 |
 |
 |
| |
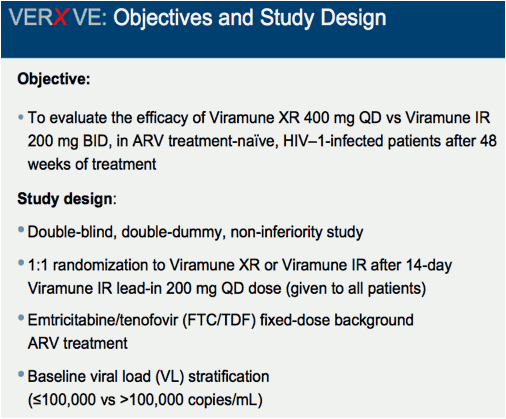
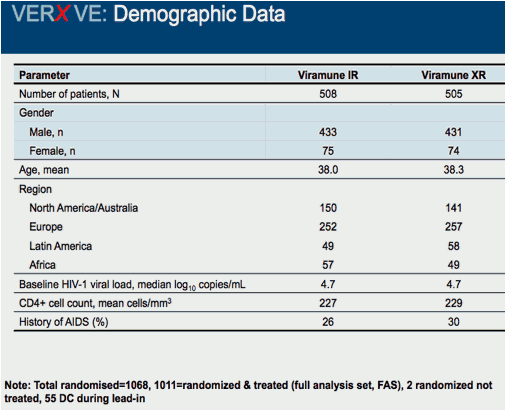
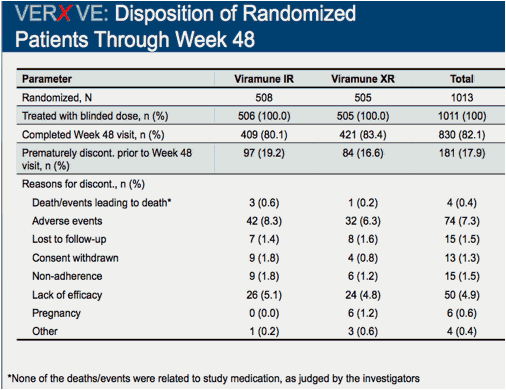
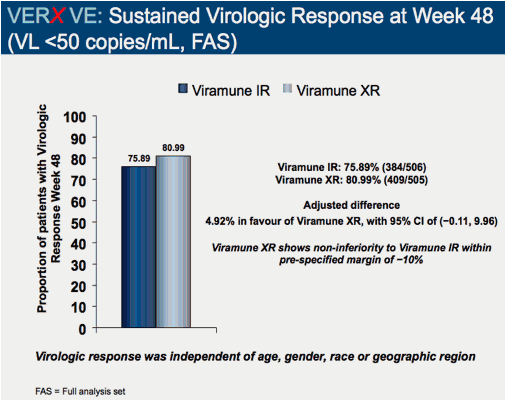
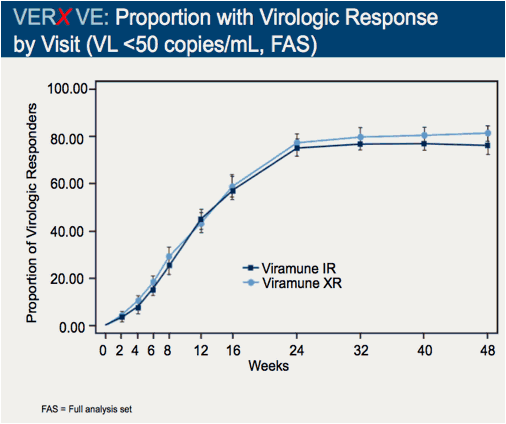
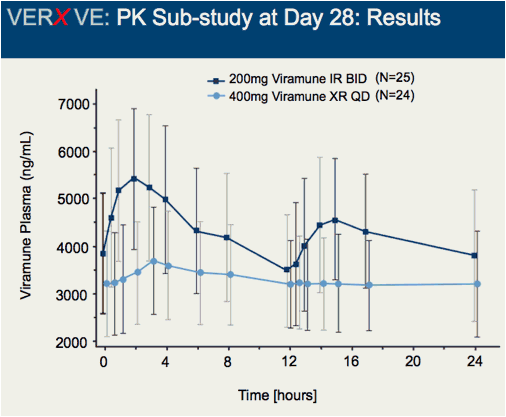
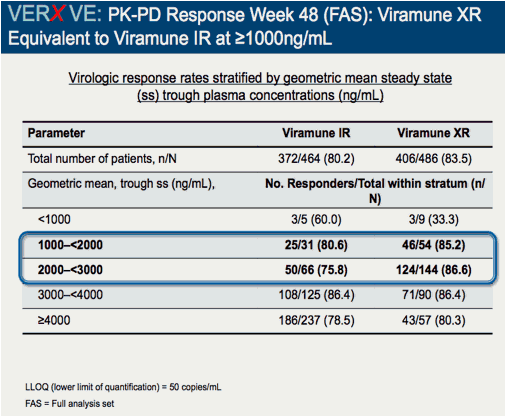
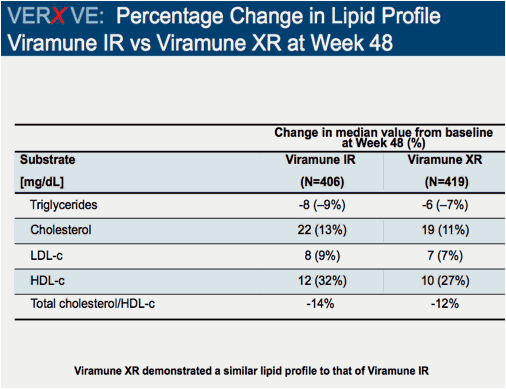
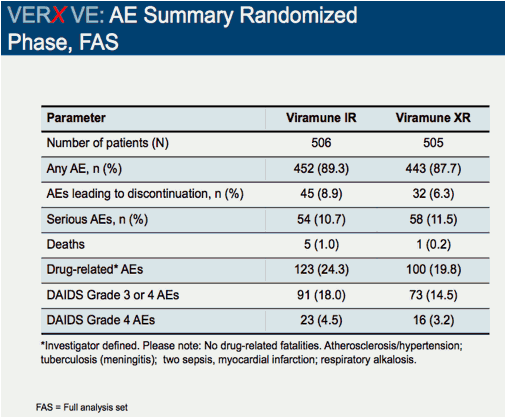
Comparison of 48 week efficacy and safety of 400mg QD nevirapine (NVP) extended release formulation (Viramune XR) versus 200mg BID nevirapine immediate release formulation (Viramune IR) in combination with emtricitabine/tenofovir in antiretroviral (ARV) naïve HIV-1 infected patients (VERxVE)
|
| |
| |
Reported by Jules Levin
18th Intl AIDS Conference July 18-23 Vienna Austria
J. Gathe, JR. Bogner, S. Santiago, A. Horban, M. Nelson, P. Cahn, J. Andrade, D. Spencer, C. Yong, T. Nguyen, W. Zhang, M. Drulak and A. Quinson*
*Boehringer Ingelheim Pharmaceuticals Inc, Ridgefield, CT, USA




|
| |
|
 |
 |
|
|