 |
 |
 |
| |
Safety and Antiviral Activity of MK-5172, a Next Generation HCV NS3/4A Protease Inhibitor with a Broad HCV Genotypic Activity Spectrum and Potent Activity Against Known Resistance Mutants, in genotype-1 and -3 HCV-Infected Patients: '75% (30/40) of GT-1, and 38% (9/24) of GT-3 HCV-infected patients achieved plasma HCV RNA below the limit of quantitation with a 7 day monotherapy course of MK-5172'
|
| |
| |
Reported by Jules Levin
HEP DART Dec 4-8 2011, Koloa, HI
Presented by Ian Fraser
IP Fraser1, A Petry1, K Van Dyck1, RB Nachbar1, I De Lepeleire1, M Robberechts1, L Han1, J Palcza1, V. Moiseev2, Z. Kobalava2, F Wagner3, M Uhle3, JA Wagner1
1 Merck Sharp & Dohme Corp., Whitehouse Station, NJ, 2ASCENT Clinical Research Solutions OOO, Moscow, Russia, 3Charité Research Organisation GmbH, Berlin, Germany
New from AASLD are:
1. Updated in vitro potency data (slide #3)
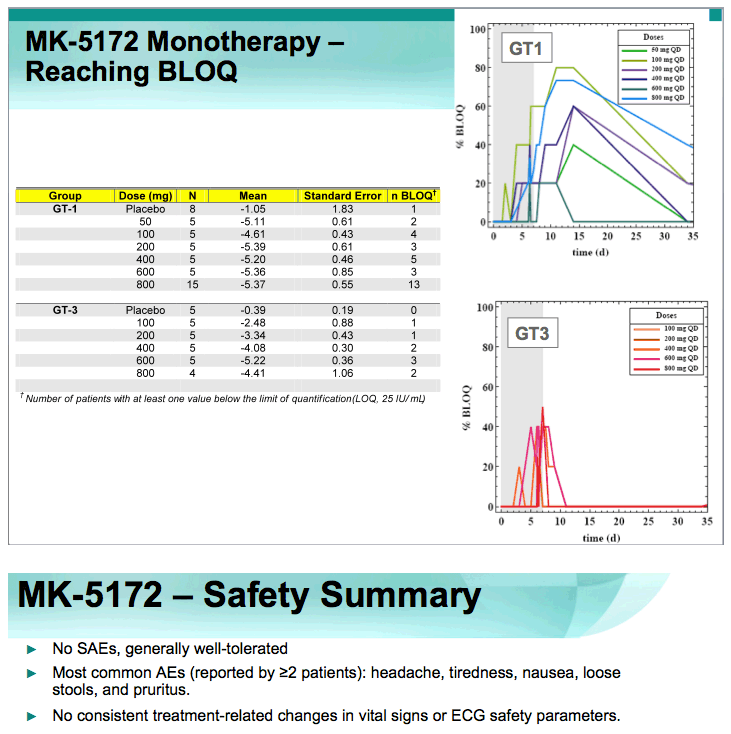
2. BLOQ data (slide #9).
ABSTRACT:
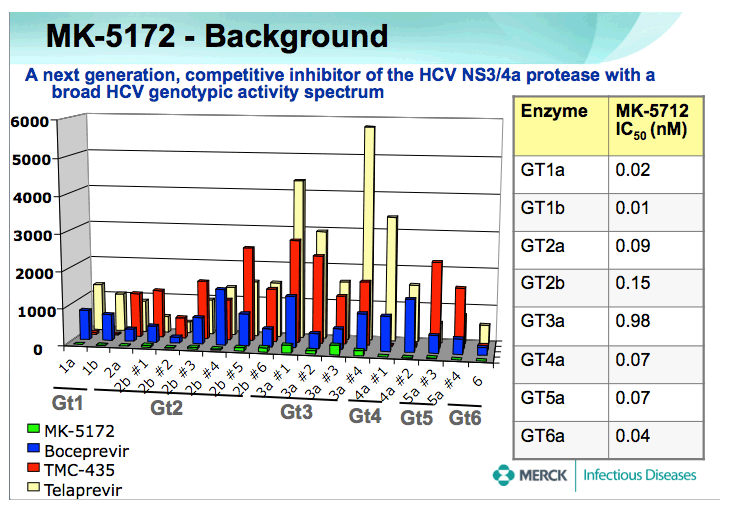
BACKGROUND: MK-5172 is a next generation, competitive inhibitor of the HCV NS3/4A protease with a broad HCV genotypic activity spectrum, and with selective, potent in vitro activity against viral variants that are resistant to other protease inhibitors in development. This placebo-controlled study assessed the safety, tolerability and antiviral efficacy of MK-5172 administered as 7 days of monotherapy to adult male patients with chronic genotype (GT) -1 or -3 HCV infection.
