 |
 |
 |
| |
Population and Clonal Analysis of Resistance-Associated Amino Acid Variants (RAVs) in Viruses From Patients Enrolled in a Phase 2b Study of MK-7009 (Vaniprevir): 'HCV Protease Resistance Persists-Clonal Sequencing-novel double/triple mutations detected up to 300 days after stopping HCV therapy'
|
| |
| |
Reported by Jules Levin
HEP DART 2011, December 4-8th 2011, Kauai, HI, USA
Carolyn McHale1, Richard Barnard1, David Nickle1, William Newhard1, Carol Cheney1, Don Graham1, Amy Himmelberger1, Julie Strizki1, Peggy Hwang1,
Niloufar Mobashery1, Amber Rivera2, Jacqueline Reeves2, and Daria Hazuda1
1Merck & Co., Inc., West Point and North Wales, PA; 2Monogram Biosciences Inc., South San Francisco, CA
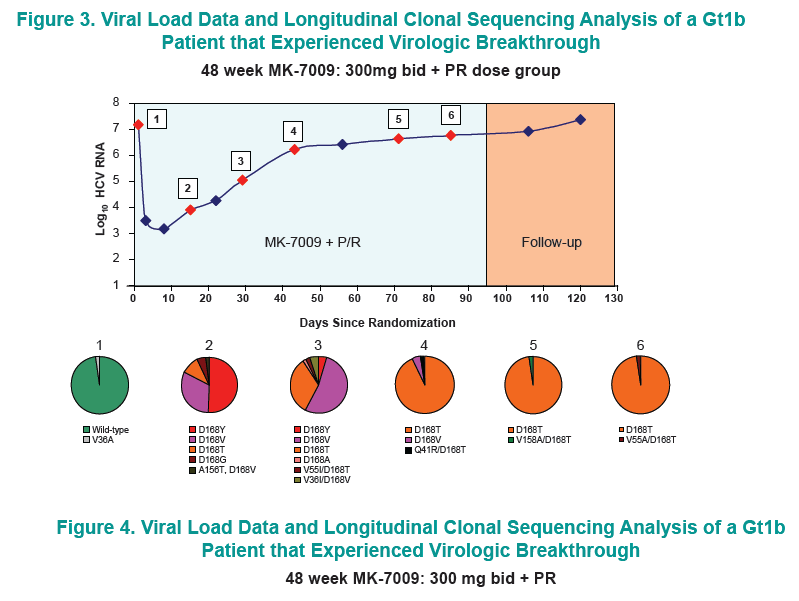
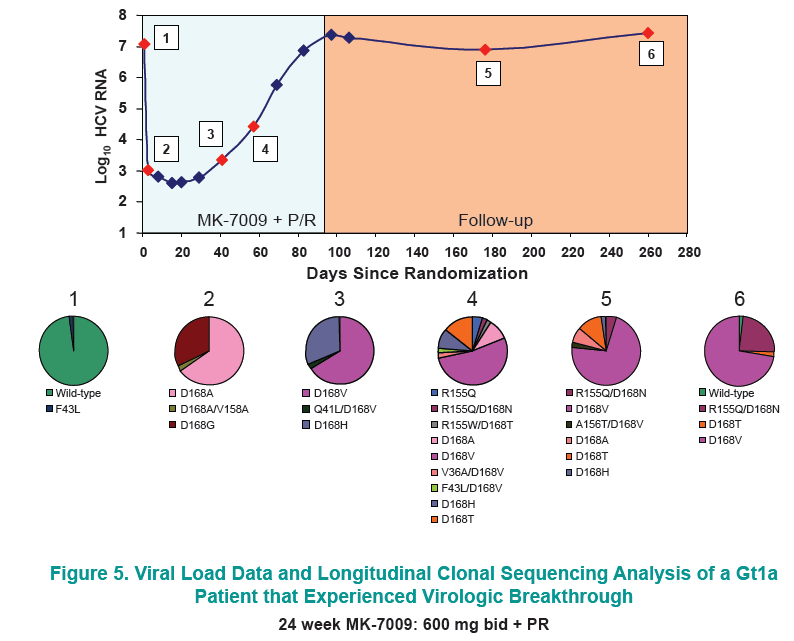
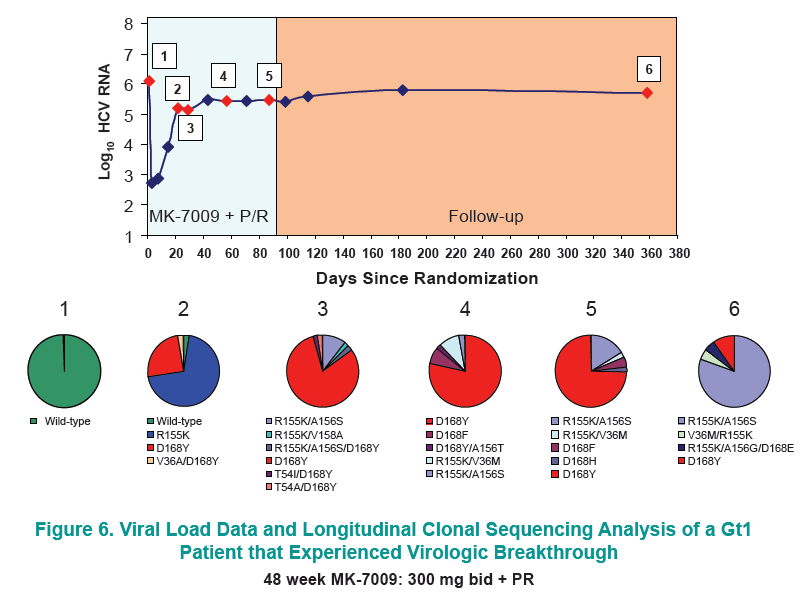
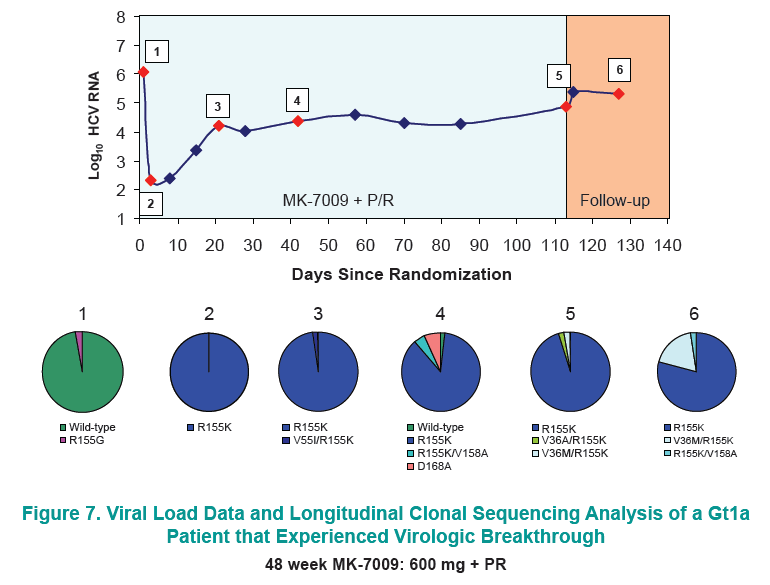
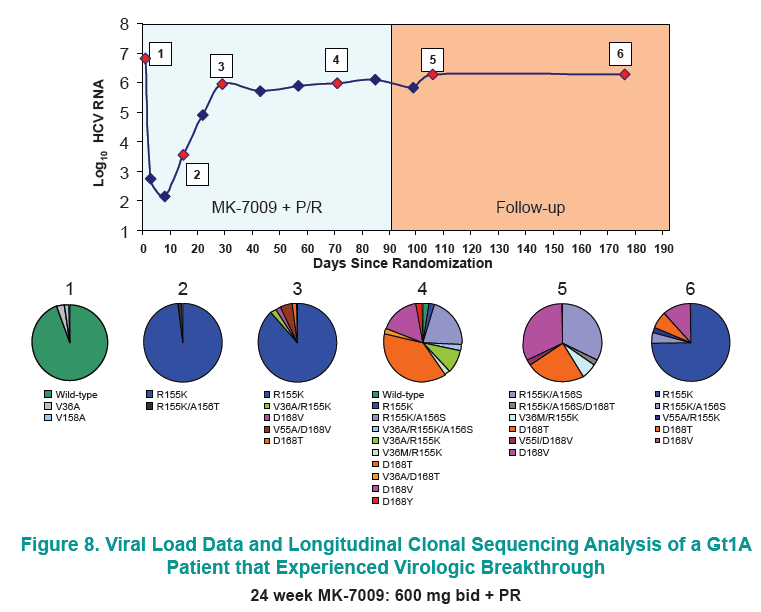
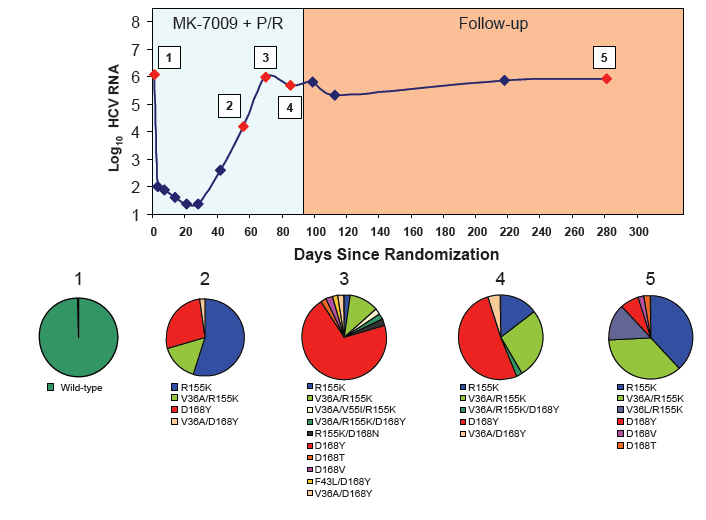
