 |
 |
 |
| |
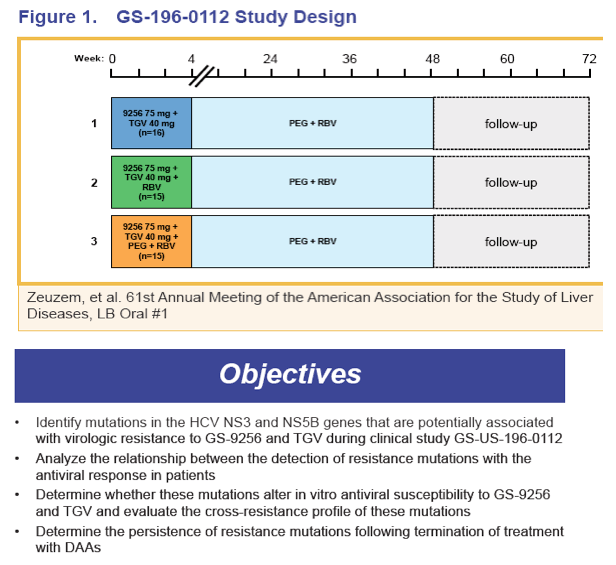
Characterization of Viral Resistance Mutations In Genotype 1 HCV Patients Receiving Combination Therapy with a Protease Inhibitor and a Polymerase Inhibitor with or without Ribavirin Gilead
|
| |
| |
Reported by Jules Levin EASL 2011 Berlin March 31-Apr 2
H. Mo, C. Hebner, B. Han, J. Harris, A. Bae, K. Wong, W. Delaney, D. Oldach and M. D. Miller
Gilead Sciences, Inc. Foster City, CA USA
Author Summary
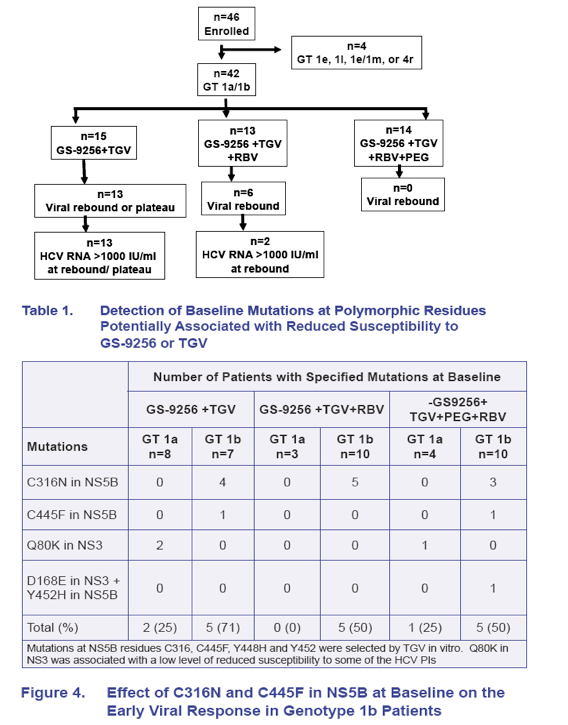
· Population sequence detected C316N, C445F and Y452H NS5B mutations in 12/27 (44%), 2/27 (7%), and 1/27 (3.5%) GT 1b patients at baseline, respectively; Q80K in NS3 was observed in 2/15 (13%) GT 1a patients at baseline (Table 1)
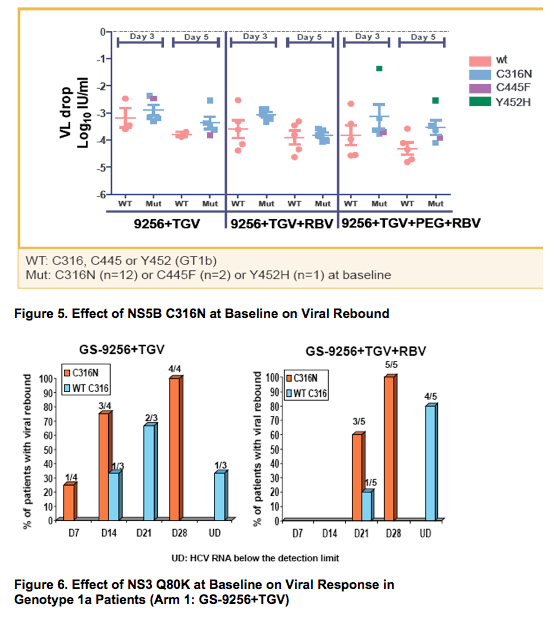
· The presence of C316N and C445F mutations in NS5B and Q80K in NS3 appears to be associated with slightly slower/poorer response and more frequent/earlier viral rebound during GS-9256+GS-9190 +/- RBV therapy (Figures 4, 5 and 6)
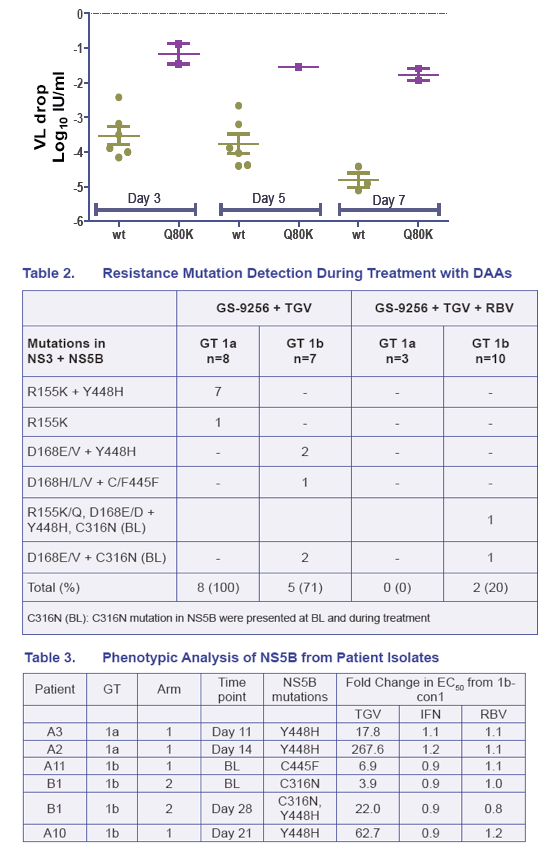
