 |
 |
 |
| |
Sustained Virologic Response and Boceprevir Resistance-Associated Variants Observed in Patients Infected With HCV
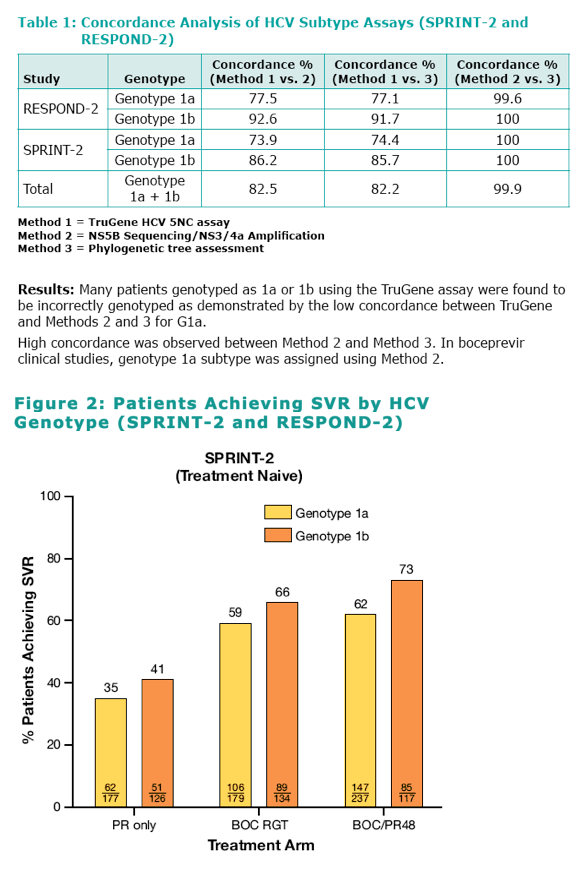
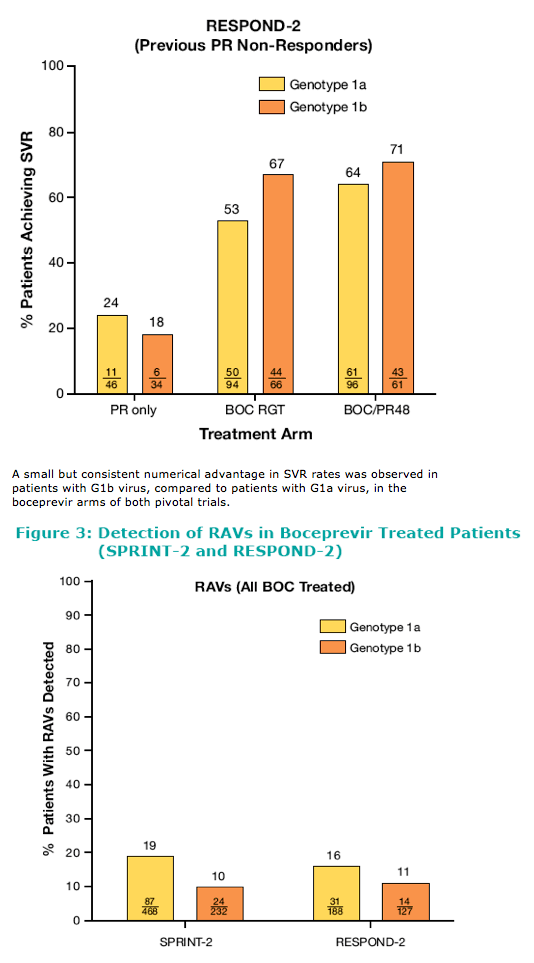
Genotype 1a/1b When Treated With Boceprevir Plus Peginterferon Alfa-2b/Ribavirin: SVR rates among patients with G1b virus were consistently higher compared with G1a patients in both SPRINT-2 and RESPOND-2
|
| |
| |
Reported by Jules Levin EASL 2011 March 31-Apr 2 Berlin Germany
C.A. Brass1*, R.J.O. Barnard1, J.A. Howe1, R.A. Ogert1, R. Ralston2, N. Boparai1, M. Burroughs1,
V. Sniukiene1, P. Mendez1, and J. Albrecht1
1Merck Sharp & Dohme Corp., Whitehouse Station, NJ, USA
2Laboratory of Virology and Infectious Disease, The Rockefeller University, New York, NY, USA *clifford.brass@merck.com
G1a is predominant in Northern Europe and North America
G1b is predominant in Southern and Eastern Europe and Japan
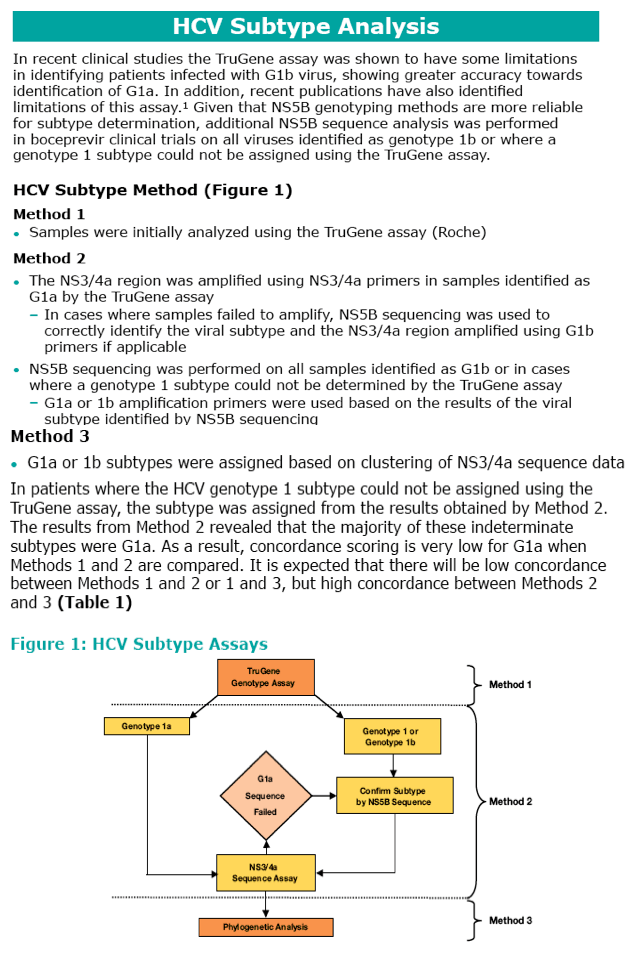
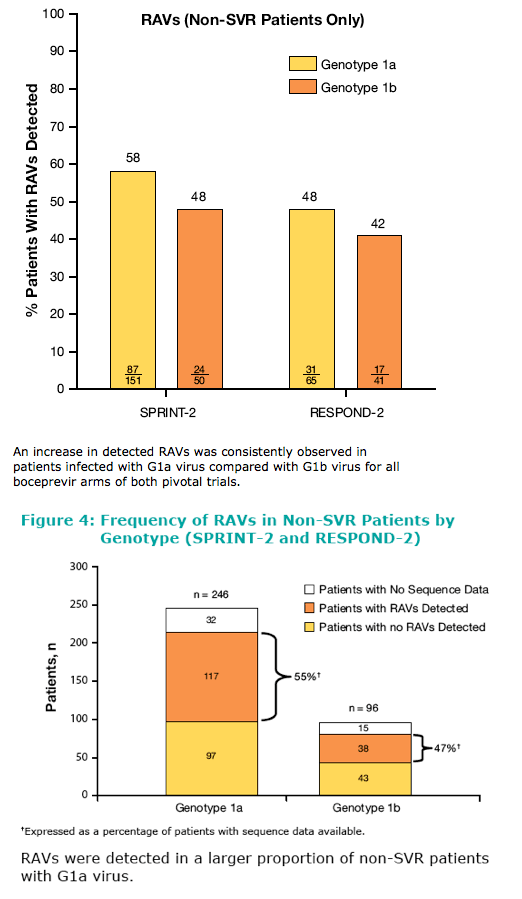
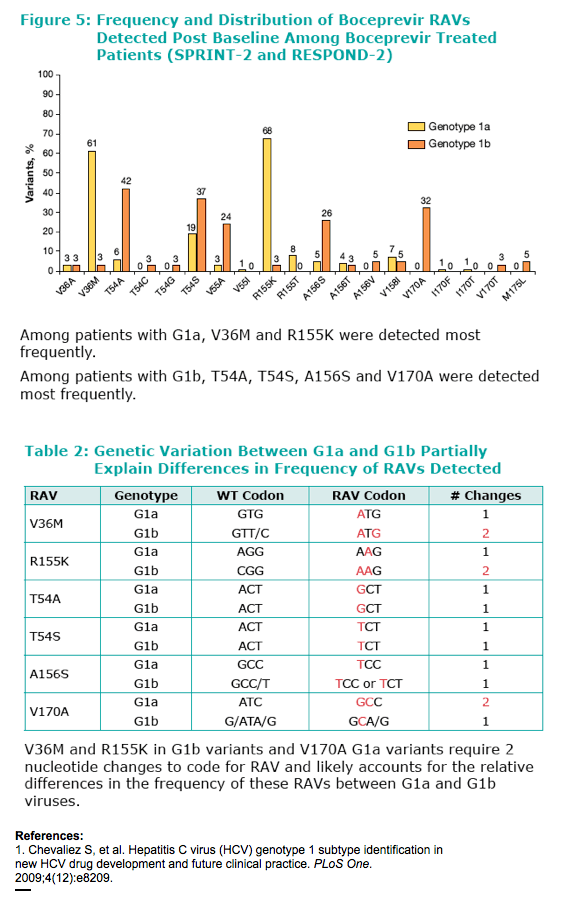
