelihood ratio tests in individual studies (see webappendix p 26 for details of statistical model).
Subsequently, we investigated the joint association between eGFR and albuminuria with risk of mortality in several analyses. We compared the risk in 32 categories of eGFR (<15, 15-29, 30-44, 45-59, 60-74, 75-89, 90-104, ≥105 mL/min/1·73 m2) and albuminuria (ACR <1·1, 1·1-3·3, 3·4-33·8, ≥33·9 mg/mmol; dipstick test result: negative, trace urine protein, 1+, 2+ or more). The category with eGFR 90-104 mL/min/1·73 m2 and the lowest albuminuria was used as the reference group. The interaction between eGFR and albuminuria was tested by likelihood ratio tests between the models incorporating spline eGFR and categorical albuminuria (ACR <3·4, 3·4-33·8, ≥33·9 mg/mmol; dipstick test result: negative or trace, 1+, and 2+ or more) with and without their interaction terms. On the basis of the model with the interaction terms, we estimated HRs at 23 points determined by the combination of eGFR (15, 45, 60, 75, 90, 95, 105, 120 mL/min/1·73 m2) and three categories of albuminuria compared with the reference of eGFR 95 mL/min/1·73 m2 plus ACR less than 3·4 mg/mmol or dipstick test result of negative or trace urine protein.
Pooled estimates of the HR and 95% CI were obtained from a random effects meta-analysis. Heterogeneity was estimated by use of the χ2 test for heterogeneity and the I2 statistic.19 Meta-analyses were done separately for studies with ACR and dipstick measurements. Since there were few participants (0·1%) with eGFR less than 15 mL/min/1·73 m2, we only reported results from participants with eGFR 15 mL/min/1·73 m2 or more. We did meta-regression analysis with a random-effects model to explore sources of heterogeneity. To test for publication bias, we created a funnel plot and undertook the Egger test.19 In all analyses, a p value of less than 0·05 was deemed significant.
Role of the funding source
The KDIGO planning committee and National Kidney Foundation staff participated in study design and data collection. The analytical team had full access to all the analyses done for each study and the writing committee had final responsibility for the decision to submit for publication informed by discussions with the collaborators.
| |
| |
| |
|
|
|