 |
 |
 |
| |
Structural Models of HIV-1 Integrase and DNA in Complex with S/GSK1349572, Raltegravir and Elvitegravir: Structure-based Rationale for INI Resistance Profiles
|
| |
| |
Reported by Jules Levin
International Drug Resistance Workshop, 8-12 June 2010, Dubrovnik, Croatia
F DeAnda1, K Hattori2, T Yoshinaga2, T Kawasuji2, MR Underwood1 1GlaxoSmithKline, Inc., RTP, NC, USA; 2Shionogi & Co., Ltd, Osaka, Japan



ABSTRACT
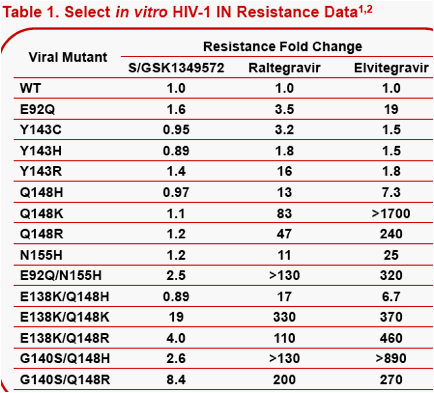
Background: The in vitroHIV-1 integrase (IN) resistance profile of S/GSK1349572 is distinct from that of raltegravir and elvitegravir. For example, while signature IN mutations at Y143, Q148 and N155 negatively affect raltegravir and elvitegravir antiviral potencies, S/GSK1349572 potency is unaffected. Herein we describe structural models of HIV-1 IN with DNA, constructed to provide a structure-based rationale for INI resistance profiles.
Methods: An HIV-1 IN model was constructed from IN structural fragments available from the RCSB Protein Data Bank (www.pdb.org), and included the N-terminal domain in 1k6y (PDB code), catalytic core domain in 2b4j, and C-terminal domain in 1ex4. The IN active site loop was modeled from prototype foamy virus (PFV) integrase in 3l2u. Two HIV-1 U5 LTR ends were also modeled from the PFV DNA in 3l2u and 3l2v; these were used to construct two distinct HIV-1 IN/DNA models. Raltegravir, elvitegravir and S/GSK1349572 were manually docked into the catalytic sites and their poses geometry-optimized.
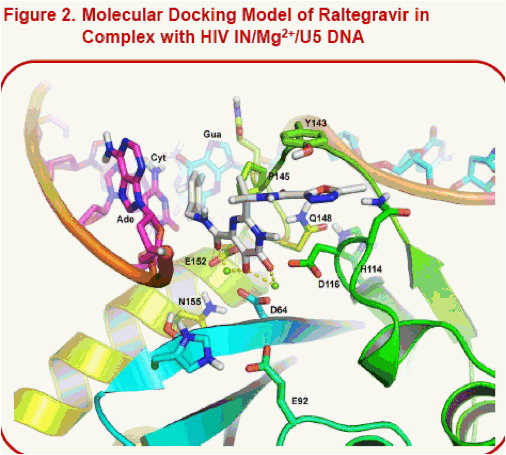
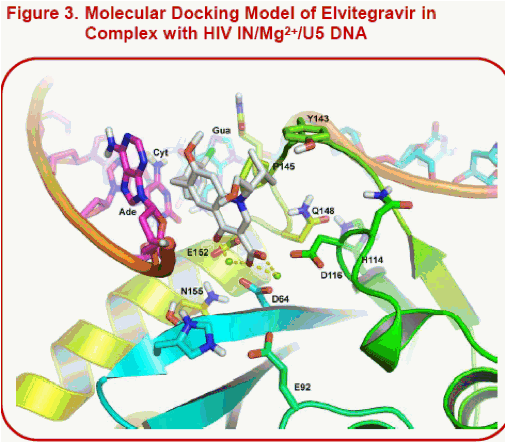
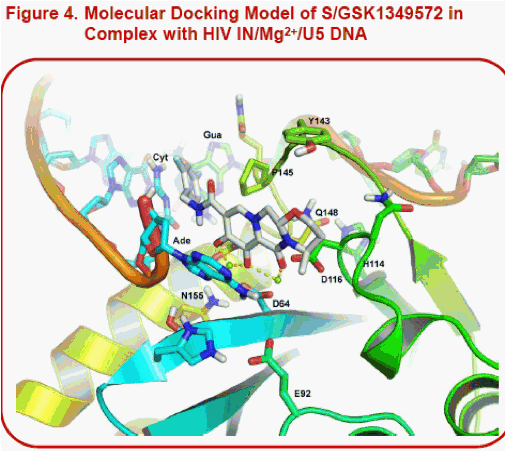
Results: Unlike raltegravir and elvitegravir, the docked pose of S/GSK1349572 shows that the inhibitor spans the width of the catalytic site from G118 to E152, but not its height from the two Mg2+to Y143 at the top of the pocket. Indeed, raltegravir's oxadiazole pi-stacks with Y143. In contrast, S/GSK1349572 lies distal to and does not interact with Y143, which may explain its activity against HIV with Y143 resistance substitutions. Our models suggest that Q148 may stabilize the active site loop in a catalytically-active conformation by forming an H-bond with H114. We hypothesize that substitutions at Q148 may disrupt the loop, potentially reducing the pocket's height and thus negatively affecting raltegravir and elvitegravir's potencies but not S/GSK1349572's potency. The N155H substitution may also reduce the pocket's height since it lies just beneath the catalytic site.
