 |
 |
 |
| |
METABOLIK: Week 48 Comparison of Metabolic Parameters and Biomarkers in Subjects Receiving Darunavir/ritonavir or Atazanavir/ritonavir
|
| |
| |
Reported by Jules Levin
Glasgow Nov 2 2010
Turner Overton,1 Judith Aberg,2 Samir Gupta,3 Robert Ryan,4 Bryan Baugh,5 Guy De La Rosa5
1Washington University School of Medicine, St. Louis, MO, USA; 2Bellevue Hospital Center at New York University, New York, NY, USA;
3Indiana University School of Medicine, Indianapolis, IN, USA; 4Tibotec Inc., Titusville, NJ, USA; 5Tibotec Therapeutics, Titusville, NJ, USA


INTRODUCTION
As life expectancy of HIV-1-infected patients increases due to improved drug efficacy, metabolic effects of antiretroviral (ARV) agents are of expanding interest
ARV agents, including protease inhibitors (PIs), may additionally contribute to metabolic complications and increased cardiovascular risk associated with HIV infection1
A previous study has suggested that darunavir has a favorable lipid profile in treatment-naïve subjects2
In the TMC114-C159 trial, the effects of darunavir boosted with low-dose ritonavir (DRV/r) on lipids, fasting glucose and fasting insulin were shown to be comparable with those of atazanavir boosted with low-dose ritonavir (ATV/r) in HIV-negative volunteers3
DRV/r and ATV/r are listed as components of the preferred PI-containing regimens in the 2009 U.S. Department of Health and Human Services HIV Treatment Guidelines and the 2010 International AIDS Society-USA
recommendations for ARV treatment4,5
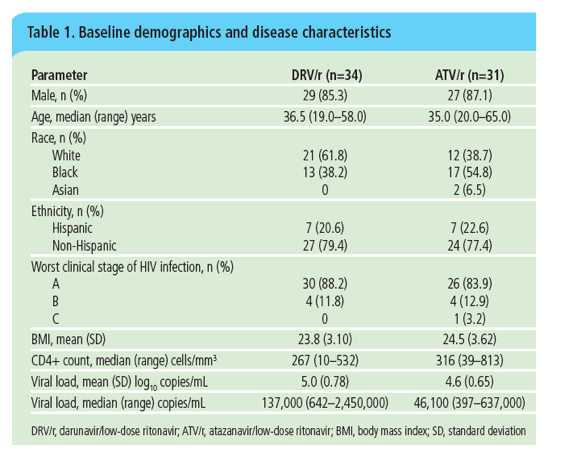
This poster presents the Week 48 results, including the Week 12 primary analysis, of METABOLIK (Metabolic Evaluation in Treatment-naïves Assessing the impact of two BOosted protease inhibitors on LIpids and other
marKers), a study evaluating the metabolic outcomes, efficacy and safety of DRV/r-based therapy compared with those of ATV/r-based therapy in treatment naïve, HIV-1-infected adults


|
| |
|
 |
 |
|
|