 |
 |
 |
| |
Long-term safety of once-daily darunavir/ritonavir versus lopinavir/ritonavir in HIV-1-infected treatment-naïve patients in ARTEMIS
|
| |
| |
Reported by Jules Levin
6th IAS Rome July 17-20 2011
Nathan Clumeck,1 Ben J Barnett,2 Prudence Ive,3 Annemie Hendrickx,4 Magda Opsomer,4 Tom Van de Casteele,4 Frank Tomaka5
1Saint-Pierre University Hospital, Brussels, Belgium; 2Harris County Hospital District, Houston, Texas, USA; 3Helen Joseph Hospital, University of the Witwatersrand, Johannesburg, South Africa; 4Tibotec BVBA, Beerse, Belgium; 6Tibotec Inc., Titusville, NJ, USA
Conclusions
· At Week 192, no new clinically relevant safety concerns were identified compared to the Week 48 and 96 findings from ARTEMIS.1,8
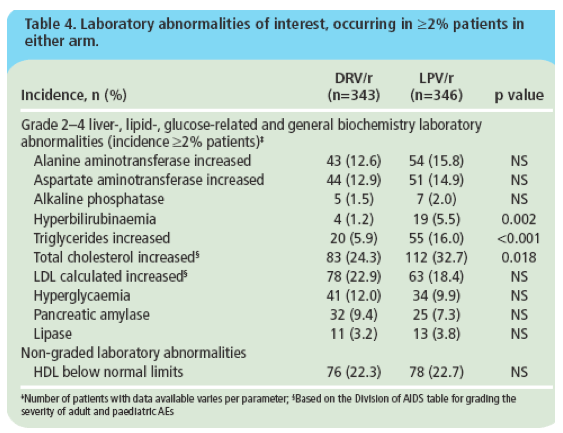
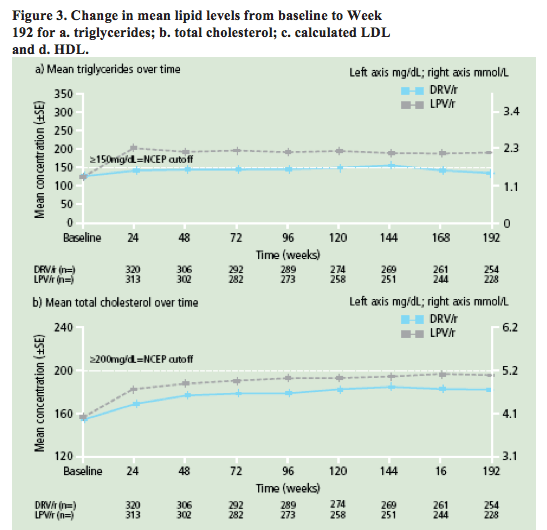
· DRV/r was associated with significantly fewer discontinuations due to AEs, a lower incidence of grade 2-4 treatment-related diarrhoea, and fewer grade 2-4 increases in triglycerides and total cholesterol than LPV/r.
· This final 192-week analysis demonstrates the long-term favourable safety and tolerability profile of once-daily DRV/r 800/100mg in treatment-naïve HIV-1-infected adults.

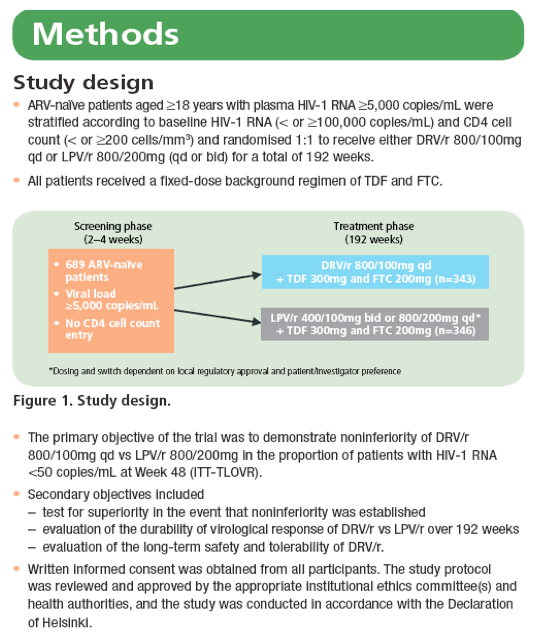

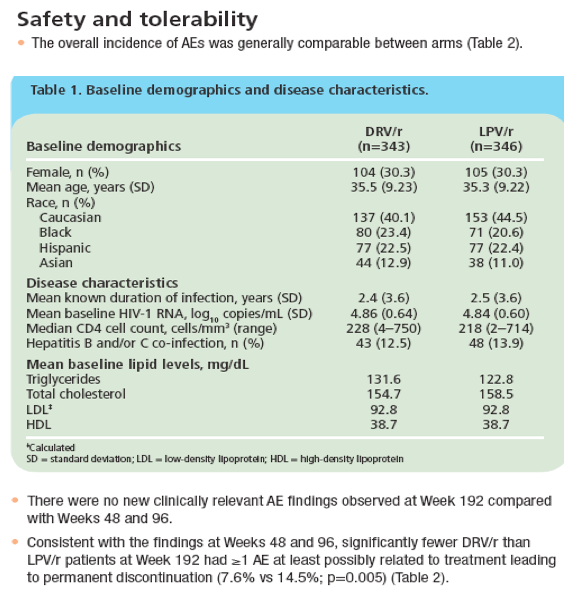
