 |
 |
 |
| |
51st Interscience Conference on Antimicrobial
Agents and Chemotherapy (ICAAC)
September 17-20, 2011
Chicago, IL
|
| |
| |
Eric S. Daar, M.D.
Chief, Division of HIV Medicine
Harbor-UCLA Medical Center
Professor of Medicine
David Geffen School of Medicine at UCLA
The 51st ICAAC included four days of presentations of data related to the pathogenesis and treatment of a wide range of infectious diseases. Despite the focus being broad there were several important presentations and posters related to HIV pathogenesis, transmission and treatment. Moreover, this meeting continued the increasing trend of expanding the data related to new therapeutics for the management of HCV infection. While the latter continues to be primarily focused on those with mono-infection, the HIV-treating world looks on closely knowing that these advancements will likely apply to upwards of a third of HIV-infected patients. This overview will touch on select presentations of interest related to the transmission and treatment of HIV and treatment of HCV infection.
Transmission
One of the most entertaining presentations of the meeting utilized a gay-specific smart phone social-networking application, GRINDR to identify individuals for a survey of risk-taking behavior (1). This smart phone "app"allow individuals to interact as well as to do so based upon geography since the GPS function enables identifying those in proximity to the user. The study recruited men who have sex with men between 18 and 29 years of age by enticing them, partly with curiosity, as well as with a $25 gift card. Despite reaching out to over 4800 men over a 5 month period of time only 375 agreed to participate, and many of these were actually identified in real time when study personnel were at sites where eligible individuals congregate, and not by the outreach on GRINDR. The ultimate study group was a median of 25 years old, 43% white, 34% Latino and 6% African American. Sixteen of the men admitted to being HIV-infected with the majority of the study group having been tested for HIV in the last year (83%). Of those surveyed 43% admitted to having unprotected receptive anal intercourse, and 47% unprotected insertive anal intercourse in the preceding month with nearly half admitting to having sex while under the influence of alcohol or drugs. Fifty six percent of participants said that they had actually found a sex partner in the past month using GRINDR, with the majority not inquiring about partner HIV status. While this study demonstrated the utility of using a social networking smart phone app to identify at-risk individuals, it also provided insight into how electronic media and networking sites are actively being utilized for identification of potential sex partners.
Another presentation reported on the results of an anonymous survey of people attending a Chicago sexually transmitted infection clinic in 2010 (2). The survey assessed HIV risk-taking behaviors, knowledge about HIV transmission and interest in pre-exposure prophylaxis (PrEP). Notably, this survey was completed prior the availability of the iPrEX study (3). Of the nearly 500 people who completed the survey, most (88%) were heterosexual, with a majority being male (63%) and African American (70%). The investigators reviewed the responses and determined that 359 were at "high risk" for HIV with 35% of these individuals being heterosexual women. Interestingly, despite the fact that most correctly answered questions regarding how HIV is transmitted (80%) there was a substantial disconnect between the investigator-defined HIV risk and those of the individuals. In fact, more than 80% of both men and women felt to be at high risk for HIV infection by the study team perceived themselves to be at low risk. Consistent with these beliefs, the overwhelming majority of women (96%) and 83% of men reported inconsistent use of condoms during intercourse. Finally, each individual was questioned about whether they would use PrEP, and if so how would they be willing to have it administered. While more than 80% of men and women stated that they would take PrEP if available, many did not feel that they would be willing to take it daily. This data provides insight into what may be needed to curb the spread of HIV. In particularly, despite the majority of surveyed individuals knowing about HIV transmission, more than 80% of those assessed as being at high risk, perceived themselves to be at low risk and this was accompanied by inconsistent condom use. Thus, education about transmission is not sufficient, but needs to include enhanced ability to communicate the actual risk for a given individual in order for them to better protect themselves in the future.
