 |
 |
 |
| |
Idenix NS5A HCV replication inhibitors with low
picomolar, pan-genotypicin vitro antiviral activity
|
| |
| |
Reported by Jules Levin EASL 2011 Berlin Germany March 30-Apr 2
Cyril B. Dousson, Massimiliano La Colla, Christopher Chapron, John P. Bilello, Joseph F. McCarville, Maria Seifer, Christophe Parsy, David Dukhan, Claire Pierra, Dominique Surleraux, David N. Standring
Idenix Pharmaceuticals, Inc., Cambridge, MA, USA and Montpellier, France
AUTHOR CONCLUSIONS
IDX380 and IDX719 were selected as lead preclinical candidates based on their overall preclinical profile to date.
These leads had low picomolar potency (< 50 pM) against HCV genotypes 1a, 1b, 2a, 3a, 4a and 5a with high selectivity indices in vitro.
Compounds had favorable pharmacokinetic properties including long half lives resulting in exposures 4,400- to 15,000-fold higher than EC50 24 hours after a single oral dose in the monkey.
Previous in vitro studies2 suggest that effective NS5A compounds in combination with direct-acting HCV antivirals from other classes, e.g., nucleoside inhibitors and protease inhibitors, will provide moderately to strongly synergistic antiviral effects.
IND/CTA enabling studies have been initiated for both IDX380 and IDX719. The early results will be used to select the final clinical drug candidate.
BACKGROUND
The NS5A protein, involved in replication and maturation, is essential to the life cycle of the hepatitis C virus (HCV) and provides an important antiviral drug target.
Our HCV drug discovery program has identified a number of NS5A-targeted compounds from different structural series that inhibit the replication of multiple HCV genotypes with picomolar (pM) potencies.
From this broad-based discovery effort, we have selected two NS5A-targeted preclinical lead candidates, IDX380 and IDX719. Here we focus on the in vitro and in vivo characterization of these compounds.
METHODS
Compounds were initially evaluated against a luciferase-tagged genotype 1b replicon in a standard assay. Active compounds were then tested in a genotype 1a replicon assay.
Intergenotypic replicons were generated by introducing the NS5A protein sequence from HCV of genotypes 1a, 2a, 3a and 4a into a genotype 1b backbone. Genotype 5a intergenotypic replicon was constructed by introducing the first 100 amino acids of genotype 5a NS5A protein into a genotype 1b backbone.
Compounds with potent activity against the NS5A genotype 1a and 1b replicons were further evaluated against the intergenotypic replicons.
The activities of IDX380 and IDX719 were evaluated against genotype 1a and 1b replicons bearing mutations at NS5A loci (Q30, L31 and Y93) that have been reported1 to confer resistance to the NS5A inhibitor class of compounds. Mutant replicons were generated by site-directed mutagenesis.
IDX380 and IDX719 were tested for inhibition of cytochrome P450 enzymes and for compound stability in simulated gastric fluid (pH 1.2) with pepsin and 30% PEG200 and simulated intestinal fluid (pH 6.9) with pancreatin and 30% PEG200.
IDX380 and IDX719 were also examined for cell permeability, protein-binding and PK characteristics in rodents and monkeys.
RESULTS
In Vitro Biological Activity
Compounds potently active against HCV replicons of multiple genotypes were identified from several distinct molecular scaffolds. The in vitro activities against genotypes 1-5 of the two lead candidates compared favorably to those of BMS-790052 (Tables 1 and 2 and Figure 1). CC50 values > 100 μM and EC50 values < 50 pM provided selectivity indices greater than 2 million.
When a given mutation was inserted into both genotype 1a and genotype 1b replicons, IDX380 and IDX719 were generally more active against genotype 1b replicons (7- to 94-fold wild type EC50) than against genotype 1a replicons (310- to 8,600-fold wild type EC50).
Fold-change and absolute EC50 values compared favorably to those of BMS-790052 for all mutants (Table 3).
IDX380 and IDX719 produced absolute EC50 values against the genotype 1a mutants approximately 2-fold to over 30-fold lower than BMS-790052
EC50 values of IDX380 and IDX719 against genotype 1b mutants ranged from nearly 2-fold to over 12-fold lower than BMS-790052.
The NS5A class of compounds shows low fold-shifts (typically 3-fold to < 10-fold) when tested against genotype 1b replicons in 40-45% human serum (data not shown).
