 |
 |
 |
| |
Rilpivirine (TMC278) tolerability over the first 12 weeks of treatmentin the Phase III ECHO and THRIVE studies
|
| |
| |
Reported by Jules Levin
51st Interscience Conference on Antimicrobial Agents and Chemotherapy, Chicago, IL, USA, September 17-20 2011
Bruce Rashbaum,1 Pierre-Marie Girard,2 Anita Rachlis,3 Diana Pedral-Sampaio,4 Aimee Wilkin,5 Simon Vanveggel,6 Katia Boven7
1Capital Medical Associates, Washington, DC, USA; 2Hôpital Saint Antoine, Paris, France; 3Sunnybrook Health Sciences Centre, University of Toronto, Toronto, Canada; 4Federal University of Bahia, Brazil;5Wake Forest University Health Sciences, Winston-Salem, NC, USA; 6Tibotec BVBA, Beerse, Belgium; 7Tibotec Inc. Titusville, NJ, USA





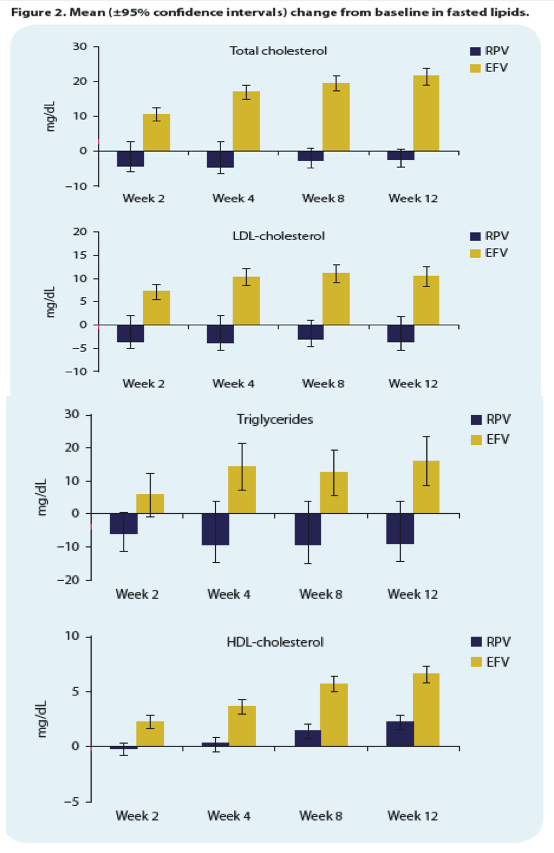
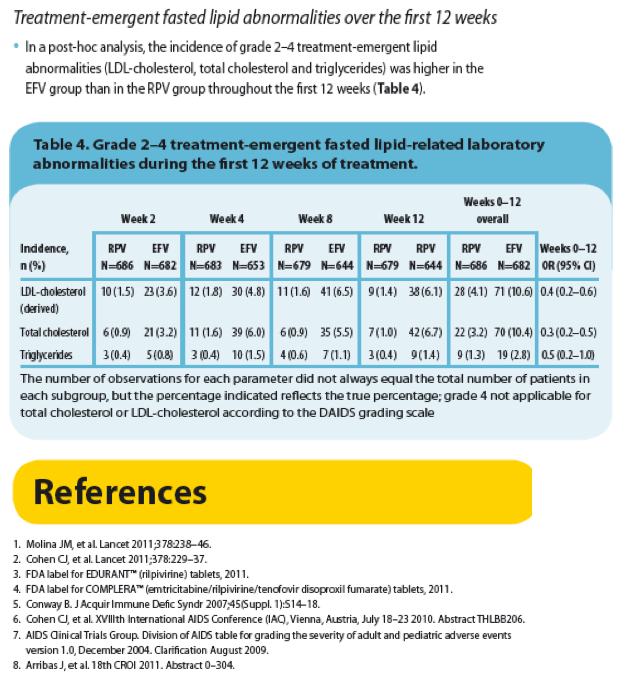
|
| |
|
 |
 |
|
|