 |
 |
 |
| |
A Finite Course of ART during Early HIV-1 Infection Modestly Delays Need for Subsequent ART Initiation: ACTG A5217, the SETPOINT Study
|
| |
| |
CROI 2010
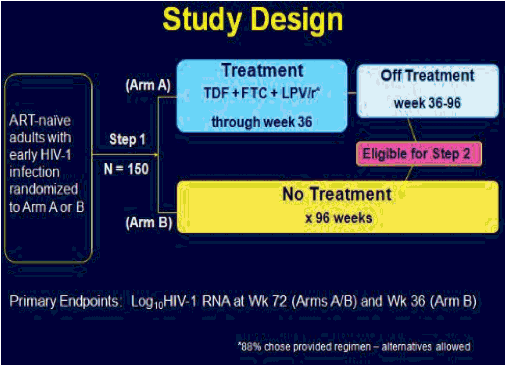
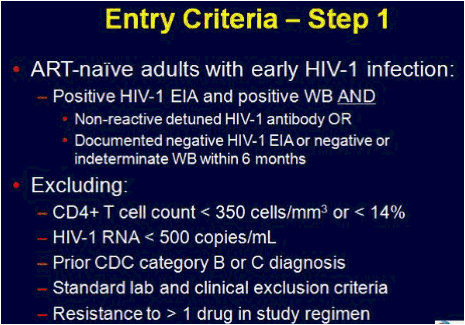
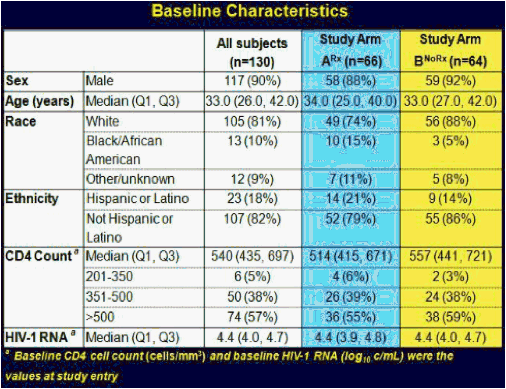
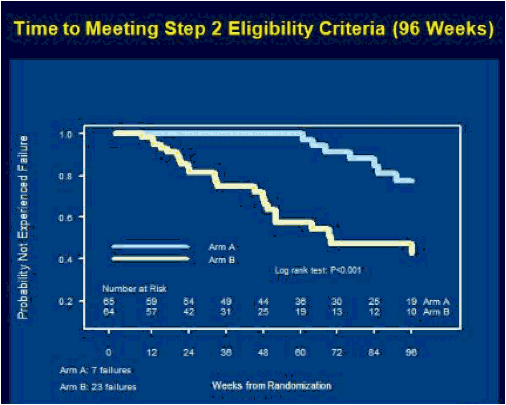
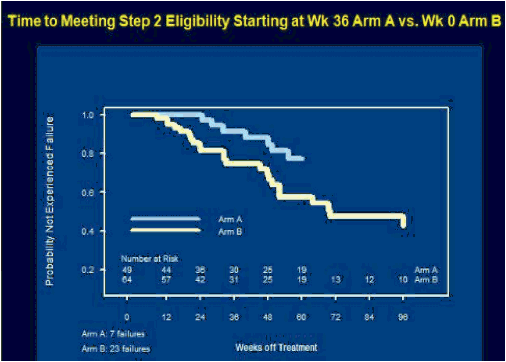
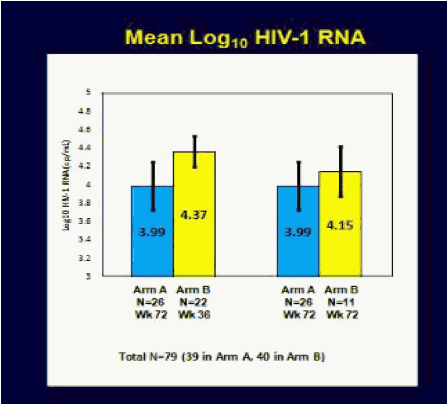
ACTG 5217, the SETPOINT study, compared a 36 week course of ARV therapy with tenofovir (TDF), emtricitabine (FTC) and lopinavir/ritonavir (LPV/r) followed by treatment interruption versus no therapy in patients with HIV infection within the previous six months, testing the hypothesis that early therapy would modify the viral load set point compared to no therapy and forestall the need to resume treatment [C Hogan, et al. Abst. 134]. The primary endpoint of the study was the viral load set point at week 72, 36 weeks after stopping therapy in the treatment arm. Subjects in either arm could start or resume therapy if they experienced a CD4 <350 cells/mm3 at or after week 12, <200 cells/mm3 at any point, an HIV RNA >750,000 copies/mL at or after week 4 or >200,000 copies/mL at or after week 12, or symptomatic HIV infection. 130 subjects were enrolled in the study, with a median CD4 of 540 cells/mm3 and a median HIV RNA of 4.4 log10 copies/mL. Following a DSMB review in June, 2009, the study was stopped because of a higher rate of disease progression in the no treatment arm. At the time of the DSMB review, 79 subjects had completed 72 weeks of study. By week 36, 27.5% of subjects in the no treatment arm reached criteria to start therapy, and by week 72 50% had met criteria for therapy. In contrast, only 10% of subjects in the early treatment group met criteria to restart therapy. The study was stopped due to futility - so many subjects in the no treatment arm had started therapy that the primary question of differences in viral load setpoint could not be determined. By comparing the time to resuming therapy for the early treatment after week 36 to the time to initiate therapy for the no treatment group from week 0, there was a delay in the time to resumption of therapy of 18 weeks that persisted over a 60 week period of time. Therefore, early treatment produced a marginal benefit compared to no treatment. The viral load set point 36 weeks after treatment discontinuation for the early treatment group (week 72) compared to week 36 for the no treatment group was 3.99 log10 copies/mL vs. 4.37 log10 copies/mL, a difference that was not statistically significant. So, early treatment followed by interruption modestly delayed the need for subsequent treatment, and therefore, there may be a slight advantage to early treatment of patients with acute infection. But the real story of this trial is that half of the people not initially started on therapy met criteria for starting therapy within the first 72 weeks of follow-up. Thus, early progression of HIV infection may be more rapid than we appreciate.