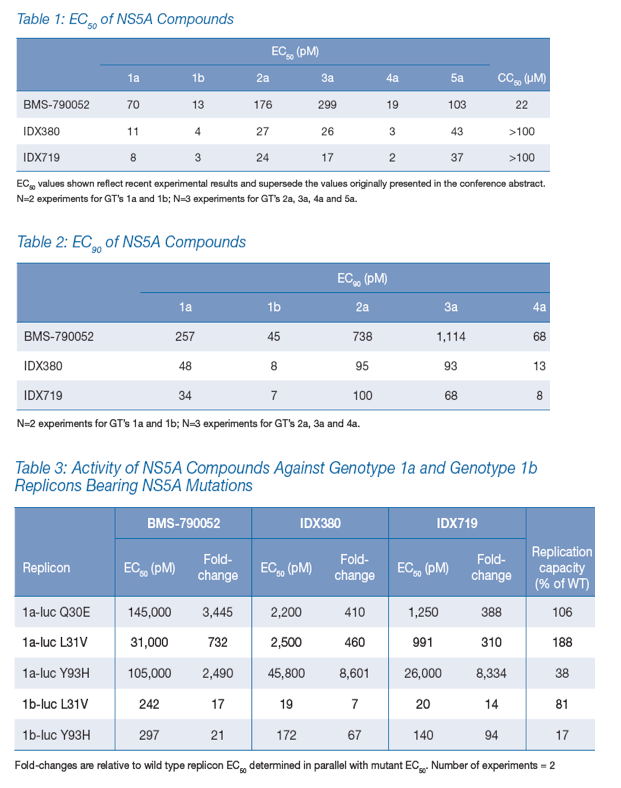
Pharmacological and Chemical Characteristics
The lead candidates did not inhibit seven CYP450 isozymes (Table 4).
IDX380 and IDX719 both remained chemically stable in simulated gastric fluid for over 2 hours and in simulated intestinal fluid for over 4 hours.
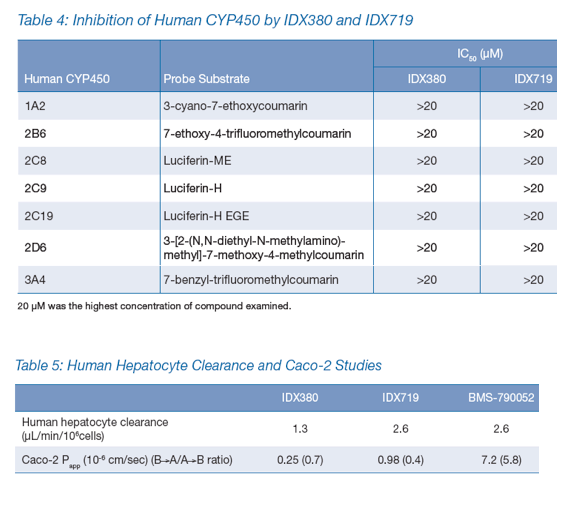
Testing in human hepatocytes shows moderate clearance, similar to BMS-790052; the Caco-2 assay shows low permeability of IDX380 and IDX719 relative to BMS-790052, but no P-glycoprotein efflux (Table 5).
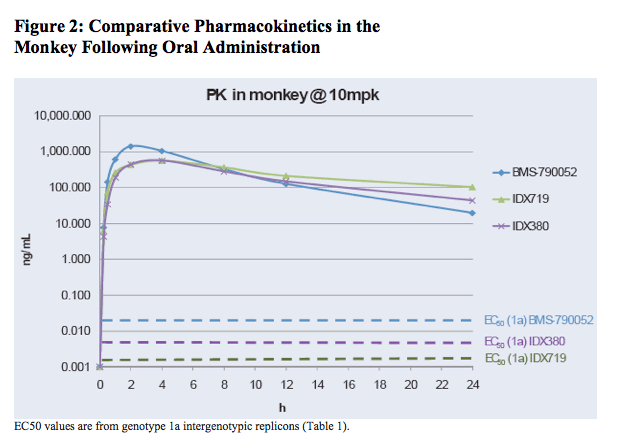
In the monkey, drug exposure levels remained far above in vitro genotype 1a EC50 values at 24 hours after an oral dose at 10 mg/kg, with IDX380 at 4,400X and IDX719 at 15,000X their respective EC50's (Figure 2). BMS-790052 exposure levels at 24 hours post-dose were 400X its genotype 1a replicon EC50.
References
1. Fridell RA, Qiu D, Wang C, et al. (2010). Antimicrob. Agents Chemother. 54:3641-3650.
2. La Colla M, Lallos LB, Serra I, et al. (2010). 45th Annual Meeting of the European Association for the Study of the Liver. April 14-18, 2010; Vienna.
|
| |
|
 |
 |
|
|