 |
 |
 |
| |
SVR and pharmacokinetics of the HCV protease inhibitor BI 201335 with PegIFN/RBV in HCV genotype-1 patients with compensated liver cirrhosis and non-response to previous PegIFN/RBV
|
| |
| |
Reported by Jules Levin EASL 2011 March 31-Apr 2 Berlin Germany
Stanislas Pol,1 Thomas Berg,2 Maurizio Bonacini,3 Marcus Schuchmann,4 Jacob Lalezari,5 Andreas Erhardt,6 Marc Bourliere,7 Michael Manns,8 Jerry Stern,9 Gerd Steinmann,10 Chan-Loi Yong,9 Joe Scherer,9 Wulf O. Boecher10
1Hôpital Cochin, Paris, France; 2Charité, Campus Virchow-Klinikum, Berlin, Germany; 3CPMC Research Institute, San Francisco, CA, USA; 4Internal Medicine 1, University Hospital Mainz, Germany; 5Quest Clinical Research, San Francisco, CA, USA; 6Internal Medicine, University Hospital Duesseldorf, Germany; 7Hôpital Saint Joseph, Marseilles, France; 8Department of Gastroenterology, Hepatology and Endocrinology, Hannover Medical School, Hannover, Germany; 9Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT, USA; 10Boehringer Ingelheim Pharma GmbH, Biberach, Germany

ABSTRACT
Background: BI 201335 is a highly potent and specific hepatitis C virus (HCV) NS3/4A protease inhibitor with good safety and antiviral activity in combination with peginterferon/ribavirin (PegIFN/RBV) in HCV genotype-1 patients with compensated liver cirrhosis and PegIFN/RBV nonresponse (Pol S, AASLD 2009). Here, we describe the sustained virological response (SVR) and pharmacokinetics after additional 44 weeks of PegIFN/RBV in this open-label, sequential group phase I trial.
Methods: Patients were treated for 28 days with 240 mg once daily (QD) or twice daily (BID) of BI 201335 in combination with PegIFN/RBV. All patients were offered continuation with PegIFN/RBV to Week 48. Plasma HCV RNA was measured by Roche COBAS TaqMan with a lower limit of quantification of 25 IU/mL. Plasma concentration of BI 201335 was measured by HPLC-MS and compared to levels in identically treated noncirrhotic patients.
Results: Mean age was 54 years, body mass index 26 kg/m2, viral load 6.2 log10 (IU/mL). No breakthroughs were observed during 28 days' treatment with BI 201335/PegIFN/RBV, while most virological failures occurred as breakthroughs on PegIFN/RBV (Table). SVR rates per-protocol were 3/6 and 1/6 patients at 240 mg QD and BID, respectively, with 1 patient still in follow-up. Serious adverse events were those typical of PegIFN/RBV. At the 240 mg BID dose, BI 201335 exhibited a supra-proportional increase in exposure, which was approximately two times increased compared to noncirrhotic patients.
Conclusions: BI 201335 combined with PegIFN/RBV, exhibited potent antiviral efficacy in nonresponder patients with liver cirrhosis, similar to that observed in non-cirrhotic patients. Thus, efficacy, safety and tolerability support the inclusion of patients with compensated liver cirrhosis into the ongoing phase II and III programme.
