 |
 |
 |
| |
Low prevalence of danoprevir resistance identified in genotype 1b HCV patients with prior null response treated with danoprevir plus low-dose ritonavir plus peginterferon alfa-2a (40KD)/ribavirin for 12 weeks
|
| |
| |
Reported by Jules Levin
Presented at the International Liver Congress 2011 [46th Annual Meeting of the European Association for the Study of the Liver (EASL)], March 30-April 3, 2011, Berlin, Germany
S. Le Pogam,1 J.M. Yan,1 M. Chhabra,2 M. Ilnicka,1 D. Chin,1 Y. Ji,2 Y. Zhang,3 N. Shulman,3 K. Klumpp,1 I. Najera1
1Roche, Nutley, NJ, USA; 2Roche, Palo Alto, CA, USA; 3Roche, South San Francisco, CA, USA
EASL: Activity of danoprevir plus low-dose ritonavir in combination with peginterferon alfa-2a (40KD) plus ribavirin in previous null responders - (04/01/11)
Conclusions
· In treatment naive patients danoprevir/r at a dose of 100/100 mg q12h, 200/100 mg q12h or 200/100 mg q24h plus peginterferon alfa-2a (40KD)/
ribavirin provided a robust virological response in treatment-naive genotype 1b and 1a patients with no viral breakthrough observed.
· In prior null responders, robust response was observed in genotype 1b patients, with only 1 of 16 patients showing evidence of
resistance-related viral breakthrough, whereas resistance-related viral breakthrough was observed in 4 of 8 genotype 1a patients.
· R155K resistance mutation persisted in three of the genotype 1a patients, while one genotype 1a patient lost the R155K resistance
mutation at the last time point of follow-up (follow-up week 24, 240 days after stopping the danoprevir treatment).
· In prior null responders there may be a need for a more intensified regimen including a second direct-acting antiviral, particularly one
with a high barrier to resistance.
Introduction
The combination of a hepatitis C virus (HCV) NS3/4A protease inhibitor (PI) in combination with peginterferon plus ribavirin has the potential to increase cure rates and reduce the required duration of treatment for patients with HCV genotype 1 infection, including treatment-naive and previous non-responders to peginterferon plus ribavirin.[1 8]
Viral resistance has emerged during treatment and is associated with viral breakthrough in patients treated with a PI. Danoprevir is a potent, highly selective and well-tolerated oral HCV NS3/4A protease inhibitor that has been studied, not only in combination with peginterferon alfa-2a (40KD) plus ribavirin,[9] but also in combination with mericitabine (RG7128), a nucleoside analogue inhibitor of the HCV NS5B polymerase in the proof of concept study of a dual oral interferon-free regimen in patients with chronic hepatitis C (INFORM-1 study).[10]
The resistance profile of danoprevir has been characterised and, unlike other HCV protease inhibitors, is largely restricted to a single amino acid substitution in the NS3 protease sequence (R155K), as observed in a 14-day monotherapy study,[11] and in a 12-week combination therapy study of danoprevir plus peginterferon alfa-2a (40KD) and ribavirin.[12] In contrast, when administered for 2 weeks in combination with mericitabine in INFORM-1, R155K was not detected in any patient, suggesting a role for mericitabine in preventing resistance to danoprevir.[10]
Objective
The objective of this study was to monitor the potential emergence of resistance to danoprevir in HCV genotype 1 patients, including both treatment-naive and prior null responders to peginterferon alfa-2a (40KD) plus ribavirin treatment, before, during and after treatment for up to 12 weeks with danoprevir plus low-dose ritonavir (r) in combination with peginterferon alfa-2a (40KD) plus ribavirin.
