| |
Hep C Nukes/Drug Development
|
| |
| |
Hep C Drug Panic! Pharmasset Stumble Ripples Through Sector
"All HCV drugs must pass the 12week safety bar. The treatment duration for oral HCV compounds is likely to be 12 weeks or 24 weeks. Therefore, all new drug candidates must pass the 12-week safety bar. Currently, the only nukes that have met this important criterion are PSI-7977 and mericitabine from Roche"
WSJ: In contrast, shares of Vertex Pharmaceuticals Inc. (VRTX) surged 8.2% Friday to $34 because it markets a new hepatitis C drug and is developing additional drugs, and the Pharmasset setback may remove potential competition. "Any setback for PSI-938...would be perceived as positive for Vertex," Barclays Capital wrote in a note to clients......"During routine safety monitoring, the company detected laboratory abnormalities associated with liver function" in people receiving a 300-milligram dose of PSI-938 once daily, Pharmasset said. Pharmasset said there were no abnormalities observed in patients who received PSI-7977 in the same clinical trial.
(fool.com) Safety (Still) Trumps Acquisitions in Hep C: The euphoria over Gilead Sciences' acquisition of Pharmasset and Roche's purchase of Anadys Pharmaceuticals seems to have caused memory loss in investors. But they got a reminder of reality today, when Pharmasset announced it was discontinuing all treatment arms in one of its phase 2b trials that contain the drug PSI-938. The drug candidate caused laboratory abnormalities in tests associated with liver function.This is nothing new. Pfizer could be further along in the hepatitis C race if ViroPharma and Wyeth's HCV-796 hadn't died back in 2008, before Pfizer bought Wyeth. HCV-796 suffered from the same potential liver issues as PSI-938. Ditto for InterMune and Roche's danoprevir at high doses. Elevated liver enzyme levels are an issue with all drugs, but they're especially problematic for hepatitis C patients, since the virus attacks the liver. And the list goes on. Boehringer Ingelheim had problems with one of its protease inhibitors. And trials involving Idenix Pharmaceuticals' IDX320 were put on clinical hold by the FDA before the company eventually scrapped the compound. Does the failure of PSI-938 and all the aforementioned compounds mean that Inhibitex and Idenix -- both down big today -- will also run into safety issues? Absolutely not. Liver toxicity is still a great mystery to drugmakers. If scientists understood what caused liver issues, they could design drugs that didn't cause it. And besides, Merck's Victrelis and Vertex Pharmaceuticals' Incivek both made it through without major safety issues. But the failure is a reminder that long-term safety data is still very important to get a drug through the clinical development marathon.
thestreet.com
By Adam Feuerstein 12/16/11 - 02:11 PM EST
PRINCETON, NJ (TheStreet) -- The discovery of liver toxicity in a Pharmasset hepatitis C drug Friday has investors panicked that similar problems could crop up in competing drugs from Inhibitex and Idenix Pharmaceuticals.
Pharmasset first spooked investors Friday morning with the announcement that hepatitis C patients were being pulled off treatment with PSI-938 because of laboratory measurements suggesting the experimental drug was negatively affecting liver function.
Importantly, PSI-938 is not the keystone jewel in Pharmasset's hepatitis C pipeline for which Gilead Sciences is paying $11 billion to acquire. Gilead's main interest is in Pharmasset's drug PSI-7977, which remains untouched by Friday's announcement.
Pharmasset said the safety problem with PSI-938 does not trigger an escape clause written into the Gilead purchase agreement that allows Gilead to walk away if a serious adverse event or safety issue is discovered in a key Pharmasset drug.
Regardless, Pharmasset shares were down 2% to $125.28 Friday, even as the company reaffirmed its belief that the Gilead deal will close in the first quarter of next year. Gilead is acquiring Pharmasset for $137 per share.
The real investor panic took place in Inhibitex, which is developing a hepatitis C drug known as INX-189 that shares similarities in chemical structure with Pharmasset's now-tainted PSI-938. With Pharmasset bought for $11 billion, investors have been betting that Inhibitex could be the next hepatitis C drug company to get gobbled up.
But if PSI-938 was causing liver damage, then perhaps treatment with INX-189 runs the same risk -- not a pleasant thought for a company trading at a high takeover premium already.
No evidence of INX-189 induced liver damage has been reported yet, but just that possibility hit Inhibitex hard with shares plunging almost 50% in Friday's pre-market trading. The stock recovered somewhat when regular trading opened but Inhibitex is still down 21% to $10.29.
Analysts rushed to defend Inhibitex, pulling out their medicinal chemistry textbooks to demonstrate that while INX-189 was similar in some respects to Pharmasset's PSI-938, the two drugs also have structural differences.