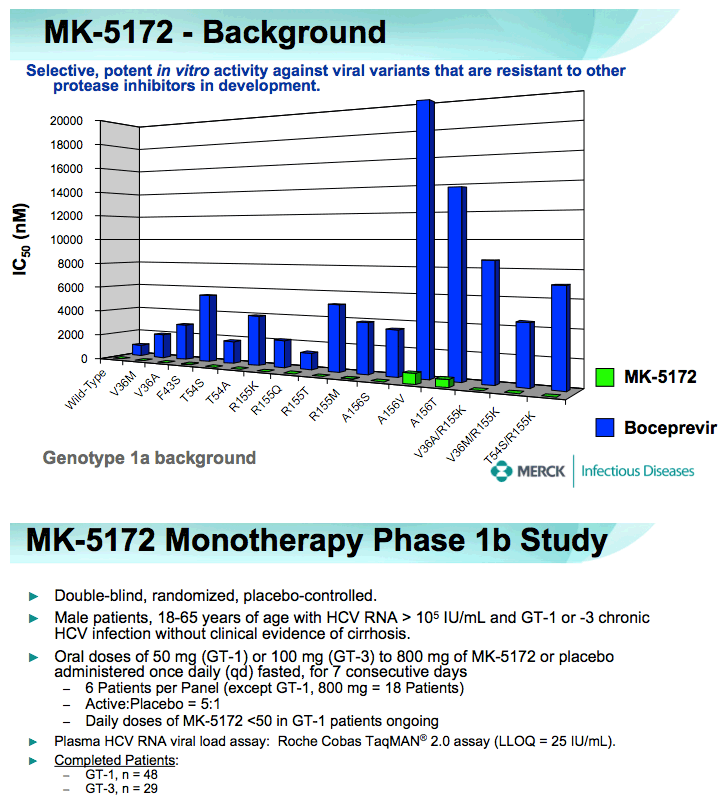
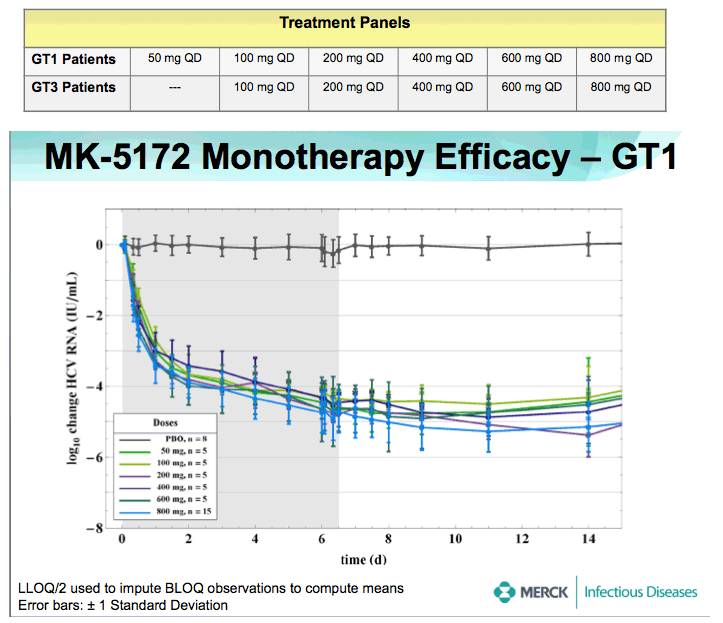
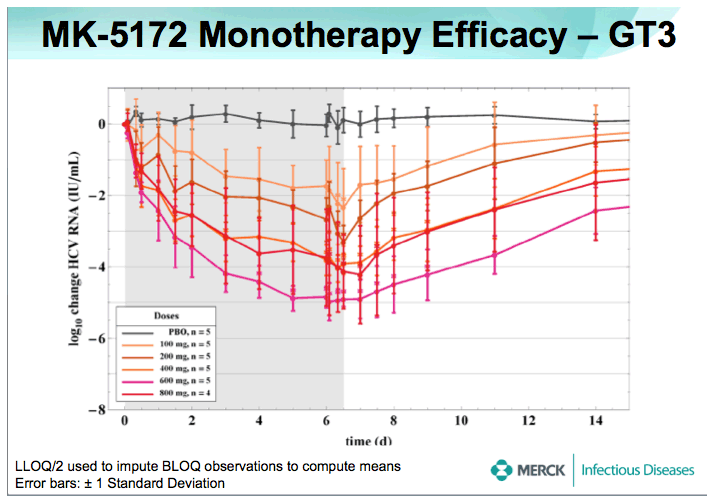
METHODS: 78 Males, 18-65 years old with HCV RNA > 105 IU/mL, and GT-1 or -3 chronic HCV infection without clinical evidence of cirrhosis, were randomized to receive placebo or doses of 50 mg (GT-1) or 100 mg (GT-3) to 800 mg MK-5172 once daily for 7 days (MK- 5172:placebo ratio of 5:1 for separate panels of GT-1 and GT-3 patients). Patients were followed for up to 2 months after the last dose. Safety and tolerability were evaluated using laboratory values, electrocardiograms (ECG), and evaluation of adverse experiences (AEs). Antiviral efficacy was assessed through quantitation of plasma HCV RNA using the Roche Cobas TaqMAN 2.0 assay (lower limit of quantitation = 25 IU/mL) at multiple time points before, during, and after the MK- 5172/placebo dosing period.
