 |
 |
 |
| |
Early HAART in TB-Treated Patients Recommended in 2 Studies at CROI for Patients with <50 CD4s
|
| |
| |
Reported by Jules Levin
CROI 2011 Boston Feb 27-March 2
In his NATAP CROI Report Eric Daar summarizes his observations:
CROI: CROI 2011 Report Selected Topics - Eric S. Daar, M.D. Chief, Division of HIV Medicine Harbor-UCLA Medical Center Professor of Medicine - (03/8/11)
"These three randomized, controlled trials consistently show that HIV-infected patients who start TB treatment with <50 CD4 cells/uL should initiate ART during the first weeks. In contrast, for those with >50 CD4 cells/uL, it is less clear that a clinical benefit is associated with starting ART within the first 2 versus 8-12 weeks of TB treatment."
ARV therapy in patients with TB
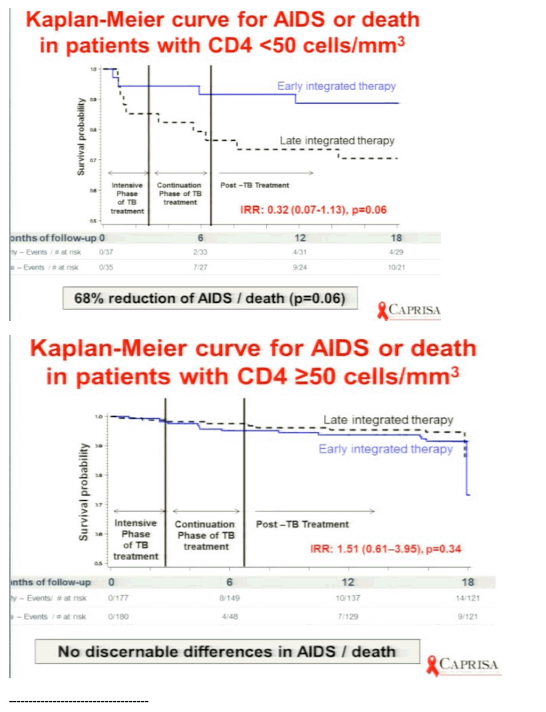
A recent study showed the benefit of earlier initiation of ARV therapy in those diagnosed with opportunistic infections, in particular pneumocystis pneumonia (24). More recently related studies have illustrated similar benefits of early ARV therapy in those with tuberculosis. The CAMELIA study enrolled patients with <200 CD4 cells/uL to start ARVs within 2 weeks or 8 weeks of initiating TB treatment. This study population had a median CD4 cell count of 25 cells/uL and showed that there was a significantly greater risk of death amongst those who waited until 8 weeks to start ARVs (25). A second study called SaPiT enrolled patients with TB and <500 CD4 cells/uL, that had a median CD4 cell count of approximately 150 cells/uL. One of the three arms was prematurely discontinued because of higher risk of progression to AIDS or death in those who delayed starting ARVs until they completed their course of TB treatment (26). The other two study groups either started ARVs within approximately 4 weeks or 8-12 weeks after initiating TB treatment. For this comparison they found that overall there was no significant difference for the primary endpoint; however, in a prespecified analysis, patients with <50 CD4 cells/uL who started ART earlier had a 68% reduced risk of progression to new AIDS defining illness or death (p=0.06) (27). In contrast, no difference was seen between study groups for those with >50 CD4 cells/uL (p=0.34). The risk of IRIS was significantly higher for patients starting ART earlier, but this was not associated with increased mortality.