For hepatitis C investors, Friday was clearly a day to regret sleeping through your college chemistry classes. Here's what JMP Securities analyst Liisa Bayko had to say to clients about INX-189:
"In our view, it is important to note that PSI-938 has a different active species (2-fluoro guanosine triphosphate) than INX-189 (2-methyl guanosine triphosphate) and that the compounds are not similar outside of the guanosine base - looking at the compounds as a whole, we believe INX-189 is more structurally similar to PSI-7977 than PSI-938. We also note that there are guanosine drugs marketed such as acyclovir, and therefore we do not believe that guanosine is the reason for the PSI-938 toxicity."
Deutstch Bank analyst Robyn Karnauskas held a call for clients Friday morning with an Inhibitex executive and a medicinal chemist who has worked on INX-189 and is familiar with other hepatitis C drugs.
On the call, Inhibitex re-affirmed the absence of liver-related toxicity in clinical studies of INX-189 to date at doses up to 100 mg. In one ongoing study, four patients treated with 100 mg of INX-189 have gone through 10 weeks of treatments, with two of those patients completing treatment.
Not all are convinced that INX-189 is in the clear.
"If you compare the activated forms of each (which happens pretty fast) the 2 structures are much much closer: specifically, varying only in that 938 has a F at C-2' in and 189 has a OH (F is a chemical isostere of OH)...The 938 result does not necessarily mean the same will hold for 189, but as I see it, it increases odds," said one buy-side analyst, referring to a side-by-side diagram of the chemical structures (just below) of INX-198 and PSI-939. This analyst had a short position in Inhibitex and was using Friday's slide to partially cover.
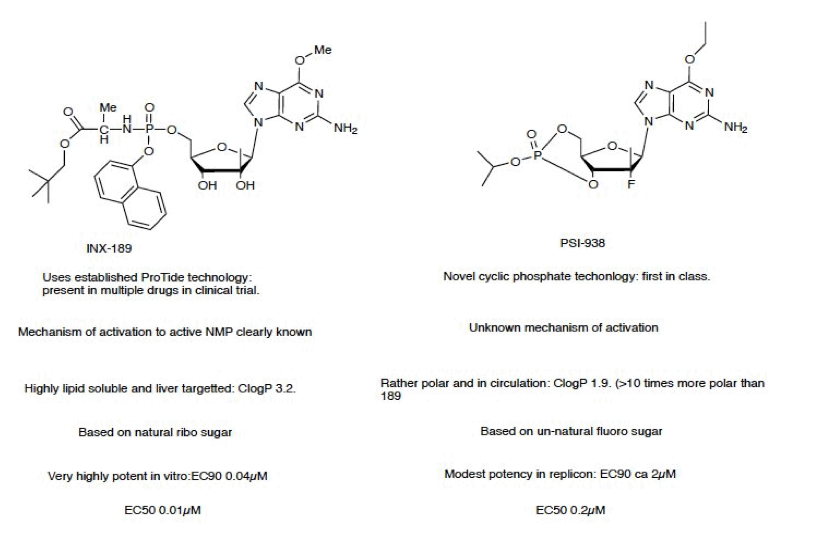
Idenix Pharmaceuticals is developing hepatitis C drugs in the same class as Inhibitex and Pharmasset, so naturally, shares were down 10% to $7 Friday.
Perhaps, the only people chuckling Friday were executives at Vertex Pharmaceuticals, which was supposed to be a hepatitis C also ran due to the looming entrant of drugs like those from Pharmasset and Inhibitex. Perhaps Vertex isn't out of the hepatitis C game quite yet. Vertex shares were up 5% to $33.20.
-----------------
Pharmasset's PSI938 Discontinued; Impact on Other Nukes in Hepatitis C?
Wm Blair & Co Analyst Equity Research Report
Y. Katherine Xu
"Currently, the only nukes that have met this important criterion are PSI-7977 and mericitabine from Roche (RHHBY $42.15). PSI-7977 has been dosed for 12 weeks or longer in over 400 patients resulting in no safety signals, and this, together with the high potency and the first-in-class status, led to Gilead's $11 billion offer. Mericitabine also passed the challenge and Roche recently announced the initiation of the Phase II DYNAMO study, which will evaluate mericitabine+Victrelis+P/R, in addition to the INFORM and MATTERHORN studies that evaluate the mericitabine+danoprevir combo with or without P or R. Today's announcement means that PSI-938 may not pass the bar. Next we need to see whether INX-189 of Inhibitex and IDX184 of Idenix pass the 12-week safety bar. If they accomplish this feat, there will be a significant valuation inflection for both drugs."
Friday morning, Pharmasset announced that it would amend the ongoing Phase IIb QUANTUM study by discontinuing all treatment arms containing PSI938; abnormal liver function tests were observed in hepatitis C virus (HCV) patients receiving 300 mg oncedaily dosing (QD) of PSI938. There are 235 patients who received PSI-938 monotherapy or PSI-7977+PSI-938 in the QUANTUM study, and the liver signal was ascribed to PSI-938. The company did not disclose when the onset of the liver signals occurred or details on the liver function tests (ALT, AST, or bilirubin), but we note that previously PSI-938 appeared safe in the 14-day NUCLEAR study, although patient numbers were small. Pharmasset commented that no laboratory abnormalities have been observed in patients receiving PSI-7977 plus ribavirin (R). Only three arms in QUANTUM will continue: PSI-7977+R for 12 weeks, PSI-7977+R for 24 weeks, and placebo. We illustrate the Phase IIb QUANTUM study in figure 1, on page 3.