INTRODUCTION
· BI 201335 is a highly potent and specific hepatitis C virus (HCV) NS3/4A protease inhibitor
· A phase I trial in HCV genotype-1 (GT-1) patients without liver cirrhosis after previous virological failure to peginterferon alfa (PegIFN) 2a and ribavirin (RBV), demonstrated 47% and 40% sustained virological response (SVR) for 240 mg BI 201335 given once daily (QD) and twice daily (BID), respectively, in combination with PegIFN/RBV for 28 days, followed by PegIFN/RBV for a further 44 weeks1
· Here we describe a phase Ib trial which assessed safety, efficacy, and pharmacokinetics (PK) of BI 201335 in GT-1 patients with compensated liver cirrhosis and virological nonresponse to previous PegIFN/RBV, a difficult-to-treat HCV population with a high unmet medical need
METHODS
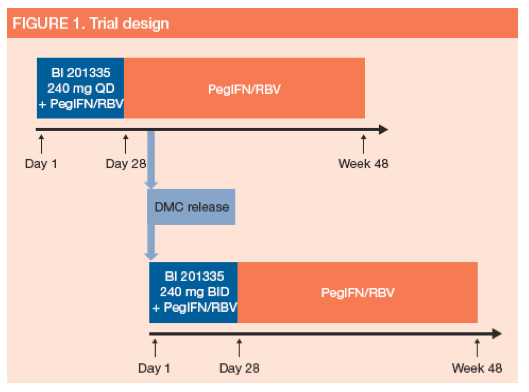
· This trial was an open label, sequential group, dose-escalating comparison of 240 mg BI 201335 QD (n=6) or twice daily (BID; n=7), given orally as a soft gelatin capsule for 28 days in combination with PegIFN alfa 2a (180 μg/week subcutaneous) and RBV (weight based: 1,000 or 1,200 mg daily) (Figure 1)
-- all patients received a single loading dose of 480 mg BI 201335 as the first dose
-- the 240 mg BID cohort started enrolment after review of safety and efficacy data from the 240 mg QD cohort, and release by a Data Monitoring Committee (DMC)
· HCV GT-1 patients were entered if they had compensated biopsy-proven liver cirrhosis (Child-Pugh-Turcotte Score A; CPT A) and had never achieved undetectable viral load (VL) under previous PegIFN/RBV treatment for at least 12 weeks
· Only patients with partial response (maximum VL reduction >1 log10 from baseline but who never achieved undetectable HCV RNA at any time) or null response (maximum VL reduction <1 log10 from baseline at any time) were entered; relapsers were excluded
· Patients with a history of decompensated liver cirrhosis were excluded
· Plasma HCV RNA was measured by Roche COBAS TaqMan assay, with a lower limit of quantification (LLOQ) of 25 IU/mL and a lower limit of detection of 10 IU/mL
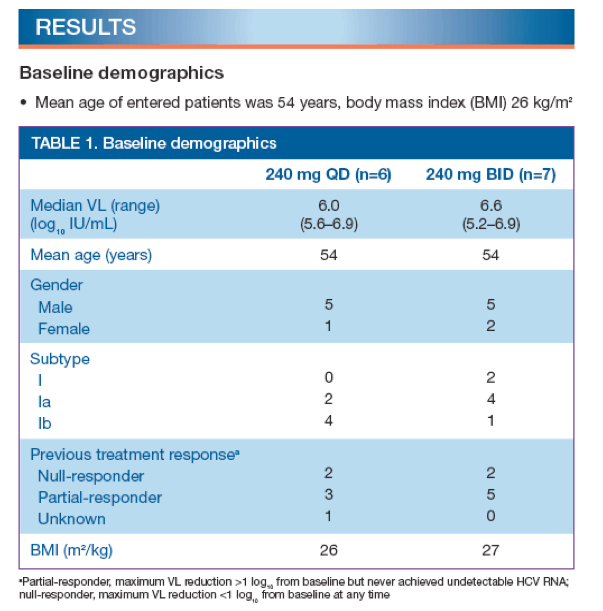
· Mean VL at baseline was 6.0 and 6.6 log10 (IU/mL) in the 240 mg QD and BID groups, respectively
--baseline demographics are shown in Table 1
Efficacy
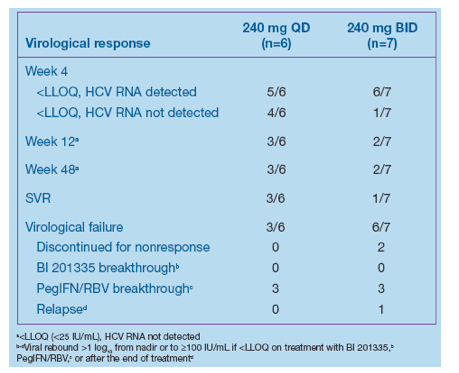
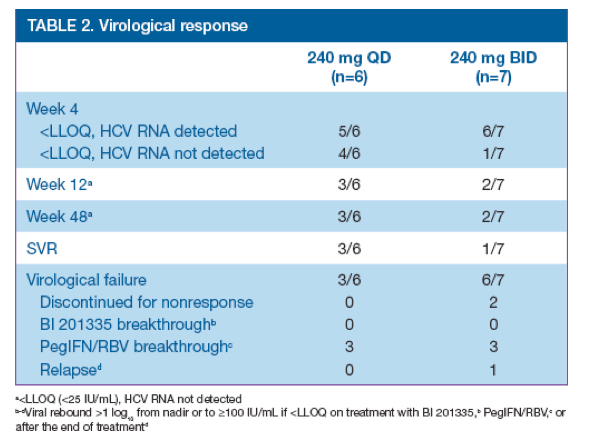
· Virological response is shown in Table 2
