 |
 |
 |
| |
Metabolic Risk Factors in Responders to Tesamorelin, a Growth Hormone-Releasing Factor (GRF) Analogue, in HIV-Infected Patients with Excess Abdominal Fat
|
| |
| |
Reported by Jules Levin
13th International Workshop on Adverse Drug Reactions
and Co-morbidities in HIV
Rome, Italy
July 14-16, 2011
AUTHOR CONCLUSIONS
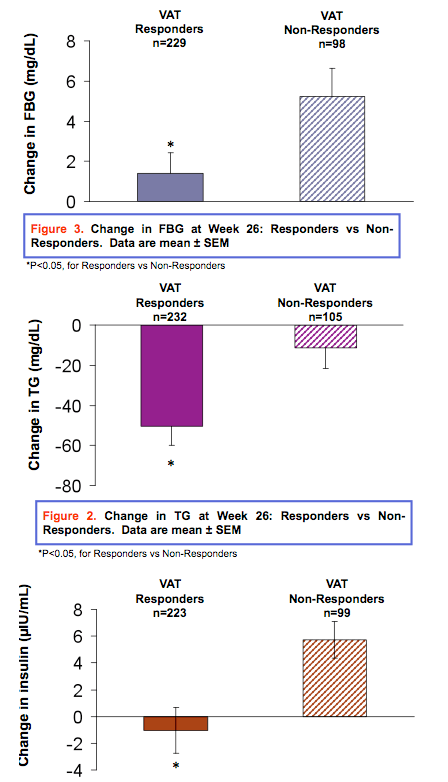
Overall, results from these analyses reveal a significantly greater responder rate in HIV-positive tesamorelin-treated patients compared to placebo (among responders there was a reduction in VAT of 27.4%). FBG and insulin were unchanged in the responders to tesamorelin, but deteriorated in non-responders. In fact, no clinically significant changes in glucose parameters occurred in the vast majority of VAT responders. Moreover, significant benefits in metabolic risk factors accrued. These data provide important new evidence for the clinical utility and metabolic benefits of VAT reduction by augmenting the physiological secretion of GH.
Julian Falutz1, Hani Assaad2, Christian Marsolais2, Josee Morin2, Jean-Claude Mamputu2, Graziella Soulban2, and Steven Grinspoon3
1Montreal General Hospital, McGill University Health Centre, Montreal, Canada; 2Theratechnologies Inc., Montreal, Canada; 3Massachusetts General Hospital and Harvard Medical School, Boston, MA, USA.
ABSTRACT
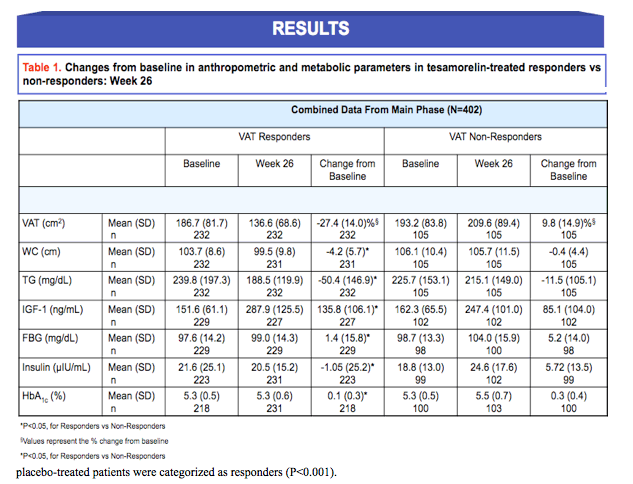
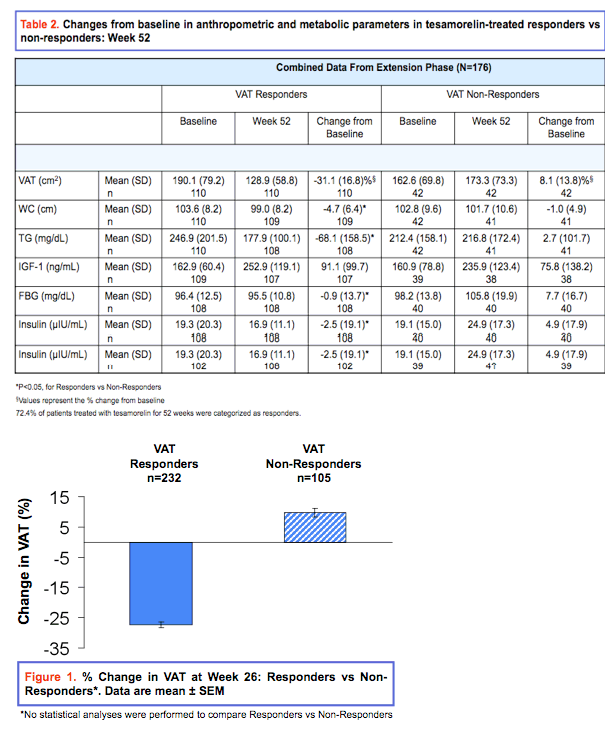
Background: Treatment with tesamorelin has been shown to significantly reduce visceral adipose tissue (VAT) in HIV patients with excess abdominal fat. Reduction in VAT may have a beneficial impact on metabolic risk factors, including glucose and triglycerides; however, the relationship between changes in these metabolic risks and the degree of VAT response to tesamorelin has not been previously investigated among this patient population. The objective of this post hoc analysis was to characterize responders in terms of change in metabolic risk factors. Responders were defined as patients with a VAT decrease from baseline ≥ 8% at Week 26 or 52 per protocol definition.
