 |
 |
 |
| |
Renal Impairment in Patients Receiving a TDF-based cART Regimen: Impact of TDF Concentration?
|
| |
| |
Isabelle Poizot-Martin*1, C Solas2, J Allemand1, V Obry-Roguet1, S Ronot-Bregigeon1, A Menard1, O Faucher1, and B Lacarelle2
1Hosp Ste Marguerite, Marseille, France and 2Hosp de La Timone, Marseille, France
Reported by Jules Levin
CROI 2011 March 2 Boston
"Our results show a high prevalence of patients with a Ctrough -TDF > 90 ng/mL and a correlation between a high Ctrough-TDF and the decrease of ClCr."
In patients with a Ctrough-TDF > 90 ng/ml,we observed:
- a lower ClCr 12 months later (p<0.001)
- a higher decrease in the ClCr (p=0.002)
- a higher proportion of patients with a decrease of ClCr > -10ml/m(p=0.017)
AUTHOR CONCLUSIONS
1. Our results show a significant correlation between the Ctrough-TDF and the decrease of the ClCr and highlight a toxic concentration- dependent effect of TDF on glomerular
filtration as proposed by Rodriguez-Novoa et al for tubular nephropathy observed in patients treated with TDF (6).
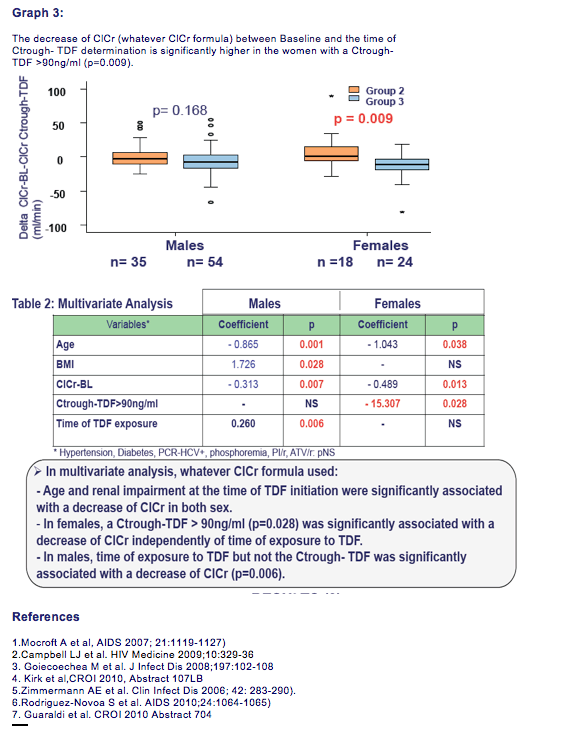
2. Age and an alteration of the glomerular filtration at the time of TDF initiation were associated with a decrease of ClCr (2) but we did not found such association with hypertension,
diabetes and HCV co- infection (1).
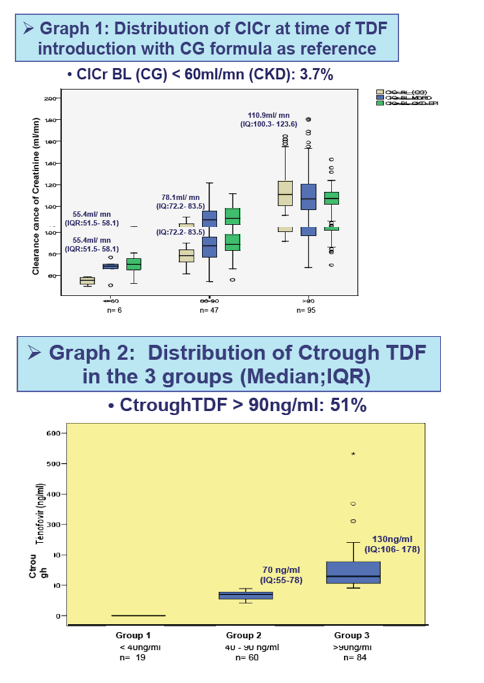
3. The high prevalence of patients with a Ctrough-TDF > 90ng/ml (51%) (graph 2) emphasizes the interest of therapeutic drug monitoring for TDF especially in women and older patients.
The impact of a dose reduction of TDF according to the Ctrough has to be evaluated. Such dose reduction is up to now recommanded in case of ClCr <50ml/mn.
4. An important variability in the ClCr estimation (graph 1), was observed according to the formula used, as reported Guaraldi et al(7). Studies to validate the most accurate estimating
equation to be used in HIV- infected patients for ClCr calculation have to be urgently performed
PATIENTS
Overall 168 patients treated by a TDF-based regimen were identified, among which 5 patients were excluded because of chronic renal failure or co-administration of nephrotoxic drugs at the time of the analysis. Data from 163 patients were included in the analysis.