No death sentence yet on PSI938. Pharmasset commented that as QUANTUM enrolled very quickly, the liver signal was detected in boluses of patients, which made it difficult to dissect details within a short time. The company made the decision to discontinue all PSI-938�containing arms within the last 24 hours to protect the PSI-7977+R arms; if more severe PSI-938 safety signals come up during the holidays, the FDA could shut down the whole QUANTUM study, which would impede the progress of the key candidate. Pharmasset intends to re-start studies on PSI-938 at lower doses to evaluate its safety as well its utility in the combination setting with PSI-7977, in particular, whether PSI-938 could eventually replace R.
PSI7977 stays intact; PSI7977+R up for testing in GT 1 patients with data out late first quarter 2012. To date, over 400 patients have been exposed to PSI-7977 for 12 weeks or longer, and no notable safety signals have been identified. The goal of the QUANTUM study is to test whether the PSI-7977+R regimen for 12 weeks could also achieve the same near-100% SVR in genotype 1 (GT 1) patients as it did in GT 2/3 patients, and if 12 weeks of PSI-7977+R treatment is not sufficient, whether 24 weeks will be. The SVR data will be available during late first quarter or early second quarter 2012, by our estimation. We believe this will be the decisive data for Gilead and Pharmasset to have a pan-genotypic, all-oral regimen in hand to take into full blown Phase III studies.
No impact on Gilead acquisition of Pharmasset; deal on target to close first quarter 2012. Both Pharmasset and Gilead (GILD $37.16; Outperform) have commented that today's announcement did not trigger the "key product event" clause within the agreement announced on November 21, and both parties' rights and obligations remain intact. Gilead stated that the value of the Pharmasset transaction is based on the potential future cash flow from PSI-7977 along with its own HCV portfolio.
All HCV drugs must pass the 12week safety bar. The treatment duration for oral HCV compounds is likely to be 12 weeks or 24 weeks. Therefore, all new drug candidates must pass the 12-week safety bar. Currently, the only nukes that have met this important criterion are PSI-7977 and mericitabine from Roche (RHHBY $42.15). PSI-7977 has been dosed for 12 weeks or longer in over 400 patients resulting in no safety signals, and this, together with the high potency and the first-in-class status, led to Gilead's $11 billion offer. Mericitabine also passed the challenge and Roche recently announced the initiation of the Phase II DYNAMO study, which will evaluate mericitabine+Victrelis+P/R, in addition to the INFORM and MATTERHORN studies that evaluate the mericitabine+danoprevir combo with or without P or R. Today's announcement means that PSI-938
may not pass the bar. Next we need to see whether INX-189 of Inhibitex and IDX184 of Idenix pass the 12-week safety bar. If they accomplish this feat, there will be a significant valuation inflection for both drugs.
What is the read through to Inhibitex and Idenix? PSI-938, INX-189, and IDX184 share the same basic nucleotide, guanosine, but have different modifications on the sugar moiety and very different pro-drug designs. We do not believe the liver signal observed with PSI-938 would necessarily manifest in INX-189 or IDX184 studies; this is likely not a guanosine nucleotide class issue. We believe that the total package of the molecule dictates its efficacy and safety profile; eventually all these drugs need to be studied in humans, and the ones that show adequate 12-week safety in humans will prevail, despite what the in vitro or preclinical studies might suggest.
For example, PSI-938 in vitro was very clean, but liver toxicity came up in humans. The same happened to the Idenix protease inhibitor IDX320. Mericitabine, on the other hand, demonstrated kidney toxicity in monkeys, but was able to pass the 12-week safety bar in humans.
Our conversation with Idenix management suggests to us that to date, a few of the first cohort of 30 patients on IDX184+P/R have reached the 12-week mark and so far no safety signals were observed that are atypical of what is routinely observed with P/R alone. Patients with abnormal liver enzymes have normalized, and patients with normal liver enzymes at baseline have stayed stable.
Our conversation with Inhibitex management suggests to us that to date, a few of the patients on INX-189+P/R have reached the 12-week mark and a few more have passed the 10-week mark. So far, no treatment interruptions occurred and liver enzymes are coming down as well.
We realize that these are very small numbers of patients, and some rare adverse events might start to manifest only when the drug is studied in hundreds of patients. However, liver enzyme levels, hematological parameters, and GI side effects, which are the "usual" potential side effects observed with nukes, usually show up during the first 12 weeks and occur at a relatively high frequency. The fact that neither Idenix nor Inhibitex observed strong safety signals to date is encouraging, in our opinion.
|
|
| |
| |
|
|
|