Raltegravir in treatment naïve and -experienced patients
Long-term follow-up data was presented from the pivotal STARTMRK trial of treatment-naïve patients randomized to tenofovir DF/emtricitabine (TDF/FTC) with either RAL or efavirenz (EFV) (4). The investigators further assessed the relationship between response and baseline characteristics, such as age, gender, race, viral load, CD4 and hepatitis co-infection. Of the 281 randomized to RAL and 282 to EFV, 227 and 211 completed 156 weeks of follow-up, respectively. Overall virologic responses for RAL and EFV by TLOVR non-complete=failure was 75.4 and 68.1% (difference = 7.3; 95% CI -0.2, 14.7) and by observed failure it was 89.5 and 84.6% (difference = 4.9; 95% CI -1.3, 11.1), respectively. Further analysis of treatment effect across pre-specified baseline subgroups using observed failure only (meaning discontinuations of randomized drug not counted as failures) did differ by baseline viral load. The overall response rates for RAL and EFV were very similar in those with viral loads >100,000 copies/mL (86 and 85%, respectively) but was different in the ≤100,000 copies/mL strata, 94 vs. 84%, respectively. The explanation for the treatment effect seen in the low viral load strata at baseline is not completely understood at this time. Notably, despite these differences, response rates were very high for both groups regardless of baseline factors and the response to RAL appears to be quite durable.
Another study better characterized the efficacy, safety, tolerability and pharmacokinetics (PK) of RAL in a diverse patient population. This was called the REALMRK Study and was an open-label, single-arm study conducted in USA, South America and Southern Africa (5). Treatment-experienced patients either failing or not tolerating current therapy, or those treatment naïve (up to 21% of the total study population) were given RAL at the approved dose of 400 mg twice daily with other approved agents. The study also targeted a minimum enrollment of 25% women and 50% African American patients. The primary endpoint was proportion with HIV RNA <50 copies/mL with discontinuation for virologic failure or adverse events (AEs) included as failures. Of the total study population, 109 men and 97 women were included with 95 (86%) and 80 (82%) competing 48 weeks of treatment, respectively. There were 153 blacks and 53 non-blacks that started treatment with similarly good retention rates. Detailed results were provided from this single arm study breaking results down by gender, race and whether patients were treatment failures for virologic failure, intolerance, or were treatment naïve. After 48 weeks the investigators concluded that RAL was efficacious regardless of gender or race and generally safe and well tolerated. In addition, PK parameters were consistent with previous data with no effect of gender or race (black vs. non-black).
Rilpivirine
Two phase III trials recently characterized the safety and efficacy of a new nonnucleoside reverse transcriptase inhibitor (NNRT), rilpivirine (RPV) in treatment-naïve patients (6, 7). The results of the individual studies led to the recent approval of this drug by the FDA for this patient population. In these trials RPV was shown to be noninferior to EFV in combination with nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs). While there was a clear tolerability advantage of RPV over EFV, there was a suggestion, particularly in the ECHO study of a greater rate of virologic failure with RPV (6). This was particularly evident amongst those coming into the study with higher viral loads. More recently, the FDA approved the single-pill fixed-dose combination of FTC/RPV/TDF for once-daily dosing. At this meeting investigators further characterized the tolerability of RPV versus EFV in these two registrational trials, focusing on the first 12 weeks of treatment (8). The analysis included data from 1368 randomized patients. During the first 12 weeks of therapy, RPV compared with EFV was shown to be associated with lower incidence of grade 2-4 AEs that were characterized as being at least possibly related to treatment (11.5 vs. 24.9%, respectively). For both study arms, the majority of these AEs occurred during the first 4 weeks, with new events being uncommon thereafter. The same was true for discontinuations due to AEs with a total of 9 for RPV and 31 for EFV, with 5 and 26, respectively, occurring during the first 4 weeks. Overall, the incidence of specific grade 2-4 AEs for RPV and EFV, respectively, were dizziness (7.7 vs. 25.2%), nausea (9.6 vs. 10.4%), abnormal dreams (6.7 vs. 10.4%), rash (3.2 vs. 13.9%), headache (5.2 vs. 5.1%) and somnolence (2.9 vs. 6.5%). Change in lipids during the first 12 weeks was also assessed with mean total cholesterol, LDL cholesterol and triglycerides increased for EFV and declined for RPV, with all changes occurring during the first 4 weeks and remaining relatively stable thereafter. In contrast, mean HDL increased in both study arms with a greater increase occurring with EFV than RPV, and a progressive increase observed from baseline to week 2, 4, 8 and 12 in both groups. This data further characterizes both the types of AEs to be expected as well as the timeline in which they are likely to occur when initiating RPV or EFV.
