 |
 |
 |
| |
Prompt, Significant Reduction of Activation and Proliferation Markers in CD4+ and CD8+ T Cell Subsets despite Incomplete Viral Suppression: Proof-of-Concept for AV-HALT-A New Class of Anti-HIV Therapeutics Inhibiting Both HIV Replication and Immune Activation
|
| |
| |
Reported by Jules Levin
CROI 2011 March 2 Boston
D Baev1, E Katabira2, R Maserati3, P Cahn4, D De Forni1, J Lisziewicz5, M Stevens6, and Franco Lori*1
1ViroStatics srl, Alghero, Italy; 2Makerere Univ Sch of Med, Kampala, Uganda; 3Fndn IRCCS Policlinico San Matteo, Pavia, Italy; 4Fndn Huesped, Buenos Aires, Argentina; 5Genetic Immunity Kft, Budapest, Hungary; and 6ViroStatics srl, Princeton, NJ, US

ABSTRACT
Background: A chronic increase in CD4+ and CD8+ T cell surface co-expression of the activation markers CD38 and HLA-DR is one of the major signs of HIV infection and progression to AIDS. Despite the achievement of undetectable viral loads, current HAART does not return these markers to non-HIV-infected values, suggesting a potential benefit from incorporating targeted immune-based therapies into future regimens. In a 28-day Phase II Proof-of-Concept study, these markers were used to monitor the effects of antiviral-hyperactivation limiting therapeutics ([AV-HALT] a new class of drugs developed to suppress both HIV and immune activation) administered as a non-maximally suppressive regimen.
Methods: Blood samples from a subset of 32 subjects receiving the AV-HALT VS411, a combination of 2 commercially available drugs, an antiviral (low-dose, slow-release 2',3'-dideoxyinosine) and an hyperactivation-limiting drug (low-dose hydroxycarbamide), were analyzed in a 28-day, multinational, double-blinded, Phase II Proof-of-Concept study using 10-color flow cytometry including activation and proliferation markers. Two-tailed paired t tests and non-parametric Wilcoxon tests were used for statistical analyses.
Results: The median decrease in HIV RNA was 1.5 log10 with only 2 subjects reaching < 50 copies/mL by day 28. Median CD4+ cell increase was +108 cells/mm3. The median CD38+/HLA-DR+ co-expression in CD4+ and CD8+ cells was significantly reduced between pre- and post-therapy (28 days) from 6.09% to 4.99% and from 25.87% to 16.98%, respectively (p values <0.005). The median CD4+ percentage increased from 33.92% to 38.46% (p <0.002). Correlations between expression levels of the proliferation marker Ki-67 and CD38+/HLA-DR+ co-expression in CD4+ and CD8+ subsets were found with r2 = 0.12 (p = 0.03) for CD3+CD4+ and 0.23 (p = 0.005) for CD3+CD8+ subsets for pre-treatment and r2 = 0.15 (p = 0.04) for CD3+CD4+ and 0.07 (p = 0.045) for CD3+CD8+ subsets for after-treatment values, respectively.
Conclusions: A rapid, significant decrease in CD38+/HLA-DR+ co-expression following 28 days of AV-HALT therapy in both CD4+ and CD8+ T cell subsets was achieved without completely suppressing viral replication. These results demonstrate that the immune system hyperactivation driving HIV disease progression can be specifically targeted and effectively reduced by the use of AV-HALT, suggesting a role for their use either alone or in combination with existing therapies.
Background:
A chronic increase in CD4+ and CD8+ T cell surface co-expression of the activation markers CD38 and HLA-DR is one of the major signs of HIV
infection and progression to AIDS. Despite the achievement of undetectable viral loads, current HAART does not return these markers to non-HIV-infected values, suggesting a potential benefit from incorporating targeted immune-based therapies into future regimens. In a 28-day Phase II Proof-of-Concept dose-ranging study, these markers were used to monitor the effects of an antiviral-hyperactivation limiting therapeutic* (AV-HALT) administered as a non-maximally suppressive regimen.
Methods:
Blood samples from a sub-set of 32 of the 58 total subjects receiving the AV-HALT VS411, a combination of two commercially available drugs, an antiviral (low-dose, slow-release 2',3'-dideoxyinosine) and a hyperactivation-limiting drug (low-dose hydroxycarbamide), were analyzed in a 28-day, multinational, double-blinded, Phase II Proof-of-Concept study using 10-color flow cytometry including activation and proliferation markers. Two-tailed paired t tests and non-parametric
Wilcoxon tests were used for statistical analyses.