Longitudinal clonal sequence analysis of six virological failure samples was performed by Monogram Biosciences......(from Jules: "after virologic failure in 6 patients with resistance associated mutations (RAVs) identified by population sequencing they remained on failing therapy for as long as 270 days after stopping therapy, and during that time on failing therapy novel double & triple RAV combinations emerged.....Clonal analysis revealed an increase in the number of RAVs detected during the MK-7009 dosing period. At later time points during MK-7009 dosing, double/triple RAVs were detected more frequently. During followup, after stopping therapy the number of double/triple mutations declined but at the end of a 24-week followup which was as long as 220 days post-therapy sometimes the novel double & triple mutation combinations remained present. The most obvious observation is this study finds the potential outcomes of remaining on failing therapy for 12 weeks, where mutations accumulate and novel double & triple mutation combinations emerge, and during the 24 week followup although the number of single, double & triple mutations decline by clonal sequencing many of them still remain, bringing into question what would the response be to retreatment with similar HCV protease inhibitors".

ABSTRACT:
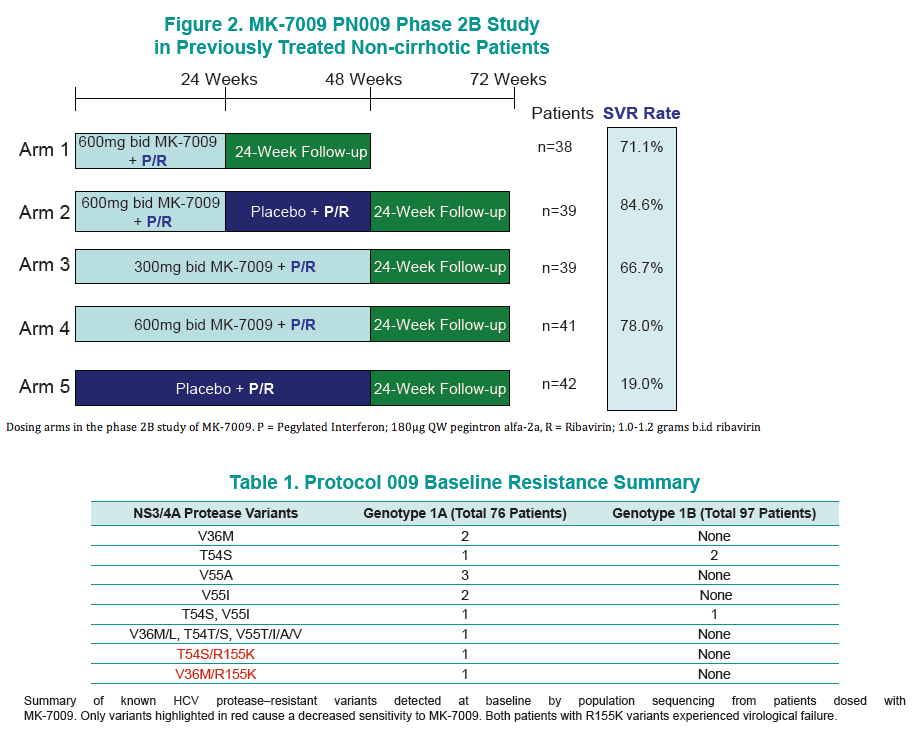
BACKGROUND: MK-7009 is an oral HCV-NS3/4A protease inhibitor being assessed in combination with peginterferon alfa-2a (P) 180 μg QW and ribavirin (R) in patients with chronic hepatitis C genotype 1 infection. This Phase 2b study evaluated four treatment regimens using MK7009 compared to 48 weeks of PR in patients who have failed previous PR therapy: 1) 24 weeks of MK-7009 600 mg bid with PR; 2) 24 weeks of MK-7009 600 mg bid with PR followed by 24 weeks of PR; 3) 48 weeks of MK-7009 300 mg bid with PR; 4) 48 weeks of MK-7009 600 mg bid with PR.