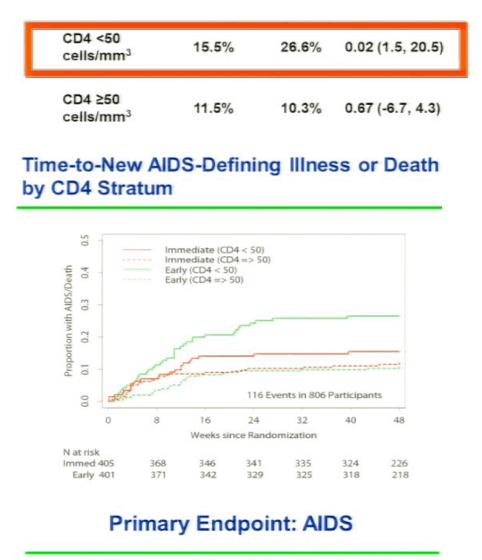
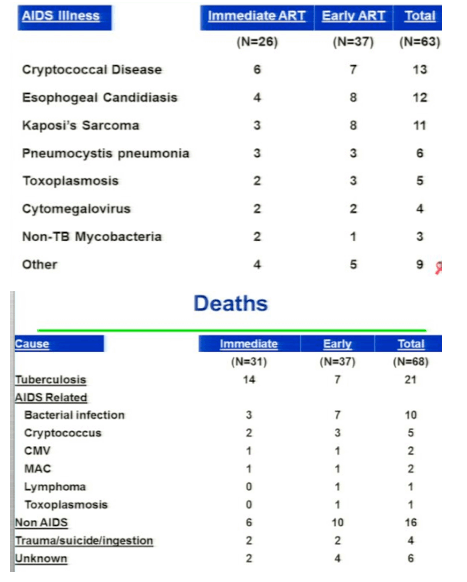
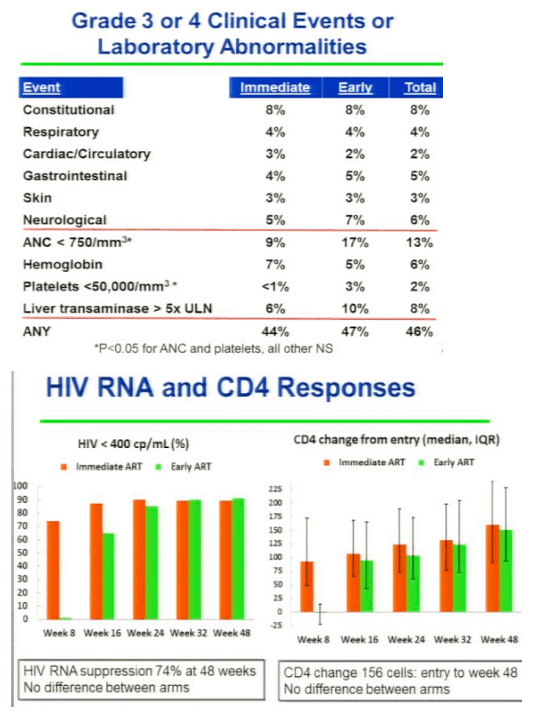
The STRIDE study (ACTG 5221) randomized 806 patients starting TB treatment who had <250 CD4 cells/uL to either initiate ARVs within 2 weeks or 8-12 weeks of TB therapy. The primary endpoint was week-48 all-cause mortality or new AIDS illnesses. The study population had a median CD4 count of 77 cells/uL and had no difference in primary endpoint at week 48 (p=0.45). In a prespecified analysis of patients with <50 CD4 cells/uL there was again a significantly greater risk of progression to AIDS or death amongst those who delayed initiation of ART until 8-12 weeks after starting TB treatment (p=0.02). Of note, there was no difference between study groups in secondary endpoints of CD4 cell increase, virologic suppression, and grade 3 or 4 adverse events. In contrast, there was an increased risk of IRIS in patients who started earlier ARV therapy. These three randomized, controlled trials consistently show that HIV-infected patients who start TB treatment with <50 CD4 cells/uL should initiate ART during the first weeks. In contrast, for those with >50 CD4 cells/uL, it is less clear that a clinical benefit is associated with starting ART within the first 2 versus 8-12 weeks of TB treatment.
-----------------------------
There were 2 studies presented at CROI back to back in the oral session:
"TB and Opportunistic Diseases"
The 1st presented was by Diane Havlir and the 2nd by Salim Abdool Karim with both finding better outcomes (AIDS/death) when starting ART early ve late in TB-treated patients, 2 studies conducted in different patient populations.