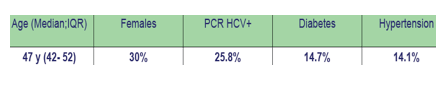
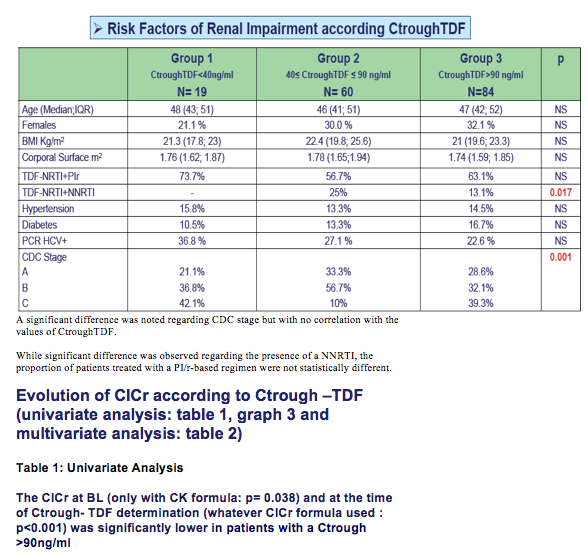
The median time of HIV follow-up was 17.2years (IQR:11.3; 21.1). The median duration of cART exposure was 8.7years (IQR:4.3;11.9) and 12.9 % of patients were CDC stage C.
INTRODUCTION
Prevalence of chronic kidney disease (CKD) in HIV infected patients is around 4 to 17% according to litterature (1). CKD has been reported in assocation with older age, female gender, hepatitis B and C infections, diabetes, hypertension and Art exposure(1,2).
Tenofovir (TDF) undergoes renal clearance by a combination of glomerular filtration and active tubular secretion. Use of TDF in patients with renal impairment, diabetes, or with boosted protease inhibitor (PI/r) increase the risk of a significant decrease of glomerular filtration (10ml/mn) calculated with the Cockroft and Gault formula (3, 4).
TDF may induce proximal renal tubular dysfunction and, in some cases a Fanconi syndrome (5). A relationship between TDF plasma concentration (Ctrough-TDF) and impairment in kidney tubular function has been recently reported(6).
Objectives
To evaluate the distribution of the Ctrough-TDF in HIV-infected patients exposed to a TDF-based cART regimen
To compare the evolution of the creatinine. clearance (ClCr) according to the Ctrough- TDF
To determine independent variables predicting TDF-induced renal dysfunction
Background: A relationship between tenofovir (TDF) plasma concentration (Ctrough-TDF) and impairment in kidney tubular function has been recently reported. To evaluate the distribution of the Ctrough-TDF in HIV-infected patients and compare the evolution of clearance of creatinine (ClCr) according to the Ctrough-TDF.
Methods: Patients taking TDF-based combination ART (cART) regimen with at least 1 determination of Ctrough-TDF were selected retrospectively from our computerized database. Determination of Ctrough-TDF levels were measured at least between 17 and 24 hours after the last TDF dose using an HPLC method (threshold values, 40 to 90 ng/mL). Explanatory variables were considered at the initiation of TDF. The renal filtration rate was assessed by measurement of ClCr using the Cockcroft-Gault formula at the time of TDF initiation (baseline ClCr), at the time of Ctrough-TDF determination (T1 to ClCr) and 12 months later (T2 to ClCr) for patients still under the same cART.
Results: Data were analyzed from 163 patients: men, 70%; median age, 47 years (42 to 52); hepatitis C virus (HCV) co-infected (PCR+), 25.8%; diabetes, 14.7%; high blood pressure, 14.1%; median delay since HIV diagnosis of 17.1 years (11.3 to 21.1).
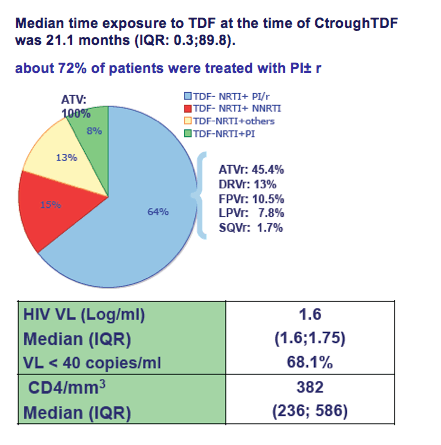
Median CD4 cell count was 382/mm3 (236 to 586), median viral load was 1.6 log/mL and 64% were treated with a PI regimen.
The Ctrough-TDF was <40 ng/mL in 19 patients (gpe1), between 40 and 90 ng/mL in 60 patients (gpe2), and >90 ng/mL in 84 patients (gpe3) with a similar median duration of TDF exposure at the time of Ctrough to TDF: 26.1 (3.8 to 51.1), 19.7 (10.8 to 50.4), and 21.9 (5.1 to 46.5) months.