Results:
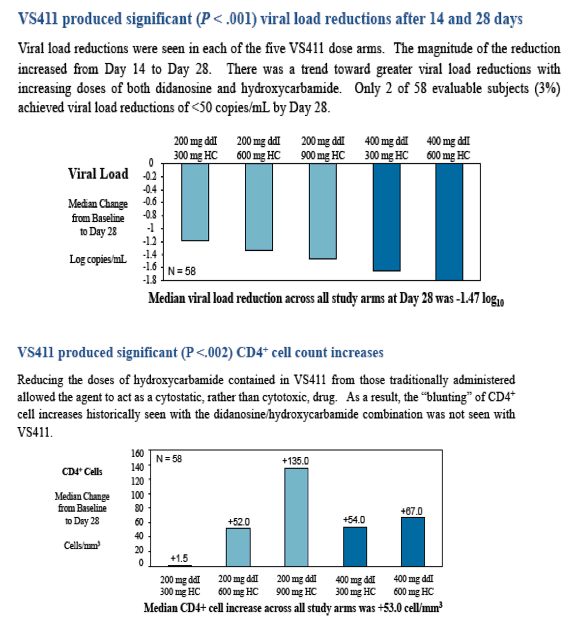
Within the 32-subject sub-study, the median decrease in HIV RNA was 1.5 log10 with only two subjects reaching < 50 copies/mL by Day 28. The median CD4+ cell increase in this group was +108 cells/mm3.
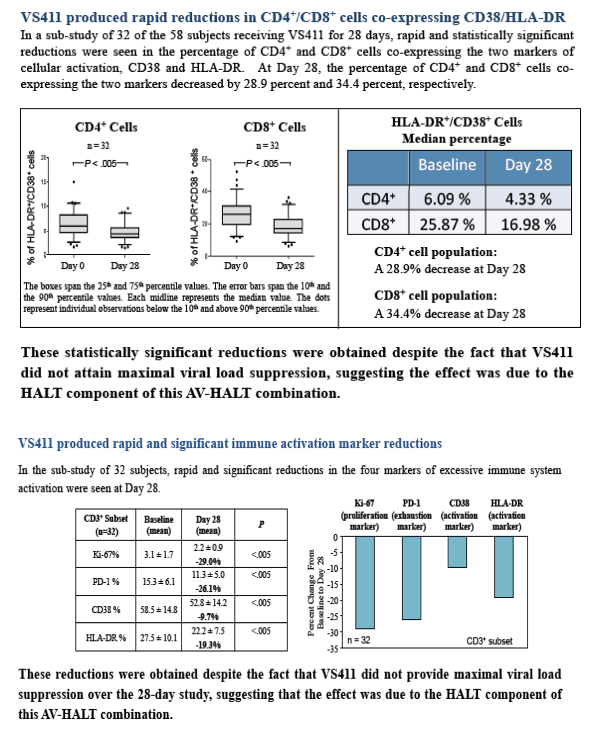
The median CD38+/HLA-DR+ co-expression in CD4+ and CD8+ cells
was significantly reduced between pre- (baseline) and post-therapy
(28 days) from 6.09% to 4.33% and from 25.87% to 16.98%,
respectively (P values <.005).
Over the 28 days of the study, the median CD4+ percentage increased from 33.92% to 38.46% ( P<.002).
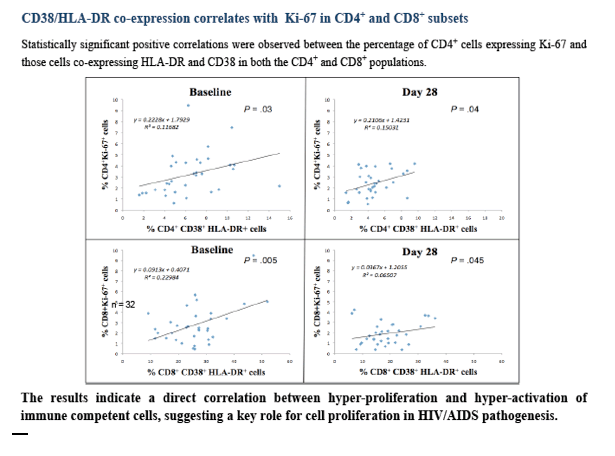
Correlations between expression levels of the proliferation marker Ki-67 and CD38+/HLA-DR+ co-expression in CD4+ and CD8+ subsets were found with r2=0.12 (P=.03) for CD3+CD4+ and 0.23 (P=.005) for CD3+CD8+ subsets for pre-treatment and r2=0.15 (P=.04) for CD3+CD4+ and 0.07 (P=.045) for CD3+CD8+ subsets for after-treatment values, respectively.
*AV-HALT: A new class of drugs developed to suppress both HIV replication and immune activation
|
| |
|
 |
 |
|
|