Link to video:
http://app2.capitalreach.com/esp1204/servlet/tc?c=10164&cn=retro&s=20445&&dp=player.jsp&e=13710&mediaType=podiumVideo
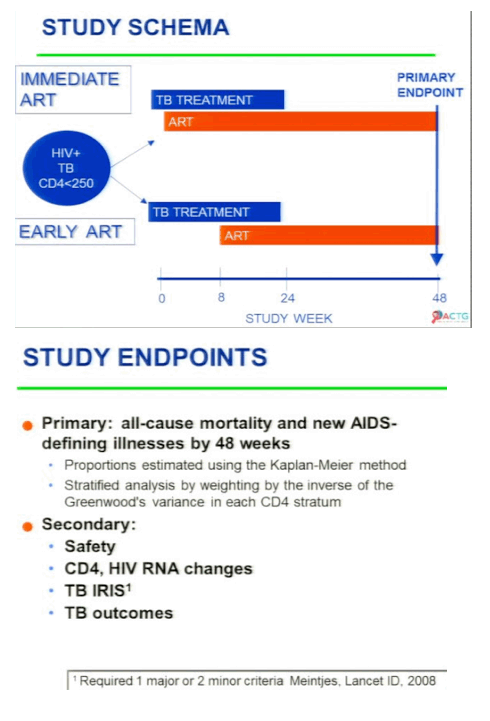
International Randomized Trial of Immediate vs Early ART in HIV+ Patients Treated for TB: ACTG 5221 STRIDE Study presented by Diane Havlir:
'Overall immediate ART did not reduce AIDS-defining ilness & death compared to early ART (median 10 days[2 wks] vs 70 days [8-12 wks]), HOWEVER for persons with CD4 <50 immediate ART reduced mortality/AIDS. Grade 3 or 4 toxicities did not differ between arms. No differences in HIV RNA suppression rates (74%) or CD4 rise between arms. TB IRIS was higher in immediate vs early arms'. "Havlir concludes: both immediate & erly ART strategies are safe & do not jeopardize CD4 or viral load suppression rates. In patients with CD4 <50, ART should be started within 2 weeks - delays increase AIDS/death. TB IRIS is more common in those receiving immediate ART, but does not increase mortality. Implementation of these findings should be a high priority in HIV & TB programs and will require coordination with hospital & outpatient programs."
Initiation of ART in patients currently treated for pulmonary TB reduces mortality. Optimal timing of ART initiation for TB/HIV co-infected patients of varying levels of immune compromise has not been established. AIDS Clinical Trials Group (ACTG) 5221 was an open-label, randomized, strategy study comparing immediate ART (2 weeks after TB treatment start) to early ART (8 to 12 weeks) among HIV+ subjects with CD4+ cells <250/mm3 treated for confirmed or suspected TB. Randomization was stratified by CD4 (<50 vs ≥50). The primary hypothesis was that immediate ART would reduce AIDS and death compared to early ART by 48 weeks. Subjects received standard rifampicin-containing TB trearment, trimethoprim/sulfamethoxazole (TMP/SMX) prophylaxis, and tenofovir/emtricitabine (TDF/FTC) + efavirenz (EFV) ART. Event proportions at 48 weeks were estimated by the Kaplan-Meier method (LFU = censor); stratified tests used Greenwood's variance. Wilcoxon and Fisher's exact tests were used as appropriate. Of 806 individuals, enrolled from 26 sites on 4 continents, 405 received immediate ART and 401 early ART. Median baseline CD4 count was 77 cells/mm3 (IQR 36 to 145) and HIV RNA of 5.43 log10 copies/mL (IQR 5.00 to 5.79). 97% of patients took EFV/TDF/FTC for HAART. The median time to starting HAART was 10 days in immediate arm and 70 days in the early arm. RESULTS: The proportion with AIDS or death in the immediate arm was 12.9% and 16.1% in the early arm and the difference was not statistically different (p=0.45), but when they did the prespecified CD4 analysis stratified by CD4 cell count for patients with <50 CD4s at baseline there was a