METHODS: For efficacy analyses, viral load measurements were made using the Roche Cobas Taqman assay with a lower limit of detection of 10 IU/mL. For resistance analyses, population resistance analysis was performed on baseline samples from all patients and on samples from non-SVR patients at the time of virologic failure, defined by the study protocol. For technical reasons, resistance sequencing was performed on samples with a viral load >1000 IU/mL. In addition, longitudinal clonal analysis of virological failure samples was performed by Monogram Biosciences.
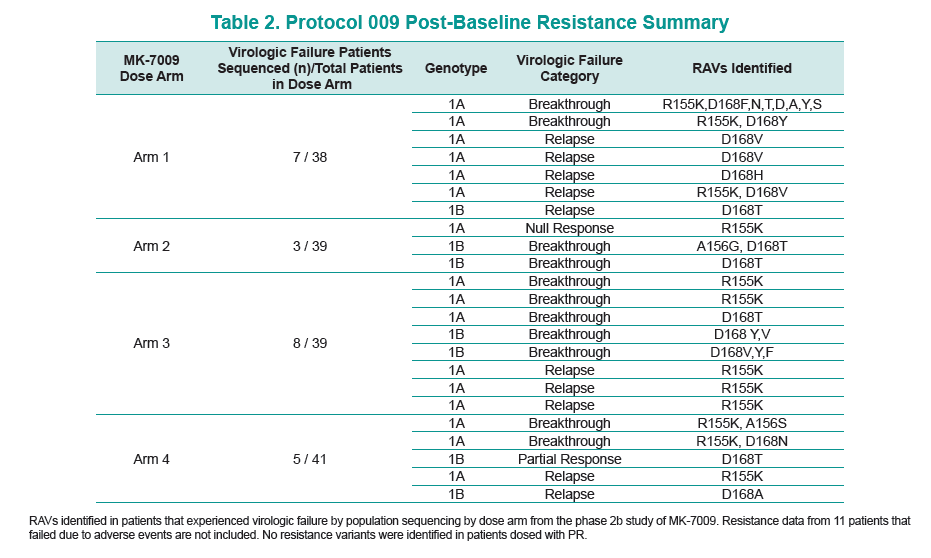
RESULTS: Baseline resistance sequence data was available for 150 patients that received MK7009. R155K was detected in 2/150 patients, both of which experienced virologic failure; this was the only MK- 7009 RAV detected at baseline.