The data with RPV, along with the approval of it as part of a single-pill fixed-dose combination provides a new option for treatment-naïve patients. The primary data demonstrates tolerability advantages over the only other single-pill fixed-dose combination (EFV/FTC/TDF), but leave some lingering concern regarding overall efficacy, particularly amongst those with higher baseline viral loads. For these reasons, neither of these options is perfect for all patients and clinicians will need to individualize therapy. The new combination pill of FTC/RPV/TDF may offer an option for those initiating an alternative regimen but need to switch due to AEs. For example, those on an EFV-based regimen with persistent neurologic symptoms or severe rash may want to change to an alternative one-pill once per day regimen. A concern raised about switching to RPV-containing therapy is that EFV induces cytochrome p450 enzymes and this could result in a reduction in RPV drug levels. A small single-arm study was performed to assess the efficacy of such a switch as well as to characterize the PK of RPV and EFV during the transition period. This study selected those on EFV with full virologic suppression that wanted to switch from EFV to RPV for any one of a number of reasons (9). Importantly, the majority of the 49 individuals enrolled had full virologic suppression for more than one year prior to the switch. Follow-up was limited to 12 weeks, but at that time 49 of 49 had viral loads <50 copies/mL. The accompanying PK study demonstrated that mean EFV levels remained in the therapeutic range for at least 4 weeks after discontinuation, while by 2 to 4 weeks RPV levels reached those seen in patients successfully treated in the phase 3 RPV trials. The investigators concluded that a switch from an EFV- to RPV-containing regimen was safe and that the initially low RPV levels were not clinically relevant. Nevertheless, there are some important caveats to consider. First, the study was limited to those who had no evidence of resistance prior to initiating antiretroviral therapy and remained fully suppressed while on EFV-based therapy. Second, the enrolled subjects were suppressed for prolonged periods of time prior to the switch and therefore the data can not necessarily be extrapolated to those starting EFV with early toxicity prompting a switch, a situation that may occur prior to full suppression or at least within the first weeks or months of treatment. Finally, the follow-up was limited to just 12 weeks.
New therapeutics
Cobicistat
There was some data presented at this meeting on new drugs that are either in the advanced stages of development or very early in clinical trials. One study focused on the new pharmacologic booster Cobicistat (COBI). This drug has now been used in at least two phase 3 trials, the preliminary results of which have been recently reported in the form of press releases. This drug has been developed to pharmacologically boost protease inhibitors; analogous to what ritonavir (RTV) has been used for to date, as well as the new integrase inhibitor elvitegravir (EVG). One of the studies randomized patients to NRTIs with atazanavir (ATV) plus either RTV or COBI. The other study compared fixed dose combination EFV/FTC/TDF with fixed dose combination COBI/EVG/FTC/TDF (QUAD). Preliminary results reported in the recent press releases stated that the primary endpoint of noninferiority to the comparator was shown for both treatment arms. Although major findings related to efficacy and tolerability will not be known until these studies are fully reported, during Phase 2b studies it was noted that COBI may be associated with small increases in creatinine. Further analyses using iohexol to determine actual glomerular filtration rates (aGFR) suggested that this increase in creatinine was not a result of a decline in aGFR but an alteration in the handling of creatinine by renal tubules, a phenomenon of no clinical relevance and previously reported with other commonly used drugs such as cimetadine and trimethoprim, as well as more recently with RPV and dolutegravir, a promising integrase inhibitor in development. The current study included two cohorts, one with estimated GFR (eGFR) ≥80 mL/min and the other with mild/moderate impairment (eGFR 50-79 mL/min) (10). It was a placebo-controlled study with those in the first cohort given COBI at treatment doses of 150 mg daily, RTV at 100 mg/ daily or neither. In the other cohort patients received COBI 150 mg daily for 7 days. Patients also received iohexol to measure aGFR. The study showed that while there were differences in eGFR, there was no change in either cohort in iohexol measured aGFR. An unexplained finding was that COBI exposure was higher in those with mild/moderate renal dysfunction than those without, the mechanism by which this occurs and the clinical relevance is yet to be defined.
