 |
 |
 |
| |
Telaprevir Substantially Improved SVR Rates Across All IL28B Genotypes in the ADVANCE Study
|
| |
| |
Reported by Jules Levin
EASL 2011 March 31 Berlin Germany
I Jacobson et al
AUTHOR SUMMARY
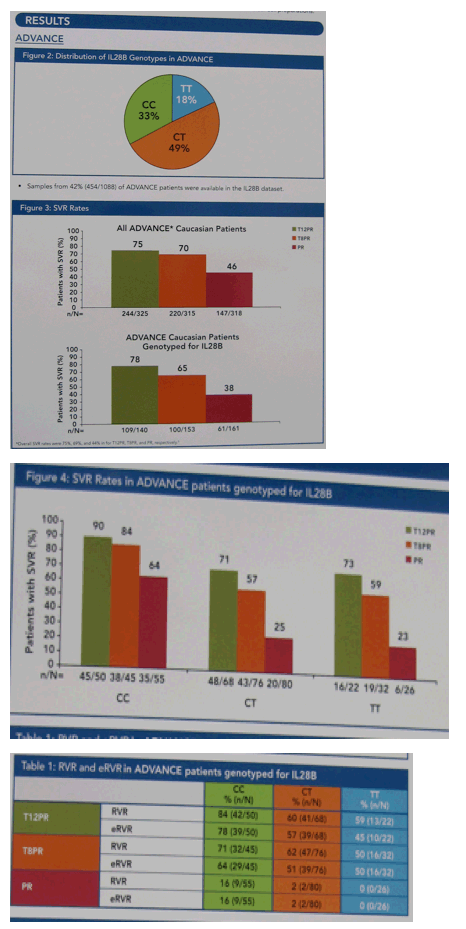
The distribution of IL28B genotypes was consistent with previously published results.
In the Phase 3 ADVANCE trial, the addition of telaprevir to peginterferon alfa-2a/RBV improved SVR across all IL28B genotypes versus peginterferon alfa-2a/RBV alone.
SVR rates were similar heterozygous (CT) and homozygous (TT) patients receiving telaprevir-based therapy.
The largest increment in SVR occurred in patients with the T allele: 71% in T12PR vs 25% in PR (CT) and 73% in T12PR vs 23% in PR (TT).
Patients with the CC genotype also experienced an increase in SVR: 90% in T12PR vs 64% in PR.
There was a larger increment in SVR for CT/TT patients with T12PR versus T8PR (71% vs 57% in CT and 73% vs 59% in TT) than for CC patients (90% vs 84%).
Telaprevir-based regimen improved RVR and eRVR rates across all IL28B genotypes compared to pegINF/rbv alone, with the highest rates in CC patients. Most patients with eRVR achieved SVR in all treatment groups and IL28B genotypes: CC (97%), CT (88%), and TT (85%).
CONCLUSIONS
Telaprevir in combination with pegIFN/rbv increased SVR across all IL28B genotypes.
The greatest increment in efficacy occurred in CT/TT patients, CC patients also had improvement in SVR rates up to 90% in T12PR, with the highest rate of RVR and eRVR
CC patients nearly always achieved SVR if they attained RVR & eRVR.
CT & TT patients with RVR or eRVR were highly likely to attain SVR.
Among patients not having eRVR, CT/TT patients had lower SVR rates than CC patients.
Further studies, including non-Caucasians, are needed to confirm these findings.
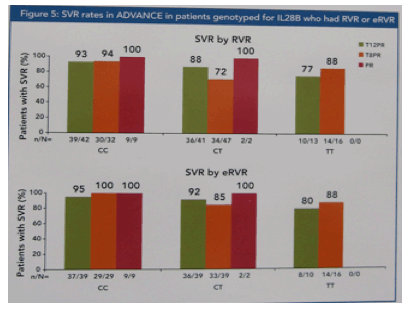
Among patients treated with a telaprevir-based regimen who had eRVR, 91% achieved SVR (97% of CC, 88% of CT, 85% of TT) and were assigned to 24 weeks of therapy.
Among patients treated with a telaprevir-based regimen who did not have eRVR, 43% achieved SVR (63% of CC, 33% of CT, 46% of TT) and were assigned to 48 weeks of therapy.
|
| |
|
 |
 |
|
|