 |
 |
 |
| |
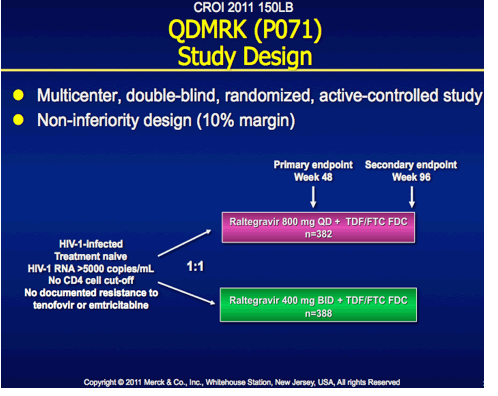
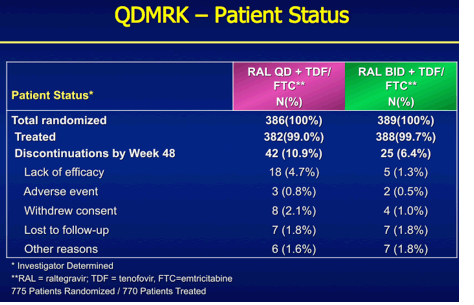
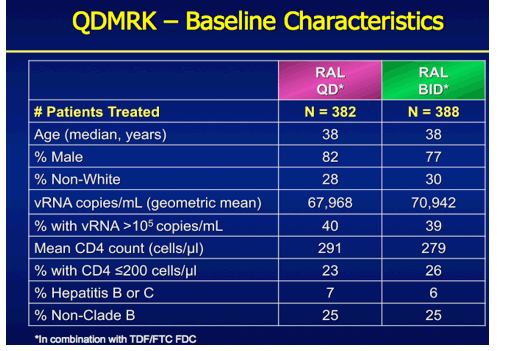
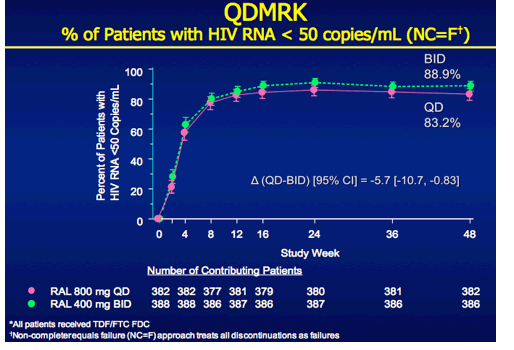
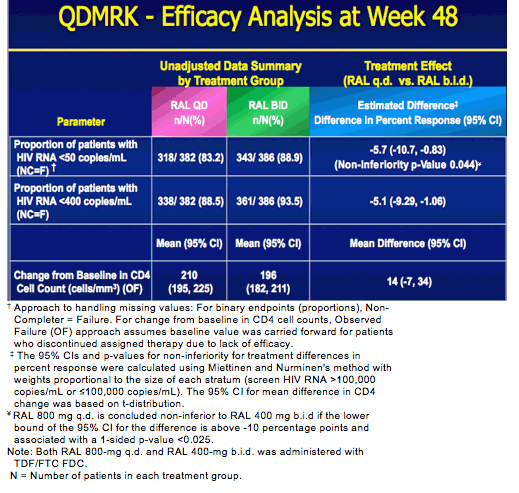
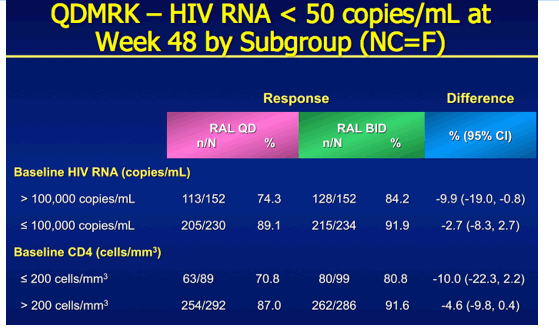
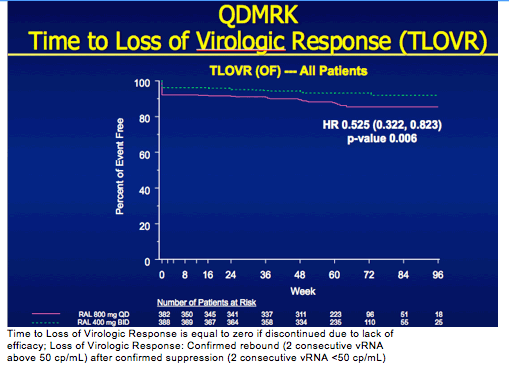
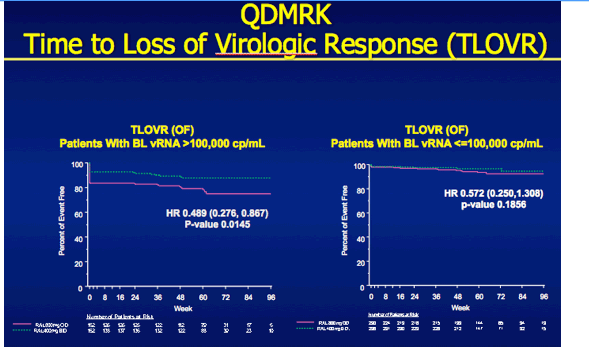
QDMRK, A Phase III Study of the Safety & Efficacy of Once Daily (QD) Versus Twice Daily (BID) Raltegravir (RAL) in Combination Therapy for Treatment-Naïve HIV-Infected Patients (Pts)
|
| |
| |
Reported by Jules Levin