Methods
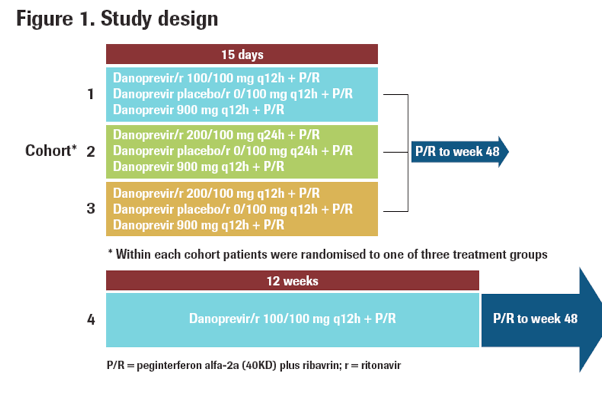
Three cohorts of treatment-naive HCV genotype 1 patients (n=34; 24 genotype 1a; 10 genotype 1b) were randomised to receive danoprevir/r, danoprevir (900 mg q12h) or placebo/r in combination with peginterferon alfa-2a (40KD) (PEGASYS®) plus ribavirin
(COPEGUS®) for 15 days (Figure 1). There were three danoprevir/r dosage cohorts:
- Cohort 1: danoprevir/r 100/100 mg q12h
- Cohort 2: danoprevir/r 200/100 mg q24h
- Cohort 3: danoprevir/r 200/100 mg q12h
A fourth cohort (n=24; 8 genotype 1a and 16 genotype 1b) included HCV genotype 1 patients with a prior null response to peginterferon/ribavirin who were re-treated with 100/100 mg q12h danoprevir/r in combination with peginterferon alfa-2a (40KD) plus
ribavirin for 12 weeks (Figure 1).
In cohorts 1 to 3 serum HCV RNA was measured by the Roche COBAS® TaqMan® HCV Test (detection limit = 15 IU/mL) at baseline and at days 1, 2, 3, 6, 9, 12, 14, 15 and 16 of danoprevir/r, danoprevir, or placebo/r plus peginterferon alfa-2a (40KD)/ribavirin treatment and also at week 4, 12 and 24 during follow-up treatment with peginterferon alfa-2a (40KD) plus ribavirin.
In cohort 4, serum HCV RNA was measured by the Roche COBAS® TaqMan® HCV Test (detection limit = 15 IU/mL) at baseline and at days 1, 2, 3, 6, 9, 12, 14, 15 and 16, and week 4, 8 and 12 of danoprevir/r + peginterferon alfa-2a (40KD)/ribavirin
treatment, at week 16, 20, 24, 36 and 48 of peginterferon alfa-2a (40KD)/ribavirin follow-up treatment, and at week 52, 60 and 72 during the follow-up treatment-free period (i.e. follow-up weeks 4, 12 and 24, respectively).
Sample selection
Population sequencing and phenotypic analyses were performed for both on treatment and follow-up samples from patients who received danoprevir/r plus peginterferon alfa-2a (40KD) plus ribavirin and who experienced one of the following:
- Viral breakthrough during danoprevir/r treatment, defined as a sustained increase (i.e. for two or more consecutive measurements) in viral load of ≥1 log10 IU/mL above a nadir that was ≥0.5 log10 IU/mL below baseline.
- Partial response during danoprevir/r treatment, defined as an initial decrease in viral load to a nadir of ≥0.5 log10 IU/mL below baseline, followed by stabilisation (i.e. at least three consecutive measurements within 0.5 log10 IU/mL of nadir), and/or an end of treatment viral load of >1000 IU/mL in a patient who has received at least 4 weeks of treatment.
-- Non-response during danoprevir/r treatment, defined as a decrease in viral load of <0.5 log10 IU/mL in a patient who has received at least 2 weeks of treatment.
Sequencing
Amplification and population sequencing covering the entire NS3/4A and/or NS3 protease coding regions was performed on baseline samples from all patients, and on-treatment and follow-up samples from selected patients treated with danoprevir/r.
Phenotyping
Phenotypic characterisation was performed by cloning the NS3 protease gene from samples collected at day 1 and at the point after viral load rebound (viral breakthrough patients) and/or at the end of danoprevir treatment (partial responders) into the NS3 protease shuttle replicon and measuring the susceptibility to danoprevir and to peginterferon alfa-2a (40KD).
Results
A total of 58 patients were enrolled, of whom 53 received either danoprevir/r + peginterferon alfa-2a (40KD)/ribavirin (25 treatment-naive patients and 24 treatment-experienced patients) or danoprevir + peginterferon alfa-2a (40KD)/ribavirin (n=4; all treatment-naive patients).
Overall danoprevir/r at a dose of 100/100 mg q12h and 200/100 mg q12h or q24h plus peginterferon alfa-2a (40KD) plus ribavirin elicited a robust virological response.