statistically significant difference with 15.5% in the immediate arm with AIDS or death vs 26.6% in the early arm (p=0.02) experiencing AIDS or death. For patients with 50 or more CD4s there was no difference with 11.5% in the immedite arm vs 103% in the early arm experiencing AIDS or death. Overall, grade 3 or 4 toxicities did not differ between arms (p = 0.29). Viral suppression (<400 copies/mL) at 48 weeks was 74% and did not differ between arms; and 60% in each arm achieved CD4 cell rise ≥100 cells/mm3 at 48 weeks. TB immune reconstitution inflammatory syndrome (IRIS) was significantly higher with immediate ART than early ART (43 [11%] vs 19 [5%], p = 0.009); but no deaths were attributed to TB IRIS
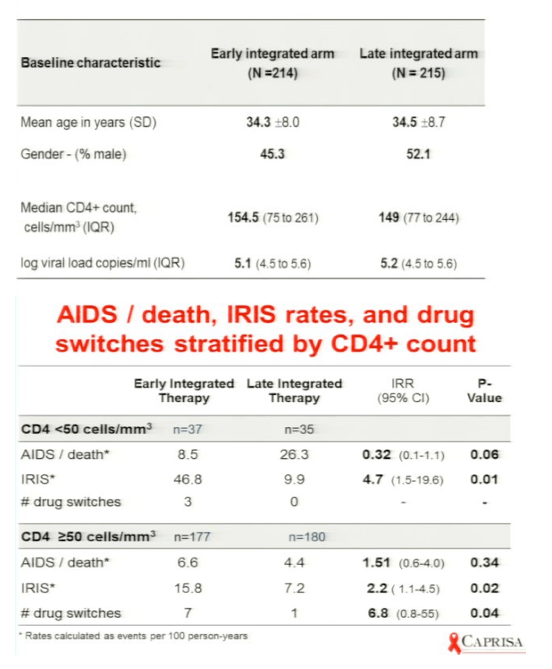
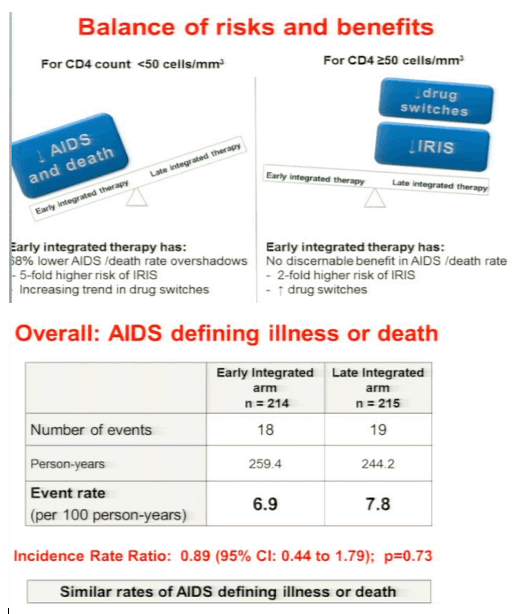
Karim concluded: in the early arm the median time for starting HAART was 8 days, he strongly recommends starting HAART early in <50 CD4s. findings support integration of TB & HIV treatment. RECOMMEND: patients with CD4 <50, early ART initiation assoon as possible after TB treatment initiation. Patients with CD4 50 or more ART initiation can be deferred to the start of the continuation phase of TB treatment. Decision on early or late initiation: use clinical judgment of capacity to manage IRIS & toxicities. In patients with baseline CD4+ counts <50 cells/mm3 (n = 72), the incidence rates of AIDS or death were 8.5 in the early therapy group compared to 26.3/100 person-years in the late therapy group (IRR 0.32; 95%CI 0.07 to 1.13; p = 0.06). Immune reconstitution inflammatory syndrome (IRIS) incidence rates in these patients were 46.8 in the early compared to 9.9 in the late therapy group (IRR, 4.71, p = 0.01). Three of the patients with CD4+ counts <50 cells/mm3, all in the early therapy group, required ART drug switches due to adverse events. In patients with baseline CD4+ counts ≥50 cells/mm3 (n = 357), the incidence rates of AIDS or death were 6.6 and 4.4/100 person-years in the early and late therapy groups, respectively (IRR, 1.51, p = 0.34). In these patients, the early therapy group had a higher IRIS incidence rate (15.8 vs 7.2/100 person-years, IRR 2.18, p = 0.02).The early therapy group also had more antiretroviral drug switches due to adverse events; 7 in the early therapy group compared to 1 in the late therapy group (p = 0.04). Conclusions: In patients with pulmonary TB/HIV co-infection with CD4+ counts <50 cells/mm3, early ART initiation within 4 weeks of TB treatment initiation was associated with better AIDS-free survival, albeit with increased risk of IRIS. However, in patients with CD4 ≥50 cells/mm3, delaying initiation of ART to the first 4 weeks of continuation phase of TB reduced the risk of IRIS and drug switches without compromising AIDS-free survival.