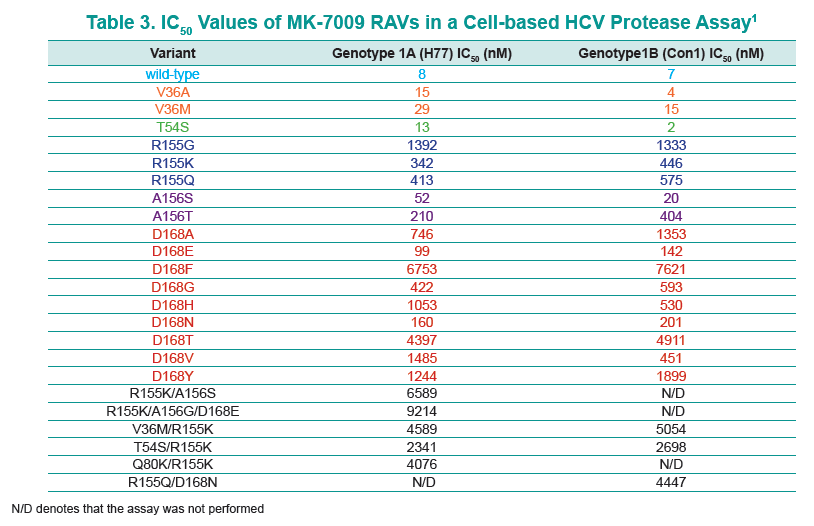
28 patients experienced virologic failure that received MK7009. Of these 22/28 (79%) had viruses with RAVs observed at positions R155, A156 and/or D168. An additional patient that discontinued study medication early had no detectable RAVs. When analyzed by MK-7009 dose-group, 7/9, 3/3, 8/11 and 5/5 virologic failure patients had viruses with detectable RAVs in dose groups 1 through 4, respectively. RAVs at R155, A156 and D168 were detected in genotype 1a infected non-SVR patients, while RAVs at D168 were detected in genotype 1b infected patients by population sequencing. Six patients with RAVs detected by population sequencing were selected for longitudinal clonal sequence analysis. This analysis revealed a number of novel double/triple MK-7009 RAV combinations such as R155K/A156S and R155K/ A156G/D168E in a genotype 1a infected patient, and R155Q/D168N in a genotype 1b infected patient. (from Jules: after failing therapy novel double/triple resistance mutations combinations emerged suggesting to me that in the clinic emergence & resolution of mutations or mutations becoming undetectable may not be as suggested by recent studies finding that mutations were not detectable 9 months or 2 years after stopping therapy, because what this study suggests is that novel mutation combinations might emerge that were not looked for in the studies recently reported finding that after therapy is stopped, 9-24 months later, mutations were not detected by population sequencing, suggesting these recently presented at conferences resistance studies may not be detecting all the resistance present.
CONCLUSION: Variants were observed at positions R155, A156 and D168 in non-SVR patients from the MK-7009 phase 2B study. In 6 patients analyzed by clonal sequencing, the pattern of RAVs became more complex over time, with the selection of double and triple RAVs that exhibited larger fold-shifts in IC50 against MK-7009.

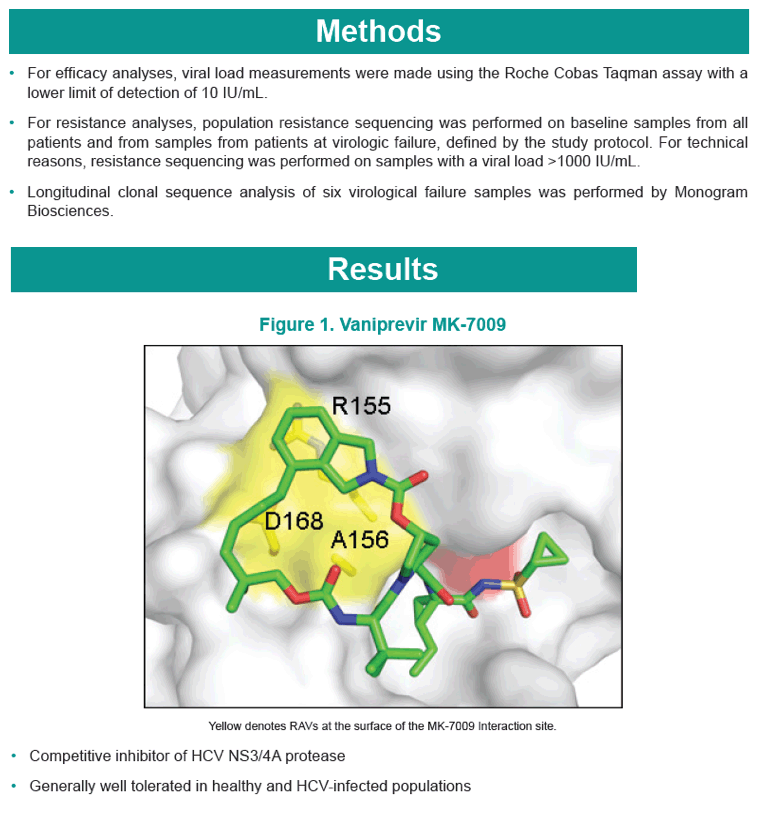
|
| |
|
 |
 |
|
|