J. Eron1, J. Rockstroh2, J. Reynes3, J. Andrade4, J. Madruga5, J. Zhao6, P. Sklar6, B-Y. Nguyen6 for the QDMRK Study Team
1Univ. of North Carolina, Chapel Hill, NC, USA; 2Univ. of Bonn, Bonn-Venusberg, Germany; 3Montpellier Univ. Hospital, Montpellier, France; 4Universidad de Guadalajara, Mexico; 5Centro de Referencia DST/AIDS, Sao Paulo, Brazil and 6Merck Research Laboratories, North Wales, PA, USA


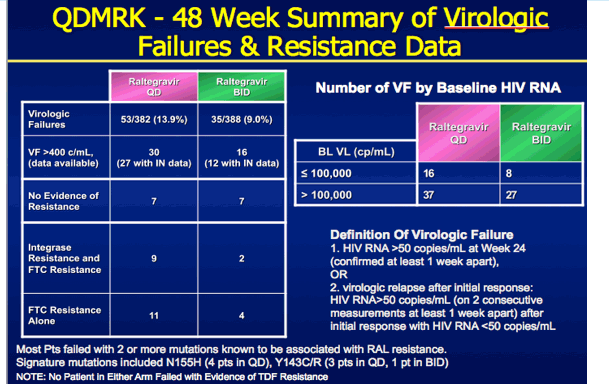
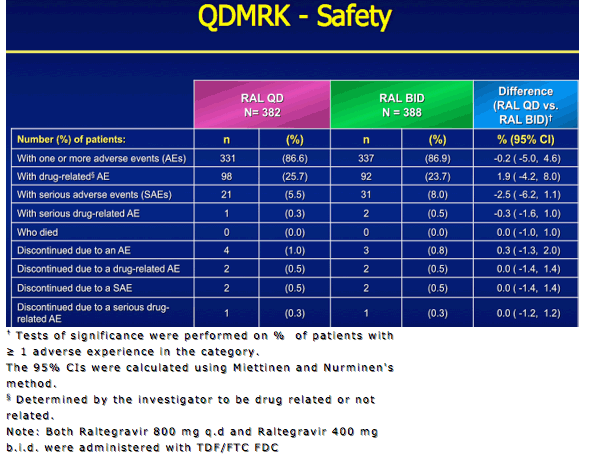
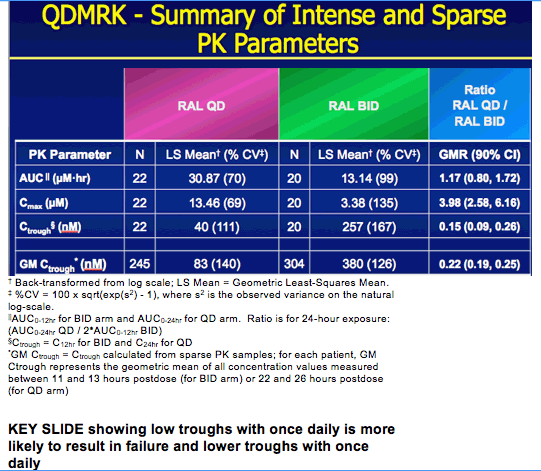
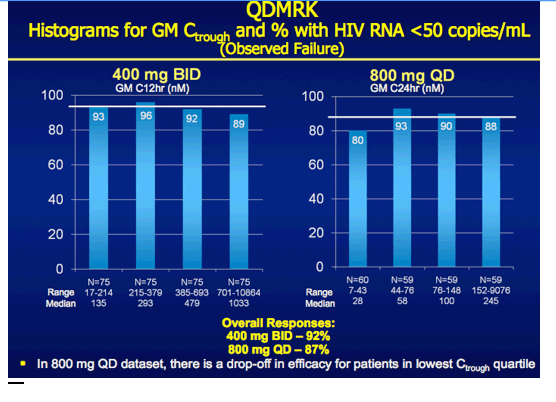
|
| |
|
 |
 |
|
|