 |
 |
 |
| |
Nucleotide Inhibitor Resistance Selections Using GT2a Infectious Virus: PSI-7851 and GS-6620 Select for A Novel Resistance Pathway Including Substitutions of M289V/L Followed By S282T
|
| |
| |
Reported by Jules Levin EASL 2011 Berlin March 31-Apr 2
G Cheng, M Fenaux, E Mabery, S Eng, B Peng, O Barauskas, L Lagpacan, D Jin, M Hung, X Liu, J Perry, J Feng, A Ray, W Delaney IV and W Zhong
Gilead Sciences, Foster City, CA , USA
Author Conclusions

Introduction
· GS-6620 is a novel nucleotide prodrug and a potent pan-genotype inhibitor of HCV currently undergoing clinical evaluation for treatment of chronic HCV infection1,2
· PSI-7977 (an isomer of PSI-7851) is an investigational nucleotide analog currently in Phase 2 for treatment of chronic HCV infection3
· The nucleotide inhibitor class has demonstrated a high barrier to viral resistance4
Background
· Nucleotide inhibitor (NI) resistance selections are difficult to perform in the HCV replicon system for the following reasons:
- poor replication fitness and the resulting low pre-existing frequency of resistance mutations
- potential to select host cell resistance
- absence of viral spread and amplifi cation
· We previously demonstrated efficient selection of clinically-relevant resistance mutations to HCV protease inhibitors using the genotype (GT) 2a HCV infection system (HCVcc)5
Objectives
· Evaluate the use of HCVcc for nucleotide inhibitor resistance selections
· Characterize and compare the in vitro resistance profiles of the nucleotide inhibitors GS-6620 and PSI-7851 using GT 2a infectious virus