Method: Two Phase III multicenter, randomized, double-blind, trials, consisting of 26-week, placebo-controlled primary intervention phase followed by 26-week extension phase were conducted. Data from the two studies were pooled. In the per-protocol population, 402 HIV patients with excess abdominal fat were treated with tesamorelin for the main phase (26 weeks) and 176 patients for both phases (52 weeks). Statistical analyses were performed on combined results from the 2 trials using ANCOVA models for efficacy and some safety parameters.
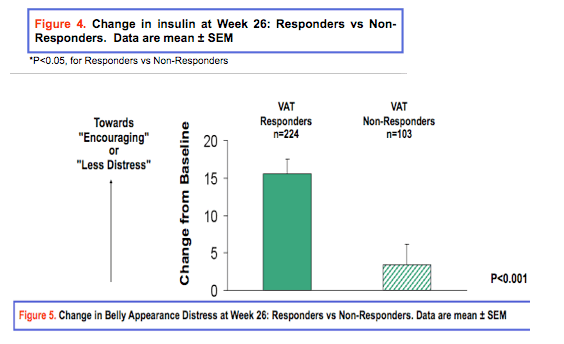
Results: Dropout rates were similar in treatment and placebo groups. At Week 26, 68.8% (n=232) of tesamorelin-treated patients vs. 32.6 % (n=56) of placebo-treated patients were categorized as responders (P<0.001). 72.4% of patients treated with tesamorelin for 52 weeks were categorized as responders. Changes from baseline [mean (standard-deviation)] in tesamorelin-treated responders versus non-responders are shown in Tables 1 and 2, for the main and the extension phases. Distress due to belly image as assessed by patients was also improved in VAT responders at Week 26 (score change: 15.51 vs. 3.40 (P<0.001)). FBG and Insulin were unchanged in responders and deteriorated in non-responders.
Conclusion: This analysis revealed a significantly greater responder rate in tesamorelin-treated patients compared to placebo. FBG and insulin were unchanged in the responders but deteriorated in non-responders. These data show that no clinically significant changes in glucose parameters occurred in the large majority of HIV patients with excess abdominal fat responding to tesamorelin, with significant reductions in VAT. Moreover, significant benefits in metabolic risk factors accrued. These data provide important new evidence for the clinical utility and metabolic benefits of strategies to reduce VAT by augmenting the biological secretion of GH.
Introduction
Abdominal obesity, particularly due to visceral adipose tissue (VAT), is strongly associated with an adverse metabolic risk profile (1,2). As such, reductions in VAT may favorably impact such risk factors. Tesamorelin, a stabilized analogue of growth hormone-releasing factor (GRF)/growth hormone-releasing hormone (GHRH), has been shown to reduce VAT in HIV-infected patients with excess abdominal fat (3). However the degree of the VAT response following treatment with tesamorelin and associated changes in metabolic risk, had hitherto not been investigated. In TheratechnologiesŐ pivotal trials with tesamorelin, responders were defined as those patients with a ≥ 8% decrease in VAT from baseline. Results from the analyses performed herein characterize responders in terms of changes in metabolic risk factors following 26 and 52-week treatments with tesamorelin.
Patients & Methods
STUDY DESIGN OF PIVOTAL TRIALS:
HIV-positive patients with excess abdominal fat in the context of antiretroviral therapy, were randomized to receive daily subcutaneous injections with either tesamorelin or placebo for 26 weeks. Patients originally on tesamorelin were re-randomized to receive either placebo or tesamorelin, while patients originally on placebo were switched to tesamorelin, all for an additional 26 weeks.
OBJECTIVE OF THIS ANALYSIS:
The objective of the current analysis was to characterize responders in terms of changes in metabolic risk factors.
STATISTICAL ANALYSES: Based on the combined results from the 2 trials, statistical analyses were performed on the change from baseline using ANCOVA models for efficacy and safety parameters.
At Week 26, 68.8% (n=232) of tesamorelin-treated patients vs. 32.6 % (n=56) of placebo-treated patients were categorized as responders (P<0.001).
REFERENCES
1. Despres JP. Health consequences of visceral obesity. Ann Med. 2001; 33:534-541.
2. Despres JP. and Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006; 444(7121):881-887.
3. Falutz J., et al. Effects of tesamorelin (TH9507), a growth hormone-releasing factor analog, in human immunodeficiency virus-infected patients with excess abdominal fat: a pooled analysis of two multicenter, double-blind placebo-controlled phase 3 trials with safety extension data. J Clin Endocrinol Metab. 2010;95(9):4291-304.
|
| |
|
 |
 |
|
|