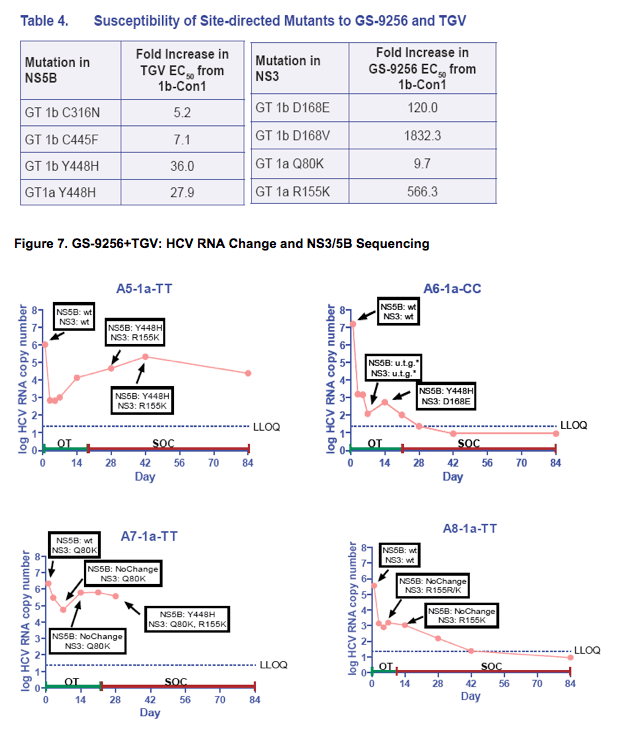
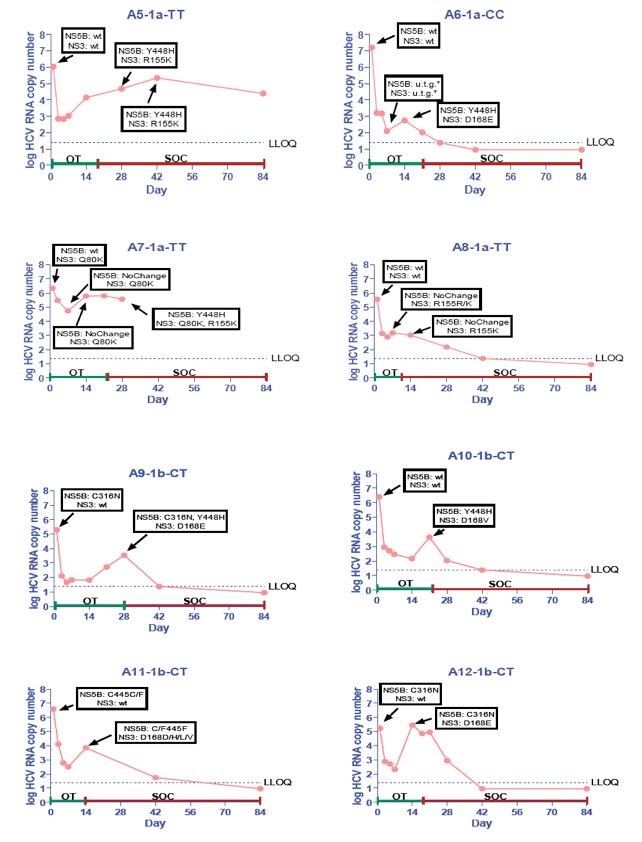
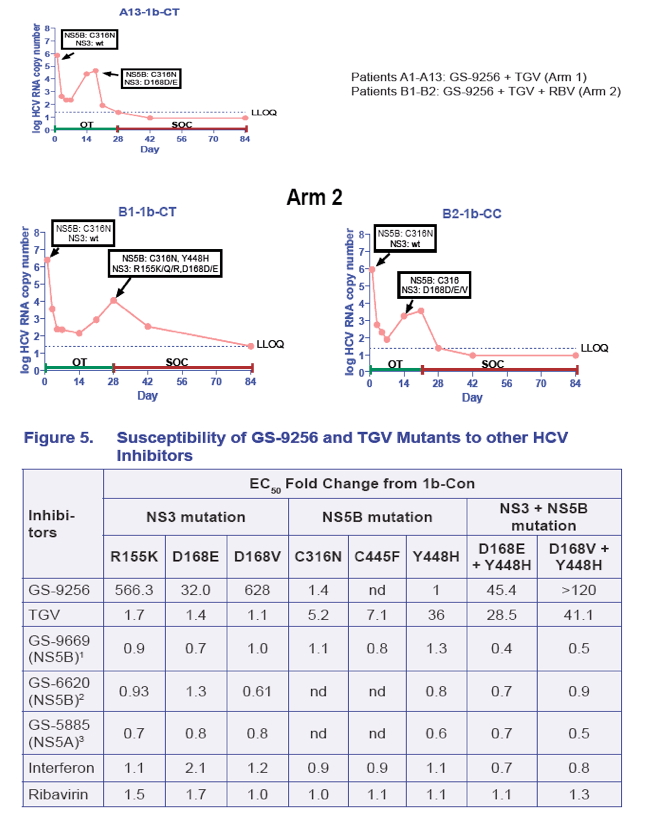
· The viral plateau and viral rebound were associated with the detection of mutations in both NS3 and NS5B in 14/15 rebound patients analyzed (Table 2 and Figure 7)
· NS3 R155K and NS5B Y448H were the most common mutations selected in GT 1a patients; NS3 D168E/V and NS5B Y448H were the most common in GT1b patient (Table 2 and Figure 7)
· In three GT 1b patients with either NS5B C316N or C445F at baseline, the viral rebound was associated with the emergence of NS3 D168E/V mutations without the selection of additional NS5B mutations (Table 2 and Figure 7)
· The GS-9256 and TGV resistance mutations persisted following a short period (2 weeks) of treatment with PEG/RBV, but were suppressed by PEG/RBV therapy in majority of patients (Figure 7)
· Phenotypic analysis showed that the C316N, C445F, and Y448H mutations confer low to moderate levels of resistance to GS-9190 (Table 3 and 4)