Conclusions: Comparison of the docked poses of S/GSK1349572, raltegravir and elvitegravir revealed clear differences in intermolecular interactions. S/GSK1349572's more streamlined architecture may contribute to its resistance to substitutions which decrease the catalytic pocket's height. Our HIV-1 IN/DNA models provide a structure-based rationale for S/GSK1349572's distinctive in vitroresistance profile and highlight its potential for clinical activity against raltegravir-resistant viruses.
Introduction
The integrase inhibitor, S/GSK1349572, has significant anti-HIV activity against not only wild type virus, but also numerous raltegravir-resistant mutants, which gives it a distinctive in vitroIN resistance profile that sets it apart from raltegravir and elvitegravir. Indeed, while signature IN mutations at Y143, Q148 and N155 negatively affect raltegravir and/or elvitegravir antiviral potencies, that of S/GSK1349572 is unaffected as reported by Seki, et al. and Sato, et al. (Table 1).1,2Herein, we describe structural models of HIV-1 IN complexed with viral DNA, which were constructed from available crystallographic data on HIV-1 and PFV IN. S/GSK1349572, raltegravir and elvitegravir were docked into the catalytic sites of these IN/DNA complexes, all in efforts to provide a structure-based rationale for the observed IN resistance profiles.

RESULTS
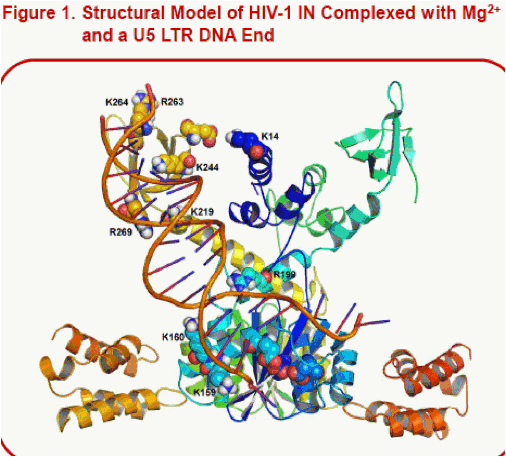
·Highlighted in Fig. 1 are nine basic residues from HIV-1 IN identified by Z. Zhao et al. to interact with the viral DNA substrate; these were identified through an experimental strategy that combined site-specific, protein-DNA cross-linking through disulfide bridging and MS protein footprinting. Based on structural data from 1ex4 and 1k6y , Z. Zhao et al. reported an IN/DNA structural model that identified a DNA binding channel brought to light by their findings.11Our HIV-1 IN/U5 DNA model is similar to that of Z. Zhao et al. and is also consistent with their biochemical results.
·Note the alternate conformations for the 3' terminal adenosine in Fig. 2 and 3 versus Fig. 4. The PFV IN/DNA//RAL complexes in 3l2t (involving Mg2+) and 3l2v (involving Mn2+) revealed two alternate conformations for this nucleotide.
References
1.Seki T., et al., 2010, 17thConference on Retroviruses and Opportunistic Infections, Poster 555.
2.Sato A., et al., 2010, 8thEuropean Drug Resistance Workshop, Poster 29.
3.Berman, H. M.; Westbrook, J.; Feng, Z., Gilliland, G.; Bhat, T. N.; Weissig, H.; Shindyalov, I. N.; Bourne, P. E. NucleicAcids Res. 2000, 28, 235-242.
4.Chen, J. C.; Krucinski, J.; Miercki, L. J.; Finer-Moore, J. S.; Tang, A. H.; Leavitt, A. D.; Stroud, R. M. Proc. Natl. Acad. Sci. USA2000, 97, 8233-8238.
5.Cherepanov, P.; Ambrosio, A. L.; Rahman, S.; Ellenberger, T.; Engelman, A. Proc. Natl. Acad. Sci. USA2005, 102, 17308-17313.
6.Wang, J. Y.; Ling, H.; Yang, W.; Craigie, R. EMBO J. 2001, 20, 7333-7343.
7.Hare, S.; Gupta, S. S.; Valkov, E.; Engelman, A.; Cherepanov, P. Nature 2010, 464, 232-236.
8.Millard, H. L. In Practical Application of Computer-Aided Drug Design; Charifson, P. S., Ed.; Marcel Dekker, 1997; p 243.
9.Insight II Molecular Modeling System, Version 2005, Accelrys, Inc., San Diego, CA, 2005.
10.Maestro, Version 9.0, Schrödinger, LLC., New York, NY, 2010.
11.Zhao, Z.; McKee, C. J.; Kessl, J. J.; Santos, W. L.; Daigle, J. E.; Engelman, A.; Verdine, G.; Kvaratskhelia, M. J. Biol. Chem. 2008, 283, 5632-5641.
|
| |
|
 |
 |
|
|