| |
Santaris Pharma A/S to Report New Clinical Data From HCV microRNA Miravirsen Phase 2a Study to Treat Hepatitis C in Late-Breaking Oral Presentation at the AASLD 2011 Annual Meeting - press release
|
| |
| |
HOERSHOLM, Denmark, and SAN DIEGO, Oct. 3, 2011 /PRNewswire via COMTEX/ -- Santaris Pharma A/S, a clinical-stage biopharmaceutical company focused on the discovery and development of microRNA and mRNA-targeted therapies, today announced the Company will report new clinical data results from the miravirsen Phase 2a proof-of-concept study to treat patients infected with the Hepatitis C virus (HCV) in a late-breaking oral presentation session at the American Association for the Study of Liver Diseases (AASLD) 2011 annual meeting.
Data from the Phase 2a trial showed that miravirsen, the first microRNA-targeted drug to enter clinical trials, provided continuous and prolonged anti-viral activity well beyond the end of active therapy in patients and was well tolerated in patients infected with HCV. These data demonstrate that miravirsen has the potential to be a once-weekly treatment for chronic HCV infection.
The abstract titled, "A Randomized, Double-blind, Placebo Controlled Safety and Anti-viral Proof of Concept Study of Miravirsen, an Oligonucleotide Targeting miR-122, In Treatment Naive Patients with Genotype 1 Chronic HCV Infection" will be presented in a late-breaking oral presentation on November 7 at 4 p.m., at The Liver Meeting® 2011, the 62nd annual meeting of the AASLD, taking place from November 4-8 in San Francisco, California. The abstract is published on the AASLD website at http://aasld2011.abstractcentral.com/login .
The randomized, double-blind, placebo-controlled, ascending multiple-dose Phase 2a study assessed the safety and tolerability of miravirsen in treatment-naive patients with chronic HCV genotype 1 infection. Patients with chronic HCV genotype 1 infection were enrolled sequentially to one of three cohorts (9 active:3 placebo per cohort). Miravirsen was given as weekly subcutaneous injections, over 29 days.
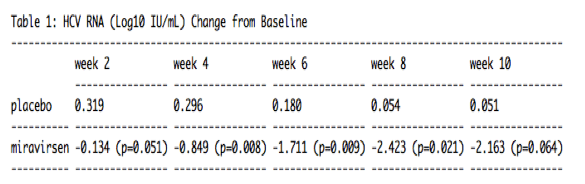
Key data that will be presented demonstrate that miravirsen was associated with dose-dependent, prolonged reductions in HCV RNA that continued to fall after the completion of miravirsen therapy (see table 1). In Cohort 2, the mean of the maximum change from baseline in HCV RNA (log10 IU/mL) over the first 10 weeks was -2.710 vs -0.152 (p =0.020) in the miravirsen and placebo groups, respectively. In Cohort 2, five subjects had a greater than or equal to a 2 log10 decrease from baseline to week 10 and one patient became HCV RNA undetectable 10 weeks after the last dose of miravirsen (week 14) without the addition of standard-of-care (SOC). Cohort 3 is ongoing.
Developed using Santaris Pharma A/S proprietary Locked Nucleic Acid (LNA) Drug Platform, miravirsen is an inhibitor of miR-122, a liver specific microRNA that the Hepatitis C virus requires for replication. Miravirsen is designed to recognize and sequester miR-122, making it unavailable to the Hepatitis C virus. As a result, the replication of the virus is effectively inhibited and the level of Hepatitis C virus is profoundly reduced.
Miravirsen (SPC3649): a novel treatment for Hepatitis C Virus (HCV)
miravirsen is a specific inhibitor of miR-122, a liver specific microRNA that the Hepatitis C virus requires for replication. Thus, unlike many other Hepatitis C therapies that directly target the virus, miravirsen works by removing a "helper" molecule (miR-122) that the virus needs in order to make new copies. As a result, miravirsen is expected to provide a very high barrier to the generation of viral resistance - a significant problem with direct acting drugs due to the very high mutation rate of the virus.
In preclinical studies, miravirsen produced a potent, dose-dependent and long lasting inhibition of miR-122 in mice, cynomologus monkeys and green African monkeys as indicated by decreases in cholesterol levels, a biomarker of miR-122 activity. In chimpanzees chronically infected with Hepatitis C, miravirsen caused a significant (350 fold) and long-lived reduction in viral load.
Indeed, as expected from the mode-of-action of miravirsen, there was no evidence of viral resistance during the extended study. The preclinical data also showed changes in the expression of key genes, signaling that the drug may help patients who do not respond to interferon treatment to become responsive. In all animal studies, including IND enabling toxicology studies, the drug has been well tolerated, suggesting that miravirsen could become an important new therapeutic in the fight against Hepatitis C.