· The NS3 Q80K polymorphism in GT 1a showed a low-level reduced susceptibility to GS-9256; however mutations at R155, D168 and A156 conferred high levels of resistance to GS-9256 (Table 4)
· The GS-9256 and GS-9190 resistant mutants tested remain fully susceptible to IFN, RBV, GS-5885 (NS5A inhibitor), GS-6620 (a novel nucleoside inhibitor) and GS-9669 (a novel non-nucleoside inhibitor) (Tables 3 and 5)


References
1. M Fenaux, Y Tian, M Matles, et al. Nonclinical and Cross-Genotypic Profi le of GS-9669, a Novel HCV NS5B Non-Nucleoside Thumb Site II Inhibitor, Poster No. 564, EASL 2011, Berlin, Germany
2. A Ray, J Feng, T Wang, et al. GS-6620, A Liver Targeted Nucleotide Prodrug with Potent Pan-Genotype Anti-Hepatitis C Virus Activity In Vitro, Poster No. 1363, EASL 2011, Berlin, Germany
3. JO Link, R Bannister, LD Beilke, et al. Nonclinical Profi le and Phase I Results in Healthy Volunteers for the Novel and Potent HCV NS5A Inhibitor GS-5885, Poster No. 1883, AASLD 2010, Boston, MA, USA
· GS-9190 project team, GS-9256 project team, Betty Peng, Karin Ku, John McHutchison, William Lee
|
| |
|
 |
 |
|
|