 |
 |
 |
| |
Preclinical Characterization of GS-9620, A Potent and Selective Oral TLR7 Agonist
|
| |
| |
Reported by Jules Levin EASL 2011 March 31-Apr 2 Berlin Germany
D Tumas1, X Zheng2, B Lu2, G Rhodes2, P Duatschek3, J Hesselgesser3, C Frey3, I Henne1, A Fosdick1, R Halcomb4, and G Wolfgang1
1Departments of Drug Safety Evaluation, 2Drug Metabolism, 3Biology, and 4Medicinal Chemistry, Gilead Sciences, Inc., 333 Lakeside Drive, Foster City, CA 94404, USA
Introduction
· Though antiviral treatments can suppress Hepatitis C virus (HCV) and Hepatitis B virus (HBV) viral load in chronic infection, novel strategies to enhance long term viral clearance and sustained immunological control represent a significant unmet need
· GS-9620 is a potent oral TLR7 agonist being developed for the treatment of chronic HBV and HCV
· The goal of GS-9620 treatment is to stimulate an innate antiviral response and enhance an antiviral adaptive immune response
· In vitro and in vivo studies were performed to characterize the selectivity, activity, pharmacodynamics (PD), pharmacokinetics (PK), and tolerability of GS-9620

Background
· Oral administration of GS-9620 caused lasting reduction in viral load and S-antigen and induction of anti-S antigen antibodies in the woodchuck model of HBV infection (Poster/Abstract 170)
· Oral administration of GS-9620 for 8 weeks reduced serum and liver viral DNA in HBV infected chimpanzees with a mean maximal reduction in serum viral load of 2.2 logs and induced dose dependent increases in serum IFN-α, ISGs in PBMCs and liver, and the activation of lymphocyte subsets (CD8+ T and NK cells) (Oral/Abstract 1771)
· GS-9620 was safe and well tolerated in oral single ascending doses up to 12 mg in healthy volunteers and had pharmacodynamic effects beginning at 2 mg. (Poster/Abstract 664)
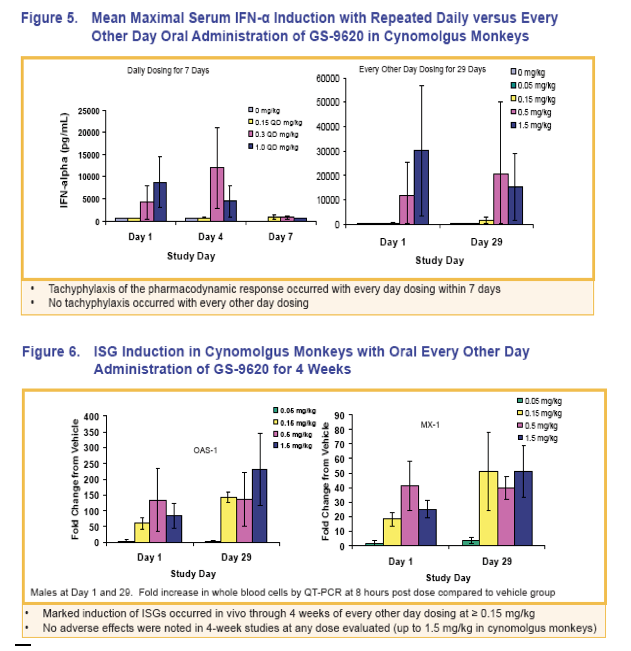
|
| |
|
 |
 |
|
|