 |
 |
 |
| |
Treatment Week 12/24 Stopping Rules for Boceprevir (BOC) Combination Therapy with Peginterferon+Ribavirin (PR):Exploratory Analyses of SPRINT-2 and RESPOND-2
|
| |
| |
Reported by Jules Levin
HEPDART- Frontiers in Drug Development for Viral Hepatitis December 4 - 8, 2011 Koloa, Hawaii
Ira M. Jacobson MD1, Patrick Marcellin MD2, Stefan Zeuzem MD3, Mark S. Sulkowski MD4, Rafael Esteban MD5, Lisa D. Pedicone PhD6, Navdeep Boparai PhD6, Margaret Burroughs MD6, Clifford A. Brass MD PhD6, Janice K. Albrecht PhD6, Jean-Pierre Bronowicki MD, PhD7
1Weill Cornell Medical College, New York, NY; 2University Paris-Diderot, Hôpital Beaujon, Clichy, France; 3J.W. Goethe University Hospital, Frankfurt, Germany; 4Johns Hopkins University School of Medicine, Baltimore, MD;5Hospital General Universitario Vall d'Hebron, Barcelona, Spain; 6Merck Sharp & Dohme Corp., Whitehouse Station, NJ; 7Universite Henri Poincare-Nancy, Vandoeuvre-lès-Nancy, France

ABSTRACT
BACKGROUND: HCV-NS3 protease inhibitor BOC combined with PR is effective for HCV-1 treatment. Stopping rules used in HCV PR therapy include <2 log viral load decline at TW12 and/or detectable HCV-RNA at TW24. Early stopping helps avoid therapy/toxicity in patients who are unlikely to achieve sustained virologic response (SVR) and may be important in minimizing the development of resistance-associated variants (RAVs) with regimens containing protease inhibitors.
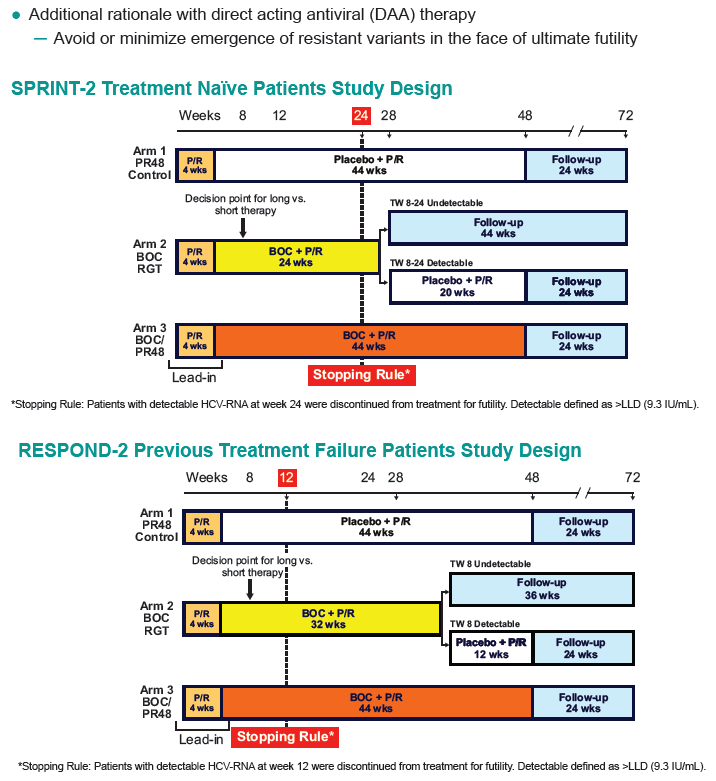