----The 17th Conference on Retroviruses and Opportunistic Infections. ARV Therapies and Therapeutic Strategies A CME Newsletter. Jointly Sponsored by Postgraduate Institute for Medicine and ViralEd, LLC
Christine Hogan*1,2, V DeGruttola3, E Daar4, X Sun3, C Del Rio5, S Fiscus6, T Frazier7, B Hare8, M Markowitz9, S Little10, and the A5217 Study Team
1Med Coll of Wisconsin, Milwaukee, US; 2Columbia Univ, Coll of Physicians and Surgeons, New York, NY, US; 3Harvard Sch of Publ Hlth, Boston, MA, US; 4Harbor-UCLA Med Ctr, Torrance, US; 5Emory Univ, Atlanta, GA, US; 6Univ of North Carolina at Chapel Hill Sch of Med, US; 7NIAID, NIH, Bethesda, MD, US; 8Univ of California, San Francisco, US; 9Aaron Diamond AIDS Res Ctr, The Rockefeller Univ, New York, NY, US; and 10Univ of California, San Diego, US
Background: Initiation of ART during early HIV-1 infection may preserve HIV-1-specific immune responses that could enhance virologic control upon treatment discontinuation.
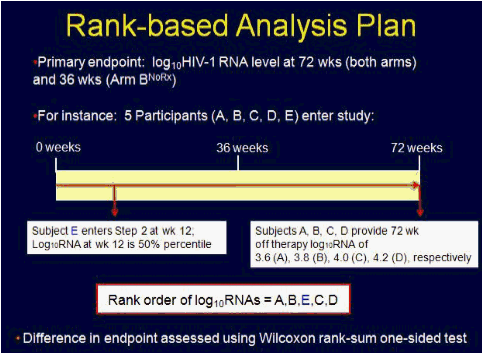
Methods: We conducted a prospective, randomized-controlled trial to assess the effect of 36 weeks of ART given during early HIV-1 infection on virologic set point at week 72. The study enrolled recently infected adults-newly seropositive or detuned-EIA consistent with infection <6 months' duration. Subjects were randomized 1:1 in step 1 to receive 36 weeks of ART and then discontinue treatment (arm A) vs no therapy (arm B). Subjects were advised to start ART (step 2) if they met disease progression criteria consistent with current treatment guidelines. Primary endpoint was a composite of meeting criteria for step 2 and log10 HIV-1 RNA at week 72 (arms A and B) and at week 36 (arm B). Difference in viral set point between the arms was assessed using the Wilcoxon rank-sum 1-sided test; those meeting criteria for step 2 were considered "failures" and assigned an unfavorable rank. Time-to-failure was assessed via KM plot and log-rank test.
Results: At the June 2009 Data and Safety Monitoring Board (DSMB) interim review, 130 of 150 subjects had been enrolled, 52 still on study. Baseline characteristics: 90% male, median age 33 years, CD4 count 540 cells/mm3 and HIV-1 RNA 4.4 log10 copies/mL. The DSMB recommended stopping the study because of a significant advantage for arm A by week 76, driven mostly by a higher rate of progression to Step 2 eligibility criteria in arm B, and their expectation that further enrollment would not yield additional conclusions. Efficacy analysis included 79 subjects randomized at least 72 weeks prior to the DSMB review. Arm A had a better outcome on the primary endpoint (P =0.005) at week 72, as well as at week 72 (Arm A) vs week 36 (arm B) (P =0.002), due primarily to the higher rate of progression in arm B-4 of 66 (6%) on arm A vs 22 of 64 (34%) on arm B met step 2 eligibility criteria by week 76, 13 (20%) on arm B by week 36. Time-to-progression was significantly shorter in arm B vs A, whether the time origin was study entry for both arms (P <0.001) or time of scheduled discontinuation of ART for arm A (P =0.035).
Conclusions: Progression to meeting criteria for initiation of ART occurred more frequently than anticipated, limiting our ability to evaluate HIV-1 RNA levels at study endpoint. However, a limited period of ART during early HIV-1 infection modestly delayed the need for subsequent initiation of ART.











|
| |
|
 |
 |
|
|