-------------------------------------
Paper # 38
International Randomized Trial of Immediate vs Early ART in HIV+ Patients Treated for TB: ACTG 5221 STRIDE Study
Diane Havlir*1, P Ive2, M Kendall3, A Luetkemeyer1, S Swindells4, J Kumwenda5, S Qasba6, E Hogg7, J Anderson3, I Sanne2, and A5521 Team
1Univ of California, San Francisco, US; 2Univ of the Witwatersrand, Johannesburg, South Africa; 3Harvard Sch of Publ Hlth, Boston, MA, US; 4Univ of Nebraska, Omaha, US; 5Johns Hopkins Univ Res Project, Blantyre, Malawi; 6NIH, Bethesda, MD, US; and 7Social & Sci Systems, Inc, Silver Spring, MD, US
Background: Initiation of ART in patients currently treated for pulmonary TB reduces mortality. Optimal timing of ART initiation for TB/HIV co-infected patients of varying levels of immune compromise has not been established.
Methods: AIDS Clinical Trials Group (ACTG) 5221 was an open-label, randomized, strategy study comparing immediate ART (2 weeks after TB treatment start) to early ART (8 to 12 weeks) among HIV+ subjects with CD4+ cells <250/mm3 treated for confirmed or suspected TB. Randomization was stratified by CD4 (<50 vs ≥50). The primary hypothesis was that immediate ART would reduce AIDS and death compared to early ART by 48 weeks. Subjects received standard rifampicin-containing TB trearment, trimethoprim/sulfamethoxazole (TMP/SMX) prophylaxis, and tenofovir/emtricitabine (TDF/FTC) + efavirenz (EFV) ART. Event proportions at 48 weeks were estimated by the Kaplan-Meier method (LFU = censor); stratified tests used Greenwood's variance. Wilcoxon and Fisher's exact tests were used as appropriate.
Results: Of 806 individuals, enrolled from 26 sites on 4 continents, 405 received immediate ART and 401 early ART. Median baseline CD4 count was 77 cells/mm3 (IQR 30 to 137) and HIV RNA of 5.43 log10 copies/mL (IQR 5.00 to 5.79). TB diagnosis was confirmed or suspected in 46% and 54% of cases. Median ART start times were 10 days (IQR 7 to 12) and 10 weeks (9.4 to 10.7) from start of TB treatment for the immediate ART and the early ART arms, respectively; 13.0% in the immediate ART arm and 16.1% in the early ART arm experienced AIDS or death at 48 weeks (p = 0.45, difference CI -1.8 to 8.1). In the low CD4 stratum (≤50, n = 285), 15.5% on immediate ART and 26.6% on early ART experienced AIDS or death (p = 0.02, difference CI 1.5 to 20.5) in pre-specified subset analyses. In the high CD4 stratum (n = 521), 11.5% on immediate ART and 10.3% on early ART experienced events (p = 0.67, difference CI -6.7 to 4.3). TB immune reconstitution inflammatory syndrome (IRIS) was significantly higher with immediate ART than early ART (43 [11%] vs 19 [5%], p = 0.009); but no deaths were attributed to TB IRIS. Overall, grade 3 or 4 toxicities did not differ between arms (p = 0.29). Viral suppression (<400 copies/mL) at 48 weeks was 74% and did not differ between arms; and 60% in each arm achieved CD4 cell rise ≥100 cells/mm3 at 48 weeks.
Conclusions: Overall, immediate ART did not reduce AIDS and death compared to early ART. For persons with CD4 ≤50 cells/mm3 immediate ART resulted in lower rates of AIDS and death compared to early ART.
---------------------------------------
Paper # 39LB
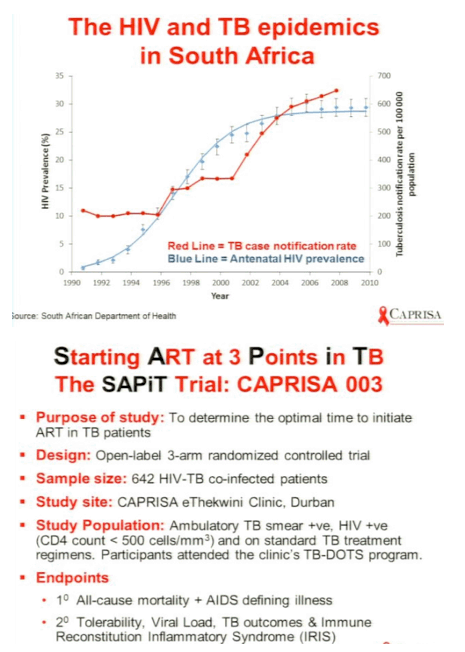
Optimal Timing of ART during TB Therapy: Findings of the SAPiT Trial
Salim Abdool Karim*1,2,3, K Naidoo1, N Padayatchi1, A Grobler1, C Baxter1, S Gengiah1, W El-Sadr4, G Friedland5, and Q Abdool Karim1,3
1Ctr for the AIDS Prgm of Res in South Africa, Durban; 2Univ of KwaZulu-Natal, Durban, South Africa; 3Columbia Univ, Mailman Sch of Publ Hlth, New York, NY, US; 4Intl Ctr for AIDS Care and Treatment Prgms, Columbia Univ, Mailman Sch of Publ Hlth, New York, NY, US; and 5Yale Univ, New Haven, CT, US
Background: Initiation of ART during TB treatment reduced mortality by 55% compared to ART initiation upon TB treatment completion. However, optimal time to initiate ART during TB treatment remains unclear.