Ibalizumab: CD4 Monoclonal Antibody
Ibalizumab is a novel IgG4 humanized monoclonal antibody that binds to an epitope in domain 2 of CD4 resulting in inhibition of HIV entry. Preclinical and phase 1 and 2 studies have demonstrated that it can be given by the intravenous or subcutaneous route with antiviral activity but no interference with CD4 cell signaling. At the 51st ICAAC data was presented from a Phase 2b study that enrolled 113 treatment-experienced patients on failing regimen where they had at least one active drug to add to a background regimen and were assigned to iballizumab 800 mg every 2 weeks or 2000 mg every 4 weeks with the primary endpoint being viral load <50 copies/mL at 24 weeks (11). The patients had mean CD4 of 109 cells/uL and relatively weak background regimens due to substantial baseline drug resistance. The percent of patients with viral loads <50 [<400] copies/mL at week 24 was 44% [58%] in the 800 mg every 2 weeks and 28% [46%] in the 2000 mg every 4 weeks arms (p=0.16) [p=0.32]. Key secondary endpoints was median change from baseline in viral load at 24 weeks being -1.8 log10 copies/mL in the 800 mg versus -1.5 log10 copies/mL in the 2000 mg study arms with percent having at least 1 log10 copies/mL reduction from baseline being 63% and 57%, respectively. Mean change from baseline in CD4 at week 24 were +37 in the 800 mg every 2 week group and +40 cells/uL in the 2000 mg every 4 week arm. Serious AEs were as expected in a population with advanced HIV disease with none being attributed to study drug. Treatment-emergent AEs occurring at ≥5% included a variety of nonspecific clinical events such as rash (12.4%), diarrhea (8.0%), headache (7.1%), upper respiratory tract infection (7.1%), nausea (7.1%), fatigue (6.2%), cough (5.3%), nasopharyngitis (5.3%) and oral candidiasis (5.3%). This drug continues to show promise for those who are highly treatment-experienced in need of new options. Moreover, it offers the potential for intermittent dosing for those in need of longer-acting agents. Further studies are underway to determine how safe and effective the drug is as a subcutaneous infusion.
Zinc Finger Nuclease
Perhaps the most novel treatment strategy presented at the 51st ICAAC was that of a gene therapy strategy designed to modify the expression of the CCR5 chemokine receptor on the surface of a patient's cells. This study attempts to build upon the intriguing observations made in the now well-described "Berlin Patient," an HIV-infected individual who received a stem cell transplant as part of his treatment for leukemia. The donor was homozygous for the CCR5 delta 32 gene which confers genetic resistance to HIV infection (12, 13). Subsequent to the stem cell transplant the Berlin Patient has remained HIV-free while off of antiretroviral therapy for several years. The goal of the current treatment strategy was to modify the cells of a HIV-infected patient by the introduction of a Zinc Finger Nuclease (ZFN) that would modify the CCR5 gene on autologous CD4+ T-cells so that a subset will be resistant to HIV infection (14). Preclinical data showed that disruption of CCR5 by ZFN did not affect CD4+ T-cell proliferation or function. The current study was an open-label, single-dose study of nine HIV-infected individuals with undetectable plasma HIV RNA on antiretroviral therapy with suboptimal CD4 response (200-500 cells/uL). All subjects were leukapheresed, after which CD4+ cells were enriched, activated and transduced by Adenoviral SB-728. These cells were expanded and cryopreserved, 25% of which had been modified by the transduction process. Each patient received a single infusion of cells with three groups of three receiving either 1.0 x 1010, 2.0 x 1010 or 3.0 x 1010 cells. There have been no serious AEs reported after a median follow-up of 337 days. The primary AEs attributed to study drug were those occurring within 24 hours of infusion with headache, chills, fever, sweats, dizziness, fatigue and "garlic" body odor, the latter attributed to substance use for the cryopreservation of cells. All of these AEs were reversible and resolved without sequelae. From an efficacy perspective, there was a notable increased in CD4+ T-cells seen shortly after infusion that then declined to a relative steady state which was approximately 100 cells/uL higher than baseline. There was also an increase in CD8+ T-cells and normalization of CD4:CD8 ratio. A critical finding in the study was persistence of identifiable CCR5 modified T-cells in the peripheral blood and in rectal mucosa, suggesting a certain level of engraftment with trafficking to lymphoid tissue. This study provided early evidence that the ZFN can be efficiently introduced into cells and persists in blood and tissue of HIV-infected patients. As with all gene therapy strategies, long-term follow-up will be needed to assure that no important AEs occur over time.