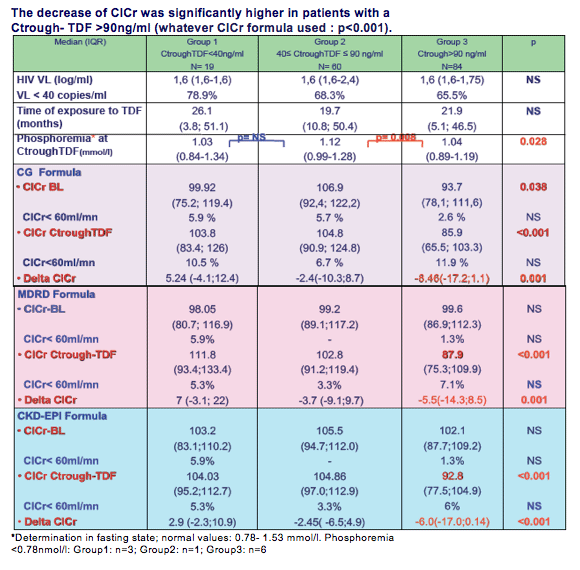
Median baseline ClCr (mL/mn) was, respectively, 99.92 (75.2 to 119.4), 106.9 (92.4 to 122.2), and 93.7 (78.1 to 111.6). Median T1 to ClCr (mL/mn) was similar for gpe1 (103.8; 83.4 to 126) and gpe2 (104.8; 90.9 to 124.8), but significantly lower in gpe3 (85.9; 65.5 to 103.3) (p <0.001).
After 12 months, a significant decrease in T2 to ClCr (mL/mn) was still observed in gpe3 (85.3; 69.6 to 99.9) (p <0.001). Median Delta (baseline to T2) to ClCr (mL/mn) was, respectively, -1.7(-8.6 to 10.95), -1.28 (-8.59 to 11.90) and -8.27 (-19.4 to -1.26) (p = 0.003).
In multivariate analysis, age (p = 0.001), renal impairment at the time of TDF initiation (p = 0.007), and a Ctrough -TDF >90 ng/mL (p = 0.028) were significantly associated with a decrease of the ClCr independently of duration of TDF exposure and body mass index.
Conclusions: Our results show a high prevalence of patients with a Ctrough -TDF > 90 ng/mL and a correlation between a high Ctrough-TDF and the decrease of ClCr.
METHODS
Cross- sectionnal and retrospective study performed in an out-door HIV clinical unit where about 1000 patients (40%HCV coinfected) are followed. Data of HIV-infected patients receiving TDF-containing cART regimen between december 2006 and January 2010 were retrospectively collected from an electronic medical
record database. Only patients with an available measurement of TDF Ctrough at least 17 hours after drug intake were included in the analysis. Patients taking drugs expected to induce nephrotoxicity were excluded.
Ctrough-TDF was quantified by HPLC. Expected interval for TDF-Ctrough was 40- 90 ng/ml. ClCr was calculated with the Cockroft and Gaul (CG), MDRD and CKD-EPI formula.
Renal failure was defined as ClCr< 60ml/mn.
Renal filtration rate was assessed at the time of TDF initiation (ClCr-BL), at the time of Ctrough TDF determination (ClCr-Ctrough-TDF) and 12 months later (ClCr- M12) for patients still under the same cART.
Patients were stratified according to Ctrough-TDF: <40ng/ml (group1), 40 -90 ng/ml (group 2) >90ng/ml (group3). Other variables included in the analysis were age, gender, weight, body mass index (BMI),corporal surface, comorbidities (hypertension, diabetes, hepatitis C), CDC stage, plasma HIV viral load
(HIV VL) and CD4 cell counts, time of exposure to TDF, phosphoremia, association of a PI/r, and association of atazanavir (ATV).
Kruskal-Wallis parametric and Mann-Whitney non-parametric tests were used for quantitative analysis. Multivariate analysis to identify the variables associated to ClCr changes were performed by using a linear regression model with SPSS 15.0 adjusted to baseline ClCr values.
RESULTS
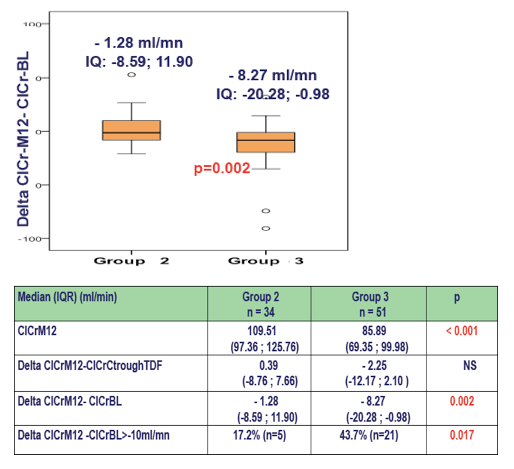
Follow up of ClCr (CG) between 10 and 14 months after Ctrough TDF measurement
Data from 98 patients still under the same cART regimen were analyzed. The median time exposure to TDF was 38.4 months (IQR:14.7; 71.1).
In patients with a Ctrough-TDF > 90 ng/ml,we observed:
- a lower ClCr 12 months later (p<0.001)
- a higher decrease in the ClCr (p=0.002)
- a higher proportion of patients with a decrease of ClCr > -10ml/m(p=0.017)
|
| |
|
 |
 |
|
|