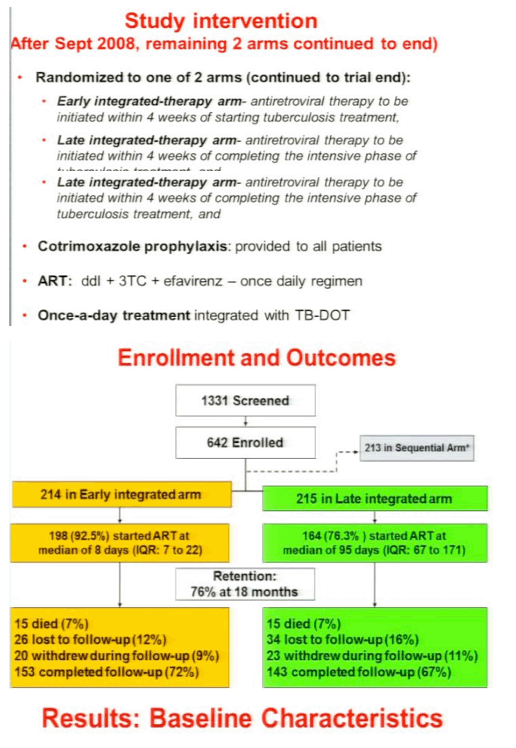
Methods: We compared outcomes of early therapy (ART initiated within 4 weeks of TB treatment initiation, n = 214) and late therapy (ART initiated within the first 4 weeks of the continuation phase of TB treatment, n = 215) in an open-label randomized controlled trial in South Africa in sputum acid-fast bacilli smear-positive patients (n = 642) with HIV and CD4+ counts <500 cells/mm3.
Results: Median CD4+ count and viral load at baseline was 150 cells/mm3 and 161,000 copies/mL, and was similar in both groups. Overall, the incidence rate of AIDS or death was 6.9 (18 of 259.4) and 7.8 (19 of 244.2) per 100 person-years in the early and late therapy groups, respectively (incidence rate ratio = 0.89, 95%CI 0.44 to 1.79; p = 0.73). In patients with baseline CD4+ counts <50 cells/mm3 (n = 72), the incidence rates of AIDS or death were 8.5 in the early therapy group compared to 26.3/100 person-years in the late therapy group (IRR 0.32; 95%CI 0.07 to 1.13; p = 0.06). Immune reconstitution inflammatory syndrome (IRIS) incidence rates in these patients were 46.8 in the early compared to 9.9 in the late therapy group (IRR, 4.71, p = 0.01). Three of the patients with CD4+ counts <50 cells/mm3, all in the early therapy group, required ART drug switches due to adverse events. In patients with baseline CD4+ counts ≥50 cells/mm3 (n = 357), the incidence rates of AIDS or death were 6.6 and 4.4/100 person-years in the early and late therapy groups, respectively (IRR, 1.51, p = 0.34). In these patients, the early therapy group had a higher IRIS incidence rate (15.8 vs 7.2/100 person-years, IRR 2.18, p = 0.02).The early therapy group also had more antiretroviral drug switches due to adverse events; 7 in the early therapy group compared to 1 in the late therapy group (p = 0.04).
Conclusions: In patients with pulmonary TB/HIV co-infection with CD4+ counts <50 cells/mm3, early ART initiation within 4 weeks of TB treatment initiation was associated with better AIDS-free survival, albeit with increased risk of IRIS. However, in patients with CD4 ≥50 cells/mm3, delaying initiation of ART to the first 4 weeks of continuation phase of TB reduced the risk of IRIS and drug switches without compromising AIDS-free survival.





|
| |
|
 |
 |
|
|