The next question that emerged from this small pilot study is whether the engrafted CCR5-modified cells would have an impact on HIV control if antiretroviral therapy was discontinued. In the primary presentation, a single patient interrupted treatment with a rapid rebound in viral load with subsequent decline to steady state of approximately 10-fold below the peak. The challenge is distinguishing whether such a rebound and then decline would have happened had ZFN modified cells not existed. A Late Breaker abstract reported on 6 aviremic patients who had CD4 >450 cells/uL and were infused with 1.0 x 1010 ZFN-modified cells and then 4 weeks after infusion underwent a 12 week treatment interruption (15). The infused cells had mean CCR5 modification of 17% (range: 11-25%) and patients were followed for median of 150 days. There was a substantial boost in CD4+ T-cells after the infusion with normalization of CD4:CD8 ratio. During the treatment interruption plasma HIV RNA rebounded at 2-6 weeks, peaking at 6-8 weeks and then declining at 12 weeks. The presentation included estimated pre-treatment plasma HIV RNA for comparison with level after initial peak. Results are obviously limited by small numbers and brief treatment interruption, but the 12 week viral load varied from approximately that of the pre-treatment levels, to at least in one case considerably lower than that prior to original initiation of antiretrovirals. This is extremely preliminary data that at least in the short-term does not demonstrate profound viral control. That said, it certainly justifies further research both into the use of this product as well as how those treated with ZFN contrasts with the many events that led to the profound success and possible "cure" seen in the Berlin Patient.
HCV Therapy
As we rapidly enter the time of direct acting antiviral agents against HCV it is important for HIV clinicians to both follow the emerging data in mono-infected patients, where these drugs are first being tested, as well as await new data related to drug-drug interactions. One of the two recently approved HCV protease inhibitors, Telapravir (TVR) is metabolized via CYP3A and non-CYP mediated pathways and is a potent inhibitor of CYP3A. Data on drug-drug interactions between TVR and antiretroviral therapy are starting to emerge. At this meeting data was presented from HIV-uninfected controls on the interaction between TVR and RAL (raltegravir) (16), the latter being primarily metabolized by conversion to glucuronide metabolite (RAL-gluc) via uridine diphosphate glucuronosyltransferase isozyme 1A1 (UGT1A1). In an open-label cross-over trial 21 healthy volunteers were given the approved dose of TVR, 750 mg every 8 hours followed by RAL at 400 mg twice-daily and a second group RAL 400 mg twice daily followed by the introduction of TVR 750 mg every 8 hours. This study showed that RAL did not influence the PK of TVR, but TVR increased RAL and RAL-gluc by 31 and 37%, respectively. This interaction is not considered clinically significant and suggests that these drugs can be used together without dose adjustment or concerns regarding effect on safety or efficacy.
Results from a Phase 2a dose finding study of pegIFN alfa-2a plus ribavirin (RBV) with or without BMS-790052 (052), a first-in-class, highly selective and potent HCV NS5A replication complex inhibitor was presented in HCV genotype 1 mono-infected patients (17). Enrolled subjects were HCV-treatment naïve and randomized to PegIFN alfa-2a plus RBV with either 052 placebo or 3, 10 or 60 mg of 052 given once daily (n=12 per study arm). Therapy was given for 48 weeks with follow-up. HCV RNA dropped dramatically in all arms containing 052 when compared to those given 052 placebo, with a suggestion of superior virologic response in the 10 and 60 mg arms. The virologic responses are summarized in the below table.
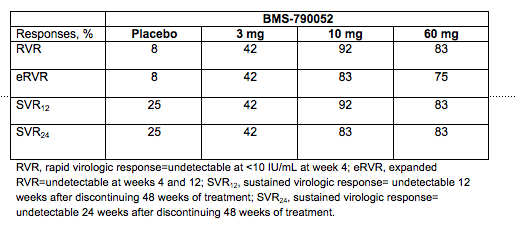
Amongst those with virologic failure emergent resistance did occur with previously identified drug resistance mutations. On treatment AEs were largely consistent with those seen in PegIFN-alfa-2a plus ribavirin therapy. There were more AE-associated treatment discontinuations in those receiving 052 at 60 mg/d than the other group (n=4), with all being for different reasons such as anxiety, rash, alopecia and lymphopenia. This data is very supportive of larger studies using this drug with PegIFN/RBV as well as in combination with other oral anti-HCV agents. Nevertheless, as is increasingly becoming a theme with HCV direct acting antivirals, resistance is a real possibility and will be major issue to be explored in future phase 3 studies.
Disclosures:
Dr. Daar has served as a consultant for Merck, Gilead, ViiV, Bristol Myers Squibb and received research support from Abbott, ViiV, Gilead, Merck and Pfizer.
References
1. Landovitz RJ, Tseng C Weissman M, et al. Epidemiology and sexual risk behavior of MSM using GRINDR in Los Angeles, California. 51st ICAAC 2011, Abstract H1-1149.
