 |
 |
 |
| |
Four-Week Treatment with GS-9256 and Tegobuvir (GS-9190) +/- RBV +/- PEG, Results in Enhanced Viral Suppression on Follow-up PEG/RBV Therapy, in Genotype 1a/1b HCV Patients
|
| |
| |
Four-Week Treatment with GS-9256 and Tegobuvir (GS-9190) +/- RBV +/- PEG, Results in Enhanced Viral Suppression on Follow-up PEG/RBV Therapy, in Genotype 1a/1b HCV Patients

INTRODUCTION
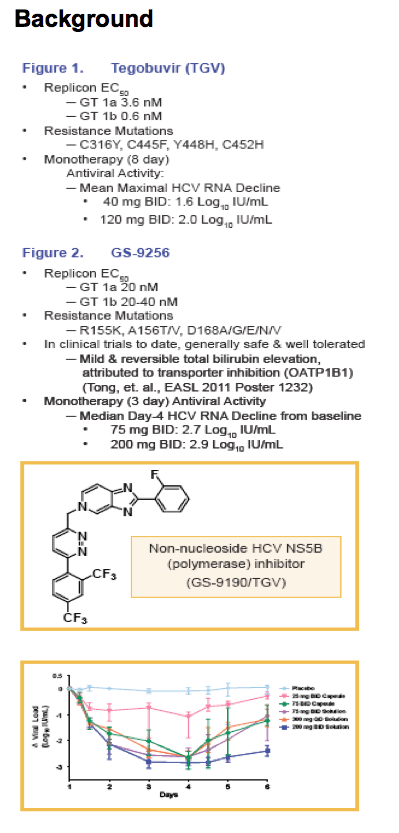
Tegobuvir (TGV) is a non-nucleoside HCV polymerase inhibitor, which interacts with NS5b close to the enzyme active site
TGV markedly enhanced EVR when administered in combination with PegIFN/RBV in Rx-naive GT1 infection (Lawitz, et. al., EASL 2011 Abstract 2396)
