 |
 |
 |
| |
Safety and Antiviral Activity of MK-5172, a Next Generation HCV NS3/4A Protease Inhibitor with a Broad HCV Genotypic Activity Spectrum and Potent Activity Against Known Resistance Mutants, in genotype-1 and -3 HCV-Infected Patients
|
| |
| |
Reported by Jules Levin
HEP DART Dec 4-8 2011, Koloa, HI
Presented by Ian Fraser
IP Fraser1, A Petry1, K Van Dyck1, RB Nachbar1, I De Lepeleire1, M Robberechts1, L Han1, J Palcza1, V. Moiseev2, Z. Kobalava2, F Wagner3, M Uhle3, JA Wagner1
1 Merck Sharp & Dohme Corp., Whitehouse Station, NJ, 2ASCENT Clinical Research Solutions OOO, Moscow, Russia, 3Charité Research Organisation GmbH, Berlin, Germany
New from AASLD are:
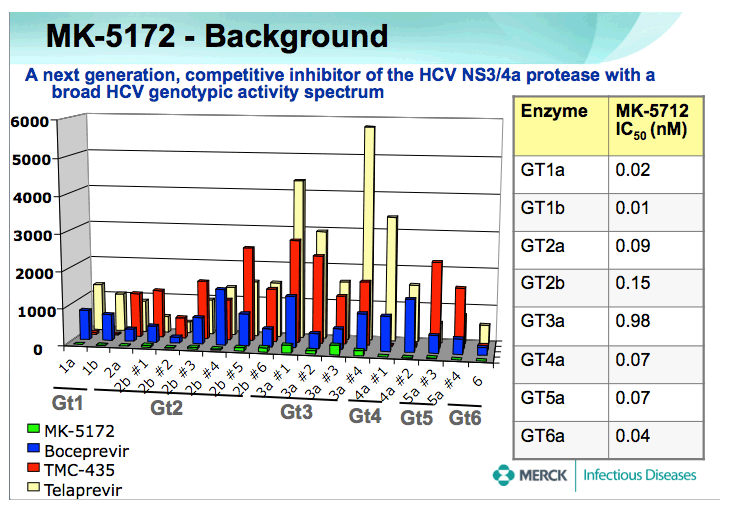
1. Updated in vitro potency data (slide #3)
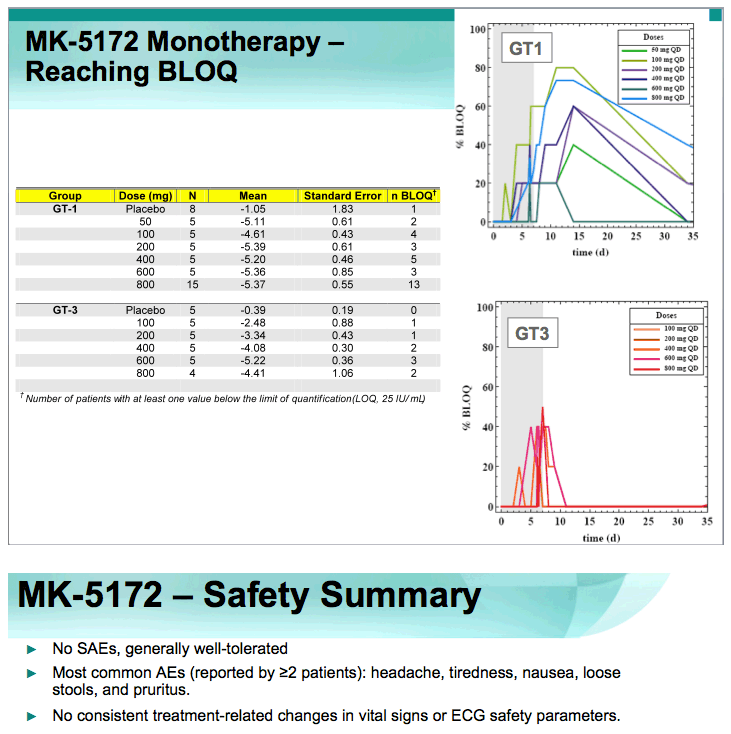
2. BLOQ data (slide #9).
⇒MK-5172 demonstrated potent and rapid viral load reductions with 7 days of monotherapy
-75% (30/40) of patients with chronic GT1 infection achieved reductions in plasma HCV RNA to BLOQ.