Miravirsen is the first microRNA-targeted drug to enter human clinical trials. Data from a Phase 1 single-ascending dose safety study in healthy volunteers show that miravirsen is well tolerated, has an attractive pharmacokinetic profile and clear dose-dependent pharmacology. Currently, mirvirsen is being studied in a multiple ascending dose study in healthy volunteers. A Phase 2 study in patients infected with Hepatitis C began in September 2010.
How does miravirsen work?
In healthy individuals, miR-122 is an abundant, liver specific microRNA that regulates a host of messenger RNAs in the liver, many of which encode proteins involved in lipid and cholesterol metabolism.
When normal liver cells are infected by the Hepatitis C virus, the virus co-opts miR-122 to two binding sites located close to the 5'end of its linear RNA genome. This binding enables the virus to replicate and create new copies of itself, which can further spread the infection throughout the liver. Miravirsen is designed to specifically recognize and sequester miR-122 thus making it unavailable to the Hepatitis C virus. As a result, the replication of the virus is effectively inhibited.
About Hepatitis C
Hepatitis C is a viral disease caused by the Hepatitis C virus that leads to swelling (inflammation) of the liver. The World Health Organization estimates about 3% of the world's population has been infected with HCV and that some 170 million are chronic carriers at risk of developing liver cirrhosis and/or liver cancer(1). Approximately 3-4 million Americans are chronically infected with an estimated 40,000 new infections per year(2). In Europe, there are about 4 million carriers(1). Treatment with the current standard of care, pegylated interferon in combination with ribavirin, is effective in only about 50% of those treated(2). Patients that are not effectively treated have an increased risk for the progression of liver disease. By 2029, total annual medical costs in the United States for people with Hepatitis C are expected to more than double, from $30 billion in 2009 to approximately $85 billion(3).
About microRNAs
MicroRNAs have emerged as an important class of small RNAs encoded in the genome. They act to control the expression of sets of genes and entire pathways and are thus thought of as master regulators of gene expression. Recent studies have demonstrated that microRNAs are associated with many disease processes. Because they are single molecular entities that dictate the expression of fundamental regulatory pathways, microRNAs represent potential drug targets for controlling many biologic and disease processes.
About Locked Nucleic Acid (LNA) Drug Platform
The LNA Drug Platform and Drug Discovery Engine developed by Santaris Pharma A/S combines the Company's proprietary LNA chemistry with its highly specialized and targeted drug development capabilities to rapidly deliver LNA-based drug candidates against RNA targets, both mRNA and microRNA, for a range of diseases including cardiometabolic disorders, infectious and inflammatory diseases, cancer and rare genetic disorders. LNA-based drugs are a promising new class of therapeutics that are enabling scientists to develop drug candidates to work through previously inaccessible clinical pathways. The LNA Drug Platform overcomes the limitations of earlier antisense and siRNA technologies to deliver potent single-stranded LNA-based drug candidates across a multitude of disease states. The unique combination of small size and very high affinity allows this new class of drugs candidates to potently and specifically inhibit RNA targets in many different tissues without the need for complex delivery vehicles. The most important features of LNA-based drugs include excellent specificity providing optimal targeting; increased affinity to targets providing improved potency; and favorable pharmacokinetic and tissue-penetrating properties that allow systemic delivery of these drugs without complex and potentially troublesome delivery vehicles.
About Santaris Pharma A/S
Santaris Pharma A/S is a privately held clinical-stage biopharmaceutical company focused on the discovery and development of RNA-targeted therapies. The Locked Nucleic Acid (LNA) Drug Platform and Drug Discovery Engine developed by Santaris Pharma A/S combine the Company's proprietary LNA chemistry with its highly specialized and targeted drug development capabilities to rapidly deliver potent single-stranded LNA-based drug candidates across a multitude of disease states. The Company's research and development activities focus on infectious diseases and cardiometabolic disorders, while partnerships with major pharmaceutical companies include a range of therapeutic areas including cancer, cardiovascular disease, infectious and inflammatory diseases, and rare genetic disorders. The Company has strategic partnerships with miRagen Therapeutics, Shire plc, Pfizer, GlaxoSmithKline, and Enzon Pharmaceuticals. As part of its broad patent estate, the Company holds exclusive worldwide rights to all therapeutic uses of LNA. Santaris Pharma A/S, founded in 2003, is headquartered in Denmark with operations in the United States. Please visit www.santaris.com for more information.
(1) World Health Organization - http://www.who.int/csr/disease/hepatitis/Hepc.pdf
(2) American Association for the Study of Liver Diseases - http://www.aasld.org/patients/Pages/LiverFastFactsHepC.aspx
(3) Institute of Medicine of the National Academies. Hepatitis and liver cancer: a national strategy for prevention and control of hepatitis B and C. Colvin HM and Mitchell AE, ed. Available at: http://www.iom.edu/Reports/2010/Hepatitis-and-Liver-Cancer-A-National-Strategy-for-Prevention-and-Control-of-Hepatitis-B-and-C.aspx .
SOURCE Santaris Pharma A/S
|
|
| |
| |
|
|
|