Treatment-naive patients (cohorts 1-3)
· In cohorts 1-3 all 29 treatment-naive patients that received either danoprevir/r plus peginterferon alfa-2a (40KD)/ribavirin or danoprevir plus
peginterferon alfa-2a (40KD)/ribavirin experienced a continuous viral load decline; none experienced viral breakthrough, partial response or non-response during 2 weeks of combination therapy (20 genotype 1a; 9 genotype 1b).
Treatment-experienced patients (cohort 4)
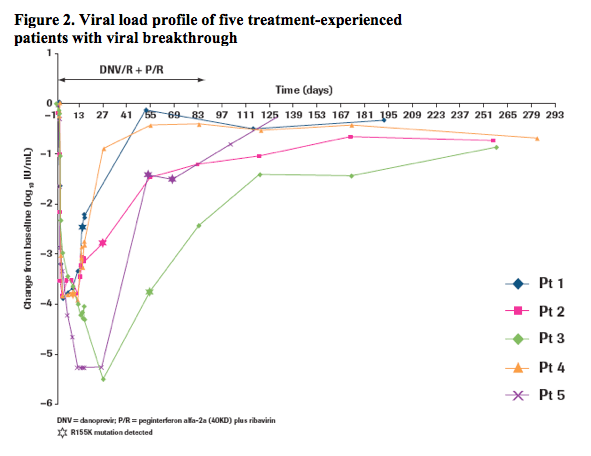
· Of the 24 prior null responder patients (8 genotype 1a; 16 genotype 1b) treated with 100/100 mg danoprevir/r + peginterferon alfa-2a (40KD)/ribavirin, viral breakthrough was observed in 5 individuals (4 genotype 1a, 1 genotype 1b), all of whom had a baseline viral load >6 log10 IU/mL (range 6.44-7.27 log10 IU/mL).
· Viral breakthrough was observed by week 3 in two genotype 1a patients, by week 4 in one genotype 1a patient, by week 8 in one genotype 1a and by week 8 in one genotype 1b patient (Figure 2).
· Viral breakthrough was associated with the selection of the R155K variant in the NS3 protease region in all cases (Table 1).
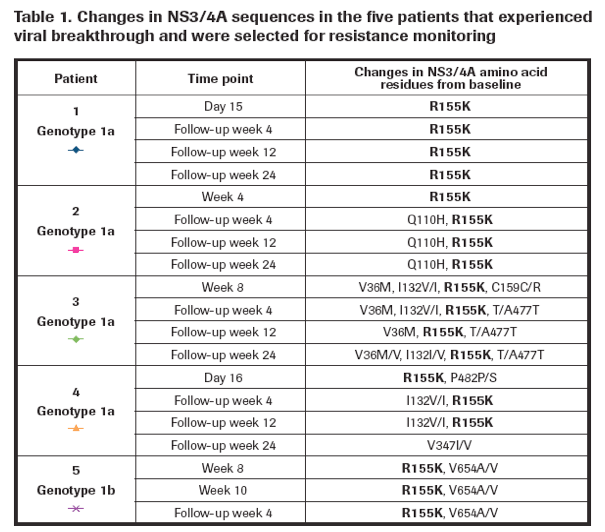
Emergence of the R155K variant was associated with at least a 218-fold shift in EC50 for danoprevir compared with baseline samples (Figure 3).
The R155K resistance mutation persisted through week 24 of the follow-up period after danoprevir had been discontinued in three genotype 1a patients.
The R155K resistance mutation persisted through at least week 4 of the follow-up period for genotype 1b patients (only data available at the time of reporting) (Table 1).
Interestingly, one (1/4) genotype 1a patient (patient 4 in Table 1) lost the R155K resistance at the last time point of follow-up (week 24), which corresponds to 240 days after stopping danoprevir treatment.
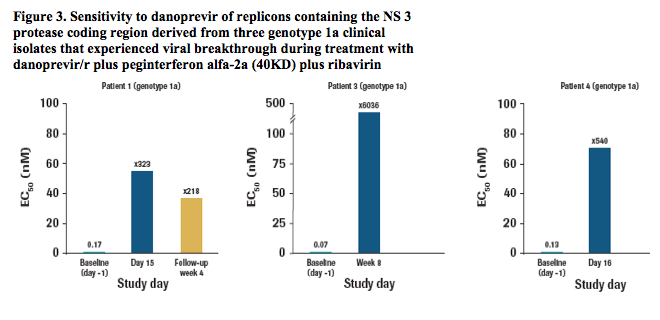
Actual EC50 values are shown for day -1 samples, and the fold-change in EC50 relative to day -1 is shown for on-treatment and follow-up time points. EC50 determination was not possible for samples from patient 2 at the week 4 time point due to very low replication capacity. Baseline sample for this patient (patient 2; day -1) was sensitive to danoprevir (0.13 ± 0.012 nM). Phenotypic characterisation of the genotype 1b samples was not available at the time of reporting.
