 |
 |
 |
| |
Metabolic Marker Changes Similar After 12 Weeks of Darunavir and Atazanavir
|
| |
| |
XVIII International AIDS Conference, July 18-23, 2010, Vienna
Mark Mascolini
IAC: METABOLIK (Metabolic Evaluation in Treatment-naïves Assessing the impact of two BOosted protease inhibitors on LIpids and other marKers): Comparison of the Metabolic Effects of Darunavir/ritonavir versus Atazanavir/ritonavir over 12Weeks - (07/22/10)
Changes in lipids, glucose, and other metabolic measures were similar in people starting darunavir/ritonavir versus atazanavir/ritonavir after 12 weeks in a small randomized study in the United States [1]. Twelve-week virologic and CD4 responses were also similar in the two groups.
The METABOLIK trial enrolled 65 antiretroviral-naive adults with a viral load above 1000 copies and any CD4 count to once-daily darunavir/ritonavir or atazanavir/ritonavir, each with tenofovir/emtricitabine. Everyone had virus sensitive to study drugs when the trial began, and no one took lipid-lowering agents from at least a month before the trial through week 12.
The darunavir and atazanavir groups differed substantially in several pretreatment variables: The darunavir group had a higher proportion of whites and a lower proportion of blacks (62% and 38%) than the atazanavir group (39% and 55%). The darunavir group had more advanced HIV infection: Median pretreatment CD4 count stood at 267 (range 10 to 532) in the darunavir arm and at 316 (range 39 to 813) in the atazanavir arm. Median viral loads were 137,000 copies (range 642 to 2,450,000) with darunavir and 46,100 (range 397 to 637,000) with atazanavir. (The investigators did not calculate the possible statistical significance of these differences.)
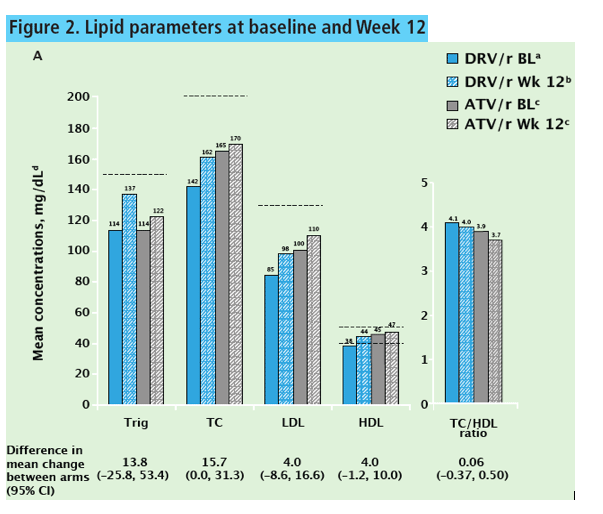
Because of dropouts and protocol violations, the 12-week metabolic parameter analysis involved 28 people taking darunavir and 27 taking atazanavir. Through the first 12 weeks, average triglycerides rose from 114 to 137 mg/dL in the darunavir group and from 114 to 122 mg/dL in the atazanavir group. Average low-density lipoprotein cholesterol climbed from 85 to 98 mg/dL in the darunavir group and from 100 to 110 mg/dL in the atazanavir group. Week-12 averages of these lipids in both groups remained below National Cholesterol Education Program (NCEP) thresholds for starting antilipid therapy. The ratio of total cholesterol to high-density lipoprotein cholesterol hardly changed over 12 weeks with darunavir (4.1 to 4.0) or atazanavir (3.9 to 3.7). from Jules: total cholesterol increased on DRV from 142 to 162 and on ATV from 165 to 170; HDL went up on on DRV from 38 to 44, on ATV from 45 to 47.
The investigators detected no clinically relevant changes in fasting glucose, fasting insulin, insulin sensitivity, or creatinine clearance through week 12 in either study group. Levels of tumor necrosis factor-alpha, an inflammation marker, fell from an average 4207 to 2751 pg/mL (a 35% drop) in the darunavir arm, and from 2957 to 2395 pg/mL in the atazanavir group (a 19% drop).
People randomized to darunavir started treatment with a viral load averaging 5.0 log copies (100,000 copies) and had a 3-log (1000-fold) decline in the first 12 weeks. People randomized to atazanavir started the study averaging 4.6 log copies (40,000 copies) and had a 12-week 2.6-log (400-fold) drop. Average CD4 counts rose from 268 to 379 with darunavir and from 327 to 395 with atazanavir.
The investigators noted that the metabolic changes recorded with darunavir in this trial reflect those recorded in the ARTEMIS study of treatment-naive people [2]. They added that lipid and glucose changes with darunavir and atazanavir generally matched those seen in healthy HIV-negative volunteers taking these protease inhibitors [3]. The researchers plan longer follow-up to detect possible long-term changes.
References
1. Aberg J, Overton T, Gupta S, et al. METABOLIK (metabolic evaluation in treatment-naives assessing the impact of two boosted protease inhibitors on lipids and other markers): comparison of the metabolic effects of darunavir/ritonavir versus atazanavir/ritonavir over 12 weeks. XVIII International AIDS Conference. July 18-23, 2010. Vienna. Abstract WEPE0111.
2. Mills AM, Nelson M, Jayaweera D, et al. Once-daily darunavir/ritonavir vs. lopinavir/ritonavir in treatment-naive, HIV-1-infected patients: 96-week analysis. AIDS. 2009;23:1679-1688.
3. Tomaka F, Lefebvre E, Sekar V, et al. Effects of ritonavir-boosted darunavir vs. ritonavir-boosted atazanavir on lipid and glucose parameters in HIV-negative, healthy volunteers. HIV Med. 200;10:318-327.
|
| |
|
 |
 |
|
|