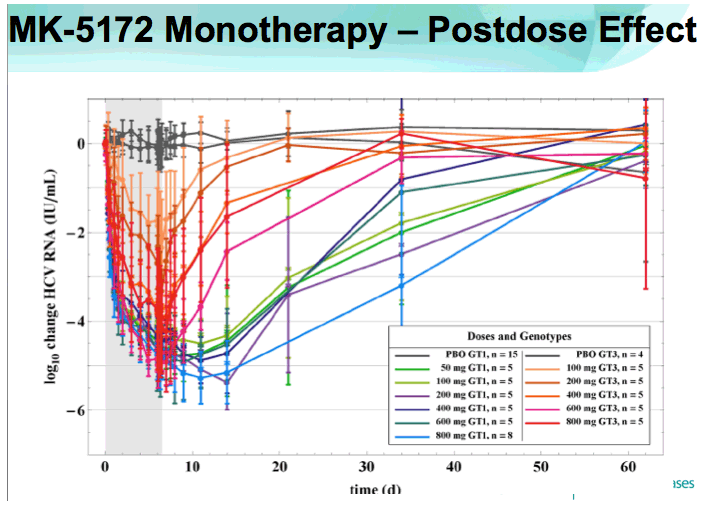
⇒Antiviral activity persisted for several days beyond the treatment period in GT-1 patients.
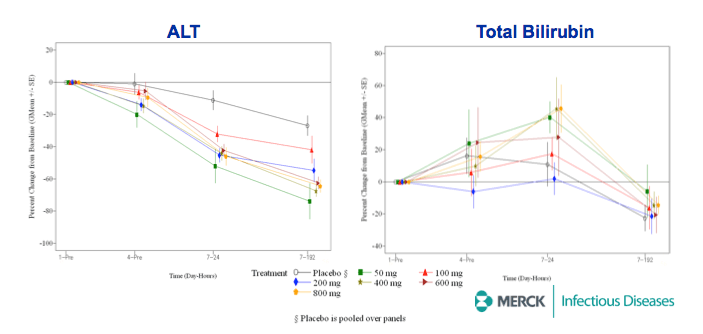
⇒MK-5172 was generally well-tolerated with no SAEs, and no discontinuations due to AEs or safety laboratory abnormalities.
-The current study is ongoing (GT1, daily doses <50mg)
⇒These findings support further clinical investigation of MK-5172 for the treatment of chronic HCV-infection.
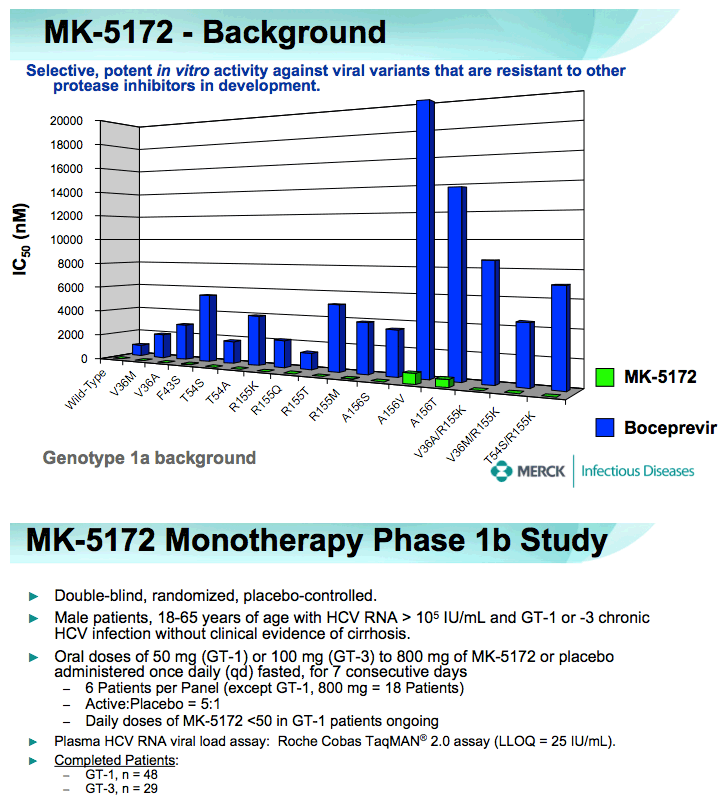
⇒Multiple oral doses of MK-5172 monotherapy up to 800 mg daily for 7 days were generally well-tolerated in HCV-infected patients.