2. Khawcharoenporn T, Kendrick S, Smith K. Human immunodeficiency virus risk perception and interest in pre-exposure prophylaxis among persons visiting a sexually-transmitted infection clinic in Chicago. 51st ICAAC 2011, Abstract H1-1148.
3. Grant R, Lama J, Anderson P, McMahan V, Liu A, Vargas L, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363:2587-99.
4. Lazzarin A, DeJesus E, Rockstroh K, et al. Durable and consistent efficacy of raltegravir (RAL) with tenofovir (TDF) and emtricitabine (FTC) across demographic and baseline prognostic factors in treatment-naïve patients from STARTMRK at week 156. 51st ICAAC 2011, Abstract H2-790.
5. Squires K, Bekker L-G, Eron J, et al. Safety, tolerability, and efficacy of raltegravir (RAL) in a diverse cohort of HIV-infected patients: 48-week results from the REALMRK Study. 51st ICAAC 2011, Abstract H2-789.
6. Molina JM, Cahn P, Grinsztejn B, et al. Rilpivirine versus efavirenz with tenofovir and emtricitabine in treatment-naïve adults infected with HIV-1 (ECHO): a phase 3 randomised double-blind active-controlled trial. Lancet 2011; 378"238-246.
7. Cohen CJ, Andrade-Villanueva J, Clotet B, et al. Rilpivirine versus efavirenz with two background nucleoside or nucleotide reverse transcriptase inhibitors in treatment-naïve adults infected with HIV-1 (THRIVE): a phase 3, randomized, non-inferiority trial. Lancet 2011; 378:229-237.
8. Rashbaum B, Girard P-M, Rachlis A, et al. Rilpivirine (TMC278) tolerability over the first 12 weeks of treatment in the Phase III ECHO and THRIVE studies. 51st ICAAC 2011, Abstract H2-805.
9. Mills A, Cohen C, DeJesus E, et al. Switching from efavirenz/emtricitabine/tenofovir disoproxil fumarate (EFV/FTC/TDF) single tablet regimen (STR) to emtricitabine/rilpivirine/tenofovir disoproxil fumarate (FTC/FPV/TDF) STR in virologically suppressed, HIV-1 infected subjects. 51st ICAAC 2011, Abstract H2-794c.
10. German P, Liu C, Warren D, et al. Effect of cobicistat on glomerular filtration rate (GFR) in subjects with normal and impaired renal function. 51st ICAAC 2011, Abstract H2-804.
11. Khanlou H, Gathe J, Schrader S, et al. Safety, efficacy and pharmacokinetics of ibalizumab in treatment-experienced HIV-1 infected patients: a Phase 2b study. 51st ICAAC 2011, Abstract H2-794b.
12. Hutter G, Nowak D, Mossner M, et al. Long-term control of HIV by CCR5 Delta32/Delta32 stem-cell transplantation. N Engl J Med 2009; 360:692-8.
13. Allers K, Hutter G, Hofmann J, et al. Evidence for the cure of HIV infection by CCR5∇32/∇32 stem cell transplantation. Blood 2011; 117:2791-9.
14. Mitsuyasu R, Lalezari J, Deeks S, et al. Adoptive transfer of Zinc Finger Nuclease CCR5 modified autologous CD4 T-cells (SB-728-T) to aviremic HIV-infected subjects with suboptimal CD4 counts (200-500 cells/mm3). 51st ICAAC 2011, Abstract H1-375.
15. Ando D, Tang WW, Stein D, et al. HAART treatment interruption following adoptive transfer of Zinc Finger Nuclease (ZFN) modified autologous CD4 T-cells (SB-728-T) to HIV-infected subjects demonstrates durable engraftment and suppression of viral load. 51st ICAAC 2011, H2-794a.
16. Van Heeswijk R, Garg V, Boogaerts G, et al. The pharmacokinetic interaction between telaprevir and raltegravir in healthy volunteers. 51st ICAAC 2011, Abstract A1-1738a.
17. Pol S, Ghalib RH, Rustgi VK et al. High rates of SVR24 for BMS-790052, an NS5A replication complex inhibitor, in combination with PegIFN-alfa-2a and ribavirin: Phase 2a trial in treatment-naïve HCV genotype 1 subjects. 51st ICAAC 2011, Abstract H1-376.
|
| |
|
 |
 |
|
|