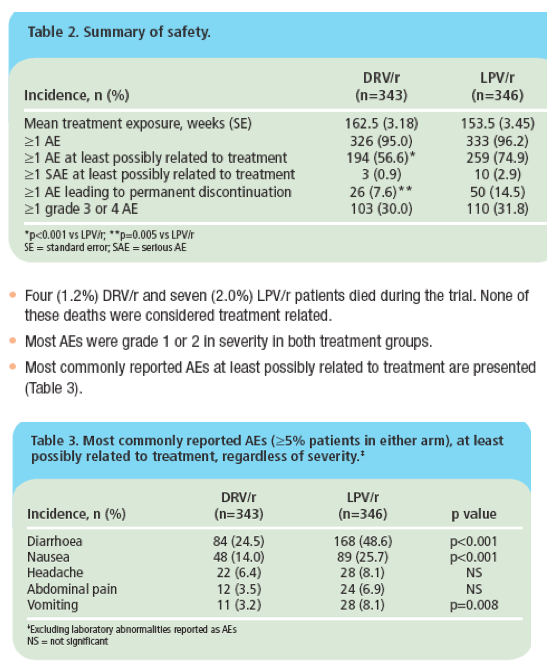
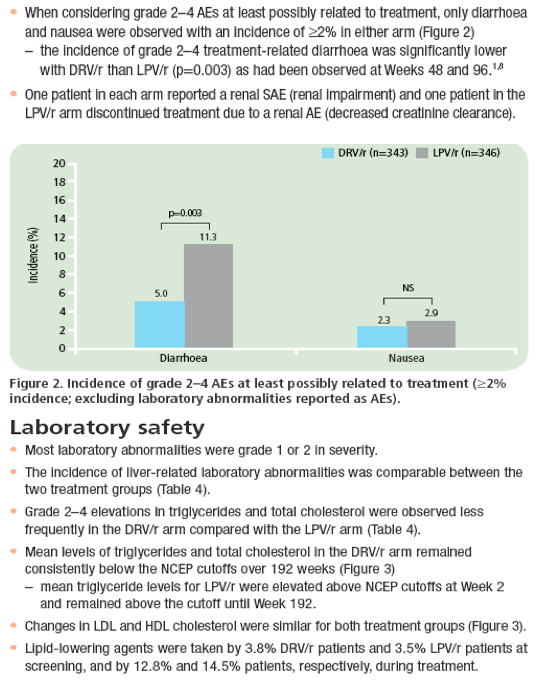
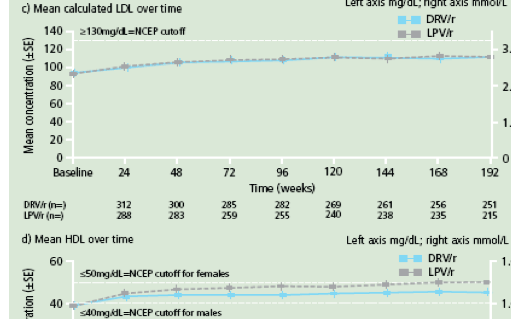
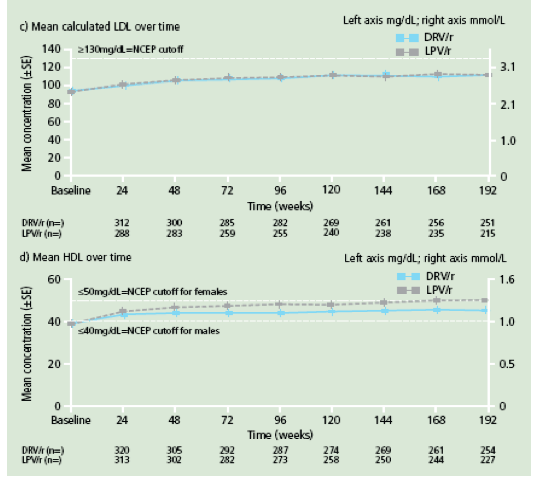
Figure 3. Change in mean lipid levels from baseline to Week 192 for a. triglycerides; b. total cholesterol; c. calculated LDL and d. HDL.
References
1. Ortiz R, et al. AIDS 2008;22:1389-97.
2. Clotet B, et al. Lancet 2007;369:1169-78.
3. Madruga JV, et al. Lancet 2007;370:49-58.
4. Cahn P, et al. AIDS 2011;25:929-39.
5. Tibotec Inc. PREZISTA® (darunavir) Full Prescribing Information. Revised December 2010 [accessed 26 May 2011]. Available at:
http://www.prezista.com/healthcare/full-prescribing-information.
6. PREZISTA® (darunavir). EPARs for authorised medicinal products for human use. 22 March 2011 [26 accessed May 2011]. Available at:
http://www.emea.europa.eu/humandocs/Humans/EPAR/prezista/prezista.htm.
7. Orkin C, et al. J Int AIDS Soc 2010;13(Suppl. 4):P3. Available at:
http://www.jiasociety.org/content/13/S4/P3/abstract.
8. Mills AM, et al. AIDS 2009;23:1679-88.
9. DAIDS Table for Grading the Severity of Adult and Pediatric Adverse Events. Version 1.0. December 2004.
10. National Cholesterol Education Program (NCEP) Expert Panel. Circulation 2002;106:3143-421.
|
| |
|
 |
 |
|
|