GS-9256 is a potent macrocyclic HCV NS3 protease inhibitor that achieved ~3 Log10 IU/mL HCV RNA decline following 3-day monotherapy in treatment naïve HCV GT1 infection (Lawitz, et. al., EASL 2010, Abstract 466)
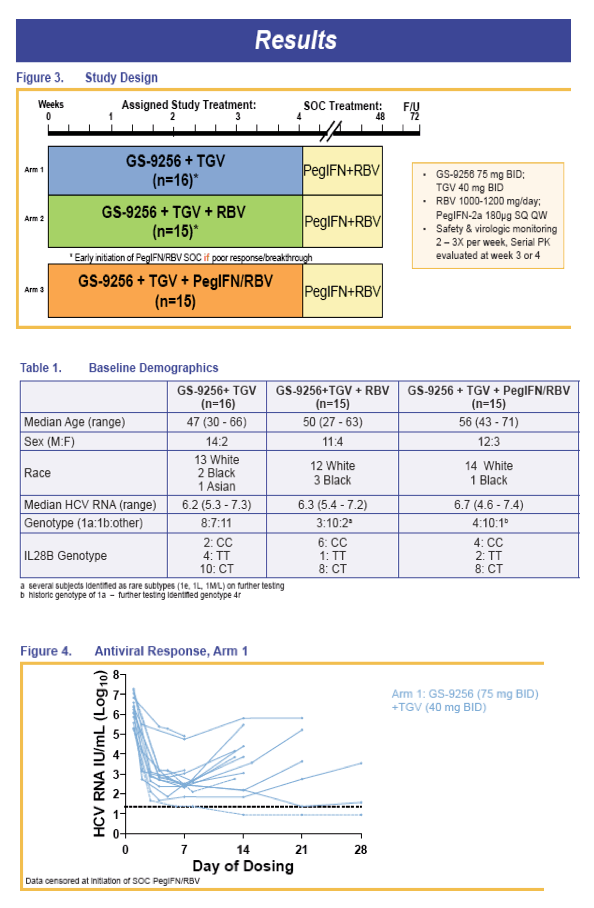
TGV + GS-9256 + PegIFN/RBV achieved 100% RVR when adminstered for 28 days in Rx-naive HCV GT1 infection (Zeuzem, et al, AASLD 2010, Abstract LB1)
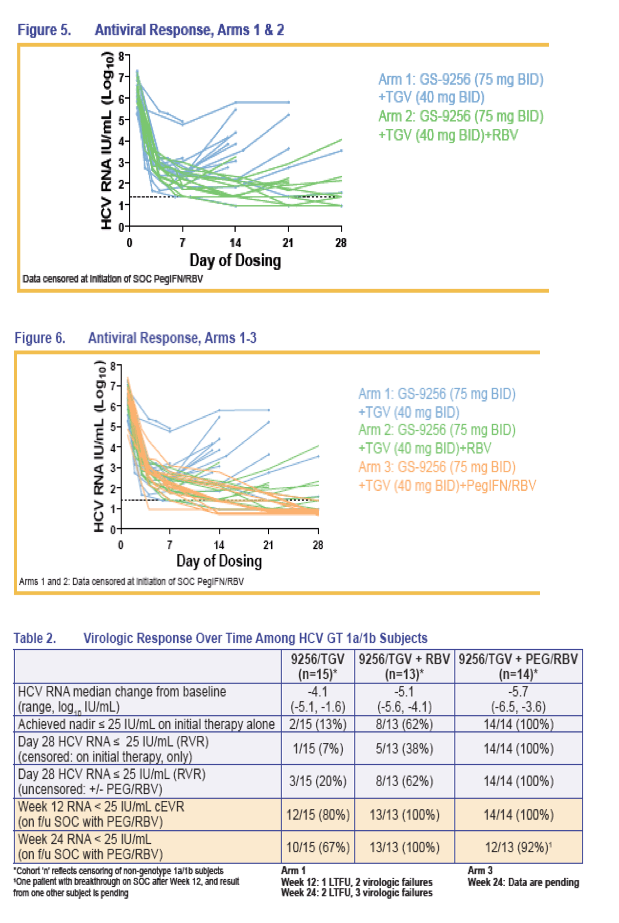
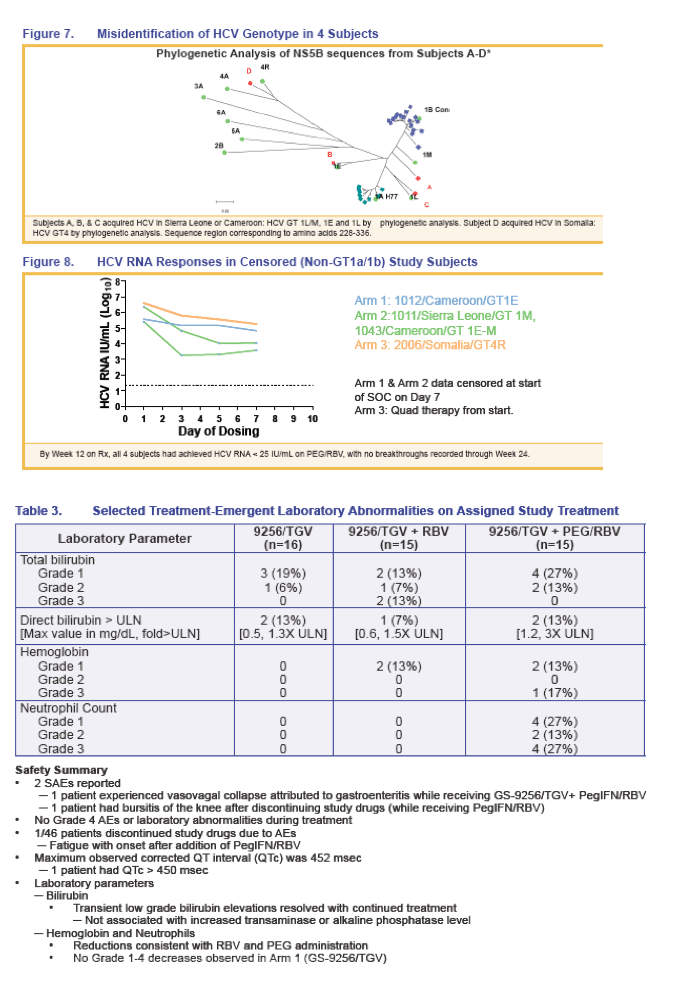
We present the preliminary results of follow-up therapy with PegIFN/RBV for patients treated for up to 28 days with TGV+GS-9256 +/- RBV +/- PegIFN prior to initiation of standard of care (SOC) therapy

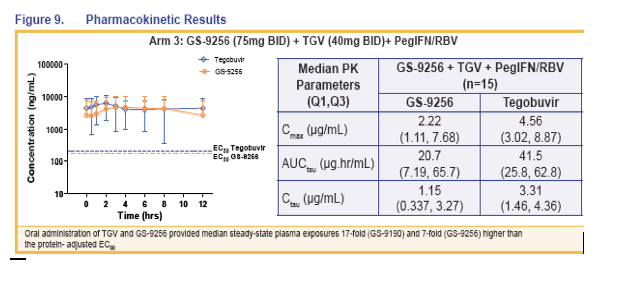
|
| |
|
 |
 |
|
|