Analysis of baseline samples in all patients (cohorts 1-4)
Population sequence analysis of the NS3/4A region from baseline samples from all 58 recruited patients did not identify any pre-existing danoprevir resistance mutations at baseline.
References
1. McHutchison JG, Everson GT, Gordon SC, et al. Telaprevir with peginterferon and ribavirin for chronic HCV genotype 1 infection. N Engl J Med 2009; 360: 1827-38
2. Hezode C, Forestier N, Dusheiko G, et al. Telaprevir and peginterferon with or without ribavirin for chronic HCV infection. N Engl J Med 2009; 360: 1839-50
3. McHutchison JG, Manns MP, Muir AJ, et al. Telaprevir for previously treated chronic HCV infection. N Engl J Med 2010; 362: 1292-1303
4. Kwo PY, Lawitz EJ, McCone J, et al. Efficacy of boceprevir, an NS3 protease inhibitor, in combination with peginterferon alfa-2b and ribavirin in treatment-naive patients with genotype 1 hepatitis C infection (SPRINT-1): an open-label, randomised, multicentre phase 2 trial. Lancet 2010; 376: 705-16
5. Sherman K, Flamm S, Afdhal N, et al. Telaprevir in combination with peginterferon alfa2a and ribavirin for 24 or 48 weeks in treatment-naive genotype 1 HCV patients who achieved an extended rapid viral response: final results of Phase 3 illuminate study [abstract LB-2]. Hepatology 2010; 52 (Suppl 1): 401A-2A
6. Jacobson I, McHutchison J, Dusheiko G, et al. Telaprevir in combination with peginterferon and ribavirin in genotype 1 HCV treatment-naive patients: final results of phase 3 ADVANCE study [abstract 211]. Hepatology 2010; 52 (Suppl 1): 427A
7. Bacon B, Gordon S, Lawitz E, et al. HCV RESPOND-2 final results: high sustained virologic response among genotype 1 previous non-responders and relapsers to peginterferon/ribavirin when re-treated with boceprevir plus Pegintron (peginterferon alfa-2b)/ribavirin [abstract 216]. Hepatology 2010; 52 (Suppl 1): 430A
8. Bronowicki J-P, McCone J, Bacon B, et al. Response-guided therapy (RGT) with boceprevir (BOC) + peginterferon alfa-2b/ribavirin (P/R) for treatment-naive patients with hepatitis C virus (HCV) genotype (G) 1 was similar to a 48-wk fixed-duration regimen with BOC + P/R in SPRINT-2 [abstract LB-15]. Hepatology 2010; 52 (Suppl 1): 881A
9. Gane E, Rouzier, R, Stedman C, et al. Ritonavir boosting of low dose RG7227/ITMN-191, HCV NS3/4A protease inhibitor results in robust reduction in HCV RNA at lower exposures than provided by unboosted regimens [abstract 38]. J Hepatol 2010; 52 (Suppl 1): S16-17
10. Gane EJ, Roberts SK, Stedman CA, et al. Oral combination therapy with a nucleoside polymerase inhibitor (RG7128) and danoprevir for chronic hepatitis C genotype 1 infection (INFORM-1): a randomised, double-blind, placebo-controlled, dose-escalation trial. Lancet 2010; 376: 1467-75
11. Sarrazin C, Lim S, Qin X, et al. Kinetic analysis of viral rebound and drug-resistant viral variant dynamics in patients treated with ITMN-191 (R7227) monotherapy suggests a high barrier to viral escape [abstract 1411]. Hepatology 2009; 50 (Suppl. 4): 953A.
12. Le Pogam S, Chhabra M, Yan JM, et al. Low rate of viral load rebound observed among treatment-naive genotype 1 patients with chronic hepatitis C treated with danoprevir (RG7227) plus Peg IFN α-2a (40KD) (PEGASYS®) plus ribavirin: interim analysis [abstract 802]. Hepatology 2010; 52 (Suppl 1):703A.
|
| |
|
 |
 |
|
|