 |
 |
 |
| |
Pharmacokinetic interaction study between TMC278, a next-generation NNRTI, and methadone
|
| |
| |
Reported by Jules Levin
11th International Workshop on Clinical Pharmacology of HIV Therapy, Sorrento, Italy, 7-9 April 2010
HM Crauwels,1 RPG van Heeswijk,1 A Vandevoorde,1 DF McNeeley,2 A Buelens,1 K Boven,2 RMW Hoetelmans1
1Tibotec BVBA, Mechelen, Belgium; 2Tibotec Inc., Titusville, NJ, USA

ABSTRACT
Introduction
TMC278 (rilpivirine) is a next-generation NNRTI with sustained efficacy and good tolerability through 96 weeks in antiretroviral (ARV)-naïve, HIV-infected patients.1 TMC278 is currently being investigated in Phase III trials at a dose of 25 mg qd, in combination with a background regimen of two NRTIs. The current trial evaluated the effect of TMC278 on the pharmacokinetics and pharmacodynamics of methadone.
Methods
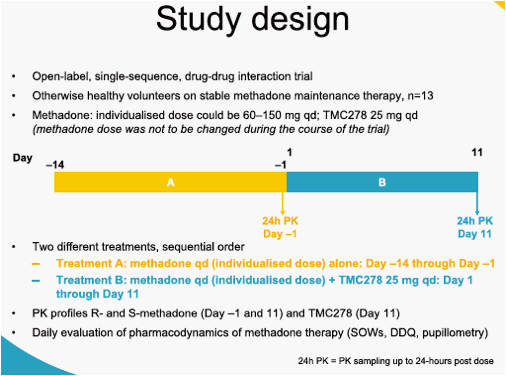
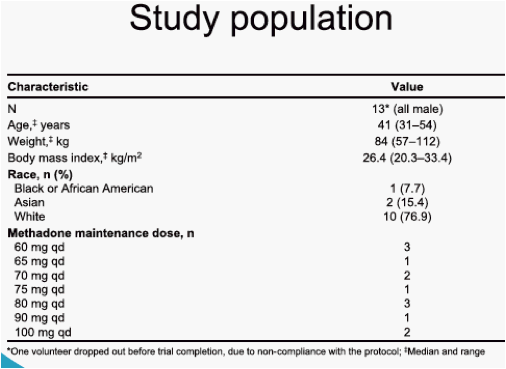
This was an open-label, single-sequence, add-on drug-drug interaction trial in 13 HIV-negative volunteers who were on stable methadone maintenance therapy. Volunteers received methadone (individualised stable methadone maintenance dose, between 60 mg and 150 mg qd) from Day -14 through Day 11, and TMC278 25 mg qd was added from Day 1 through Day 11. All intakes were witnessed, and all treatments were taken following breakfast. Pharmacokinetic (PK) profiles up to 24 hours after intake were determined on Day -1 (methadone alone) and on Day 11 (methadone + TMC278) for R- and S-methadone, and on Day 11 for TMC278. Plasma samples were analysed using validated liquid chromatography-mass spectrometry/mass spectrometry (LC-MS/MS) methods. PK parameters were calculated using non-compartmental analysis. The least square means (LSM) ratios (test/reference) and associated 90% confidence intervals (CI) were calculated based on log-transformed PK parameters. Pharmacodynamic assessments of symptoms of methadone withdrawal (Short Opiate Withdrawal Scale [SOWS], Desires for Drugs Questionnaire [DDQ], pupillometry) were performed on Day -7 and daily from Day -3 until Day 11, within 2 hours before the intake of methadone.
