 |
 |
 |
| |
The Effect of Low-dose Ritonavir on the Pharmacokinetics of the Investigational HCV Protease Inhibitor Telaprevir in Healthy Volunteers
|
| |
| |
Reported by Jules Levin
CROI 2011 March 2 Boston
V Garg1, X Luo1, L McNair1*, R van Heeswijk2, and RS Kauffman1
1Vertex Pharmaceuticals Incorporated, Cambridge, MA, USA ,2Tibotec BVBA, Beerse, Belgium
INTRODUCTION
Telaprevir (TVR), an inhibitor of the hepatitis C virus (HCV) NS3/4A protease,1,2
is being developed as a treatment for hepatitis C infection in combination with
pegylated interferon and ribavirin. In vitro studies suggested that TVR is both a
substrate and an inhibitor of CYP3A. In addition, in vivo studies in rats demonstrated
an increased plasma exposure of TVR when co-administered with ritonavir (RTV).3
The objective of this study was to evaluate the effect of low-dose RTV on the
pharmacokinetics (PK) of TVR in human volunteers.
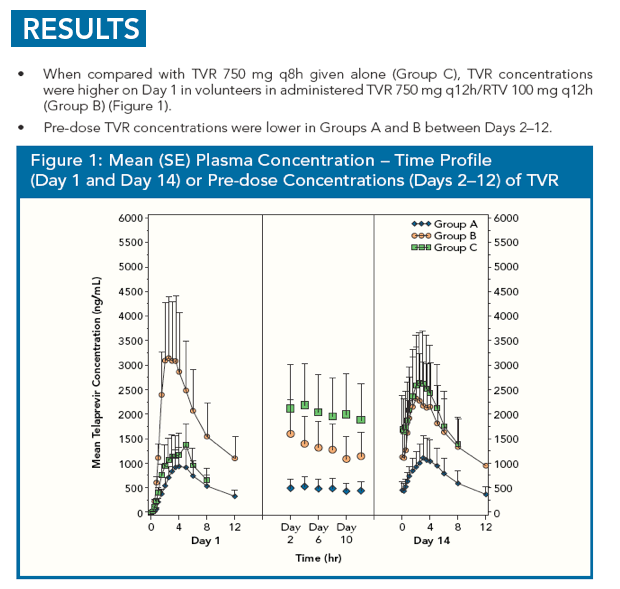
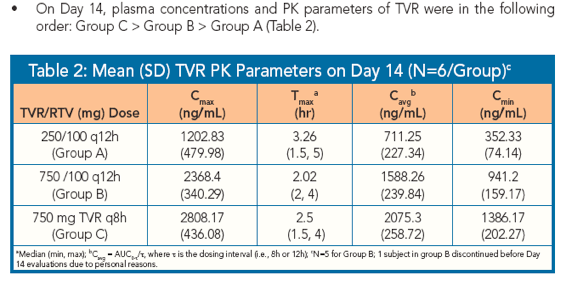
|
| |
|
 |
 |
|
|