Many patients genotyped as 1a or 1b using the TruGene assay were found to be incorrectly genotyped as demonstrated by the low concordance between TruGene and Methods 2 and 3 for G1a. High concordance was observed between Method 2 and Method 3. In boceprevir clinical studies, genotype 1a subtype was assigned using Method 2. Viral genotype was determined at screening using the TruGene assay. Subsequently, genotyping was determined by a combination of NS5b sequencing and/or inferred from positive amplification using subtype-specific primers (Virco BVM, Belgium). Population sequencing of the NS3 region was performed by VircoAn increase in detected RAVs (mutations, Resistance-Associated amino acid Variants) was consistently observed in patients infected with G1a virus compared with G1b virus for all boceprevir arms of both pivotal trials. RAVs were detected in a larger proportion of non-SVR patients with G1a virus.

INTRODUCTION
To compare the rate of sustained virologic response (SVR) of boceprevir (BOC) dosed in combination with peginterferon alfa-2b (P) plus ribavirin (R) standard of care therapy in patients with genotype 1a (G1a) and genotype 1b (G1b) in the SPRINT-2 and RESPOND-2 clinical trials
To compare the frequency of Resistance-Associated amino acid Variants (RAVs) between G1a and G1b viruses among non-SVR patients enrolled in SPRINT-2 and RESPOND-2


|
| |
|
 |
 |
|
|