· No breakthroughs were observed during 28 days' treatment with BI 201335 plus PegIFN/RBV; most virological failures occurred as breakthroughs on PegIFN/RBV
· SVR rates per-protocol were 3/6 and 1/7 patients in the 240 mg QD and BID groups, respectively
Safety and tolerability
· Safety and tolerability of 4 weeks' triple therapy was good in both dose groups and similar to noncirrhotic patients.1,2 Adverse events (AEs) were mostly mild-to-moderate and typical of PegIFN/RBV
-- details have been presented previously3
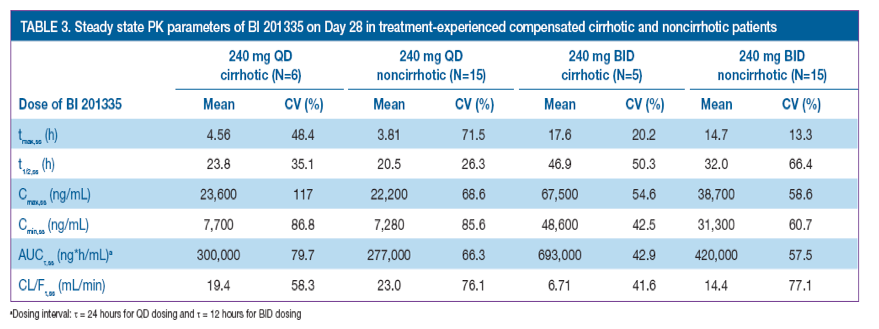
PK
· Steady state PK parameters are shown in Table 3
· In the 240 mg QD group, mean tmax,ss and t1/2,ss were similar between patients with compensated cirrhosis and without cirrhosis4
· In the 240 mg BID group, values of tmax,ss were also similar between patients with and without compensated cirrhosis; however, t1/2,ss was approximately 14 hours longer for patients with compensated cirrhosis
· Mean CL/Fτ,ss values were similar for patients with and without compensated cirrhosis, receiving 240 mg QD BI 201335
· In contrast, in the BID group, the value of CL/Fτ,ss for patients with compensated cirrhosis was approximately half that of patients without cirrhosis, indicating substantial impairment in oral clearance
References
1. Berg T, et al. Virological response and safety of 4 weeks' treatment with the protease inhibitor BI 201335 combined with 48 weeks of peginterferon alfa 2a and ribavirin for treatment of HCV GT-1 patients who failed peginterferon/ribavirin. Hepatology 2010;52(Suppl. 1). Abstract 804.
2. Manns MP, et al. Potency, safety and pharmacokinetics of the NS3/4A protease inhibitor BI201335 in patients with chronic HCV genotype-1 infection. J Hepatol 2010 Nov 11 [Epub ahead of print].
3. Pol S, et al. Virological response and safety of BI 201335 protease inhibitor, peginterferon alfa 2a and ribavirin treatment of HCV genotype-1 patients with compensated liver cirrhosis and non-response to previous peginterferon/ribavirin. Hepatology 2009;50(Suppl. 4). Abstract LB16.
4. Yong C-L, et al. BI 201335 pharmacokinetics and early effect on viral load in HCV genotype-1 patients. J Hepatol 2011;54(Suppl. 1). Abstract 1249.
5. Sulkowski MS, et al. SILEN-C2: sustained virologic response (SVR) and safety of BI 201335 combined with peginterferon alfa-2a and ribavirin (P/R) in chronic HCV genotype-1 patients with non-response to P/R. J Hepatol 2011;54(Suppl. 1). Abstract 66.
|
| |
|
 |
 |
|
|