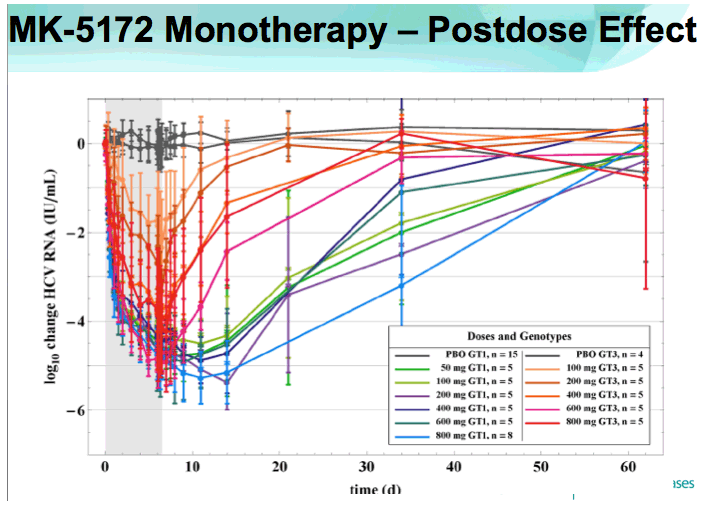
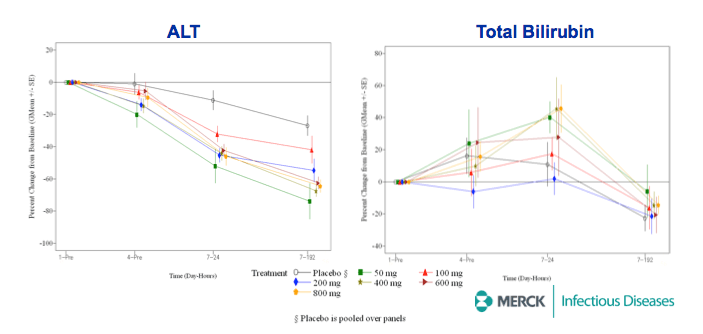
RESULTS: Plasma HCV RNA declined rapidly after initiation of dosing. Maximum mean reductions from baseline HCV-RNA were 5.4 and 5.2 log10 IU/ mL for GT-1 and GT-3 patients, respectively. No on-treatment viral rebound was observed. Mean viral load reductions with 7 days of MK-5172 monotherapy in GT-1 patients were similar among all dose groups from 50- to 800-mg qd. However, more patients achieved viral load suppression below the limit of quantitation at higher doses, with a greater proportion of patients with plasma HCV RNA persisting below the limit of quantitation at higher doses. In GT-3 patients, suppression of HCV RNA was reduced at doses below 400 mg. In GT-1 patients, HCV-RNA continued to decline after cessation of dosing, with the mean time to nadir more than 2 days after last dose. One month after completion of dosing, HCV-RNA remained below baseline levels in some individuals. MK-5172 was generally well-tolerated, with all adverse experiences transient, and the majority being mild in intensity.
CONCLUSIONS: MK-5172 administered once daily exhibits potent antiviral activity during 7 days of monotherapy in patients with chronic GT-1 and GT-3 HCV infections. Following a rapid and profound decline from baseline, plasma HCV RNA remained suppressed for several days beyond the treatment period in both genotypes. 75% (30/40) of GT-1, and 38% (9/24) of GT-3 HCV-infected patients achieved plasma HCV RNA below the limit of quantitation with a 7 day monotherapy course of MK-5172. MK-5172 was generally well-tolerated. These findings support continued clinical investigation.


|
| |
|
 |
 |
|
|