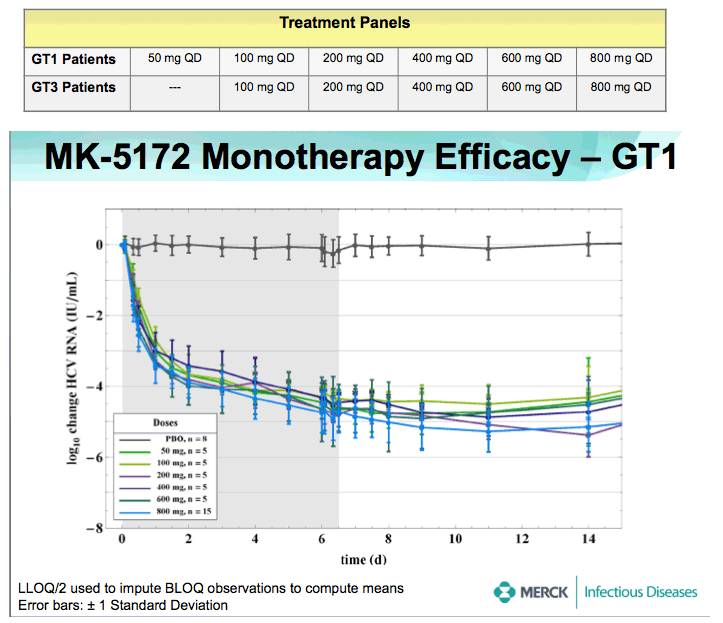
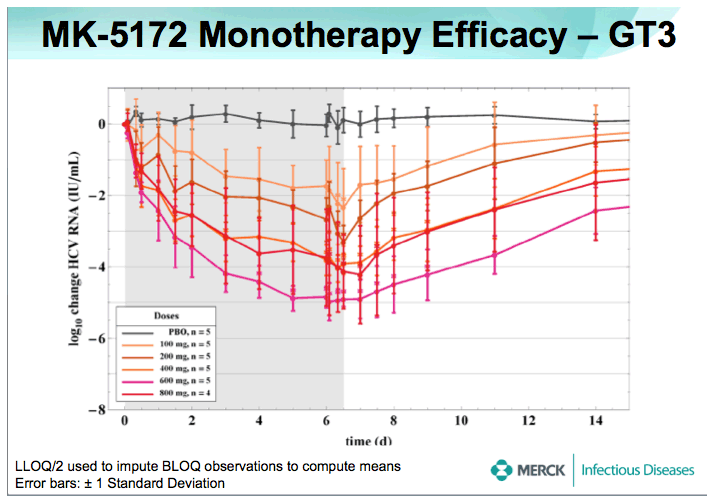
⇒HCV viral RNA declined rapidly after the first dose of MK-5172.
-Mean maximum reductions from baseline of HCV viral RNA (SEs) were 5.4 (0.38) and 5.22 (0.16) log10 IU/mL for GT-1 and -3, respectively.
⇒No on-treatment viral load rebound was observed in any patient.
⇒Mean viral load reductions with 7 days of MK-5172 monotherapy in GT-1 patients were similar among all dose groups from 50- to 800-mg qd. However, more patients achieved viral load suppression to below the limit of quantitation at higher doses, with a greater proportion of patients with plasma HCV RNA persisting below the limit of quantitation at higher doses.
-75% (30/40) of GT- 1, and 38% (9/24) of GT- 3 HCV-infected patients achieved HCV RNA levels below the limit of quantitation with a 7 day monotherapy course of MK-5172
-87% (13/15) of GT-1 HCV-infected patients treated with 800 mg MK-5172 qd monotherapy achieved undetectable plasma HCV RNA below the limit of quantitation.
⇒Mean time to viral load nadir > 2 days after last dose.
⇒By the 1 month follow-up visit, mean plasma levels of HCV RNA were still below baseline for all dose levels in GT-1 patients, and for the highest dose levels in GT-3 patients


|
| |
|
 |
 |
|
|