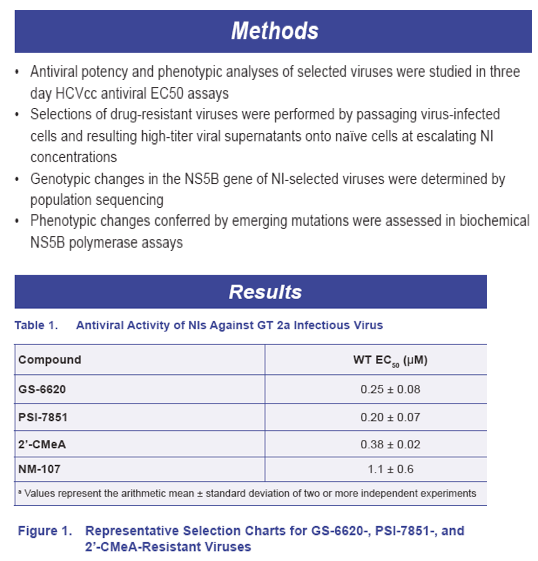
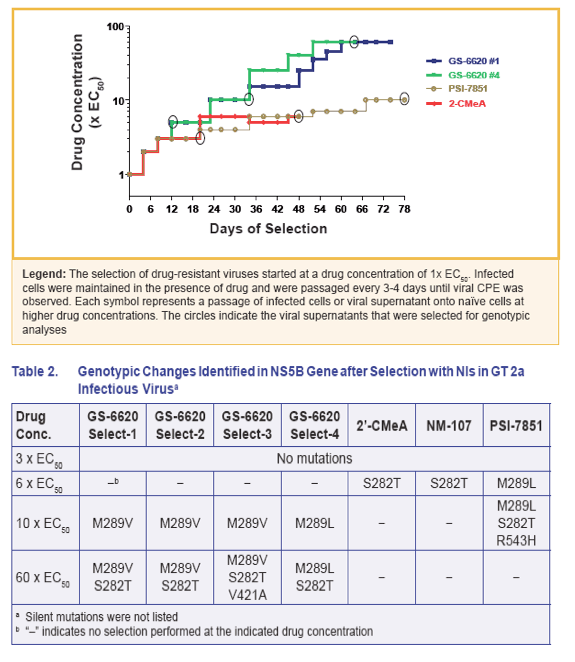
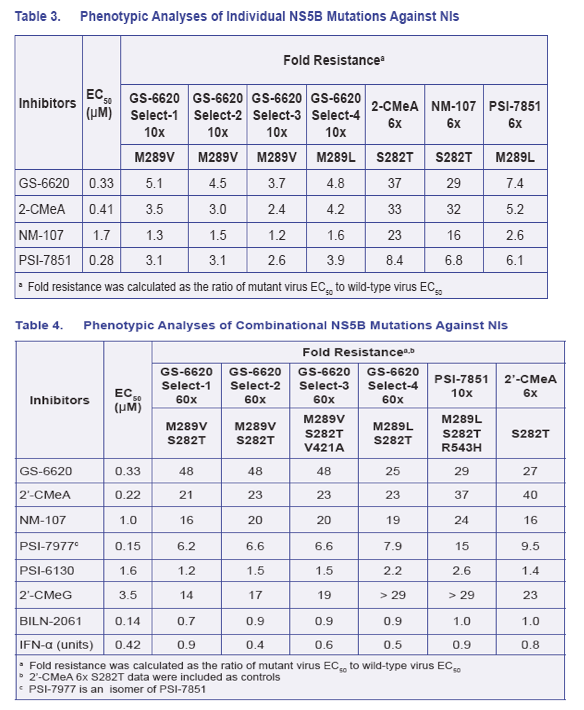
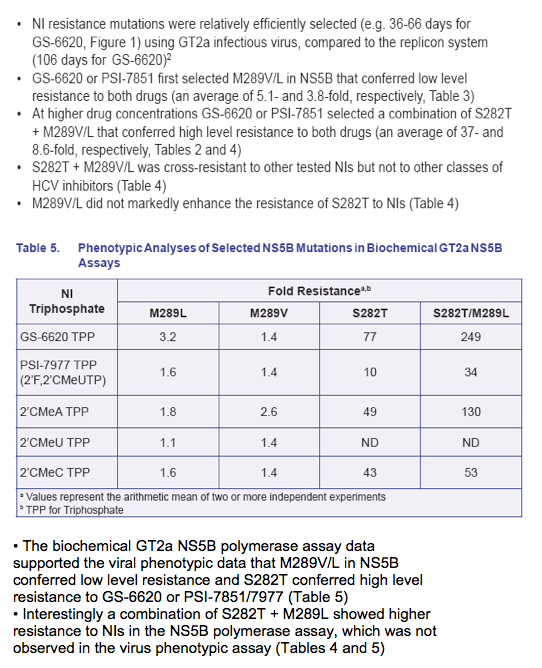
· The biochemical GT2a NS5B polymerase assay data supported the viral phenotypic data that M289V/L in NS5B conferred low level resistance and S282T conferred high level resistance to GS-6620 or PSI-7851/7977 (Table 5)
· Interestingly a combination of S282T + M289L showed higher resistance to NIs in the NS5B polymerase assay, which was not observed in the virus phenotypic assay (Tables 4 and 5)
References
1. Ray A, Feng J, Wang T, et al. GS-6620: A Liver Targeted Nucleotide Prodrug with Potent Pan-Genotype Anti-Hepatitis C Virus Activity In Vitro. Poster No. 1233, EASL 2011
2. Fenaux M, Cheng G, Mabery E, et al. GS-6620, a Novel Anti-hepatitis C Virus Nucleotide Prodrug, Has a High In Vitro Barrier to Resistance. Poster No. 1205, EASL 2011
3. Lawitz E, Lalezari J, Rodriguez-Torres M, et al. High Rapid Virologic Response (RVR) with PSI-7977 Daily Dosing plus PEG-IFN/RBV in a 28-day Phase 2a Trial. 61st AASLD, Boston, MA Oct 29-Nov 2, 2010
4. McCown MF, Rajyaguru S, Le Pogam S, Ali S, Jiang WR, Kang H, et al. The hepatitis C virus replicon presents a higher barrier to resistance to nucleoside analogs than to nonnucleoside polymerase or protease inhibitors. Antimicrob Agents Chemother 2008;52 (5):1604-12.
5. Cheng G, Chan K, Yang H, et al. Selection of Clinically-relevant Protease Inhibitor Resistant Viruses Using the Genotype 2a HCV Infection System Antimicrob Agents Chemother, 2011 in press
|
| |
|
 |
 |
|
|