Results
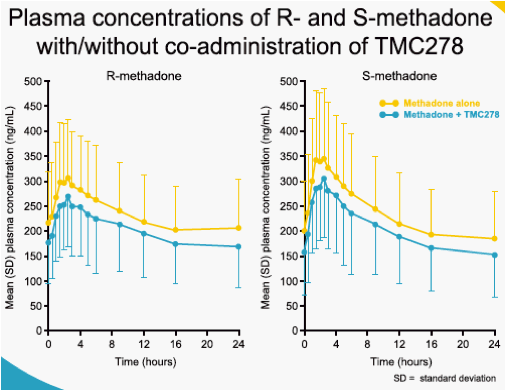
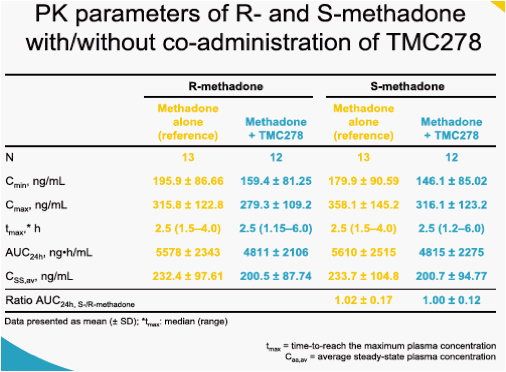
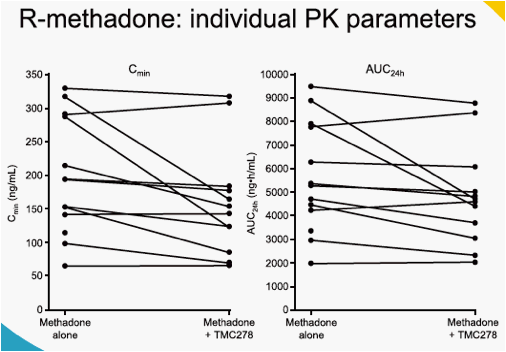
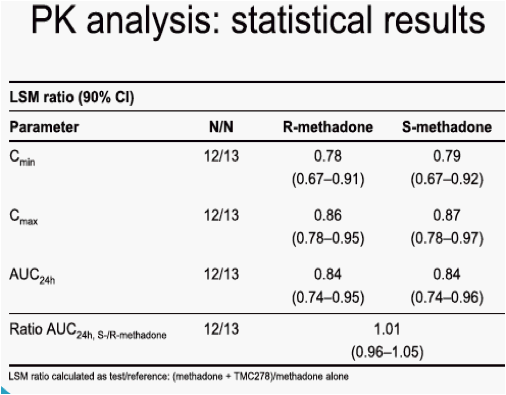
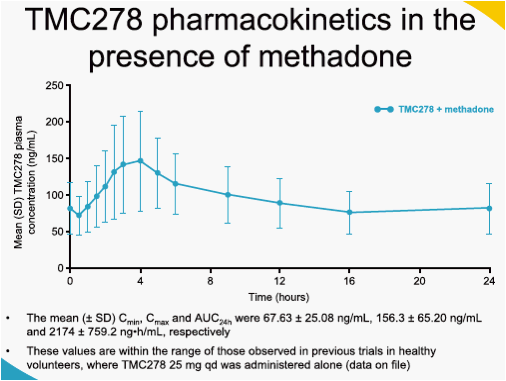
When TMC278 25 mg qd was added onto a stable methadone maintenance therapy, the mean minimum plasma concentration (Cmin), maximum plasma concentration (Cmax) and area under the plasma concentration-time curve from time of administration to 24-hours after dosing (AUC24h) of the biologically active R-isomer of methadone decreased by 22% (LSM ratio 0.78; 90% CI: 0.67-0.91), 14% (0.86; 0.78-0.95) and 16% (0.84; 0.74-0.95), respectively, as compared with administration of methadone alone. The mean Cmin, Cmax and AUC24h of the inactive S-methadone decreased by 21% (0.79; 0.67-0.92), 13% (0.87; 0.78-0.97) and 16% (0.84; 0.74-0.96), respectively. The AUC ratio for S-/R-methadone was similar during co-administration with TMC278 or when methadone was administered alone (1.01; 0.96-1.05). The exposure to TMC278 in the presence of methadone was within the expected range. During co-administration with TMC278, no clinically relevant changes in the pharmacodynamic assessments of methadone withdrawal symptoms (SOWS, DDQ scores, pupil dilation) were observed. Co-administration of methadone and TMC278 was generally safe and well tolerated. No grade 3 or 4 adverse events (AEs) and no serious AEs were reported. There were no discontinuations due to AEs.
Conclusions
No a-priori adjustment of the methadone dosage is required when initiating co-administration with TMC278. Clinical monitoring for withdrawal symptoms is, however, recommended, as methadone maintenance therapy may need to be adjusted in some patients.




REFERENCES
1. Pozniak A, et al. AIDS 2010;24:55-65.
2. Azijn H, et al. AAC 2010;54:718-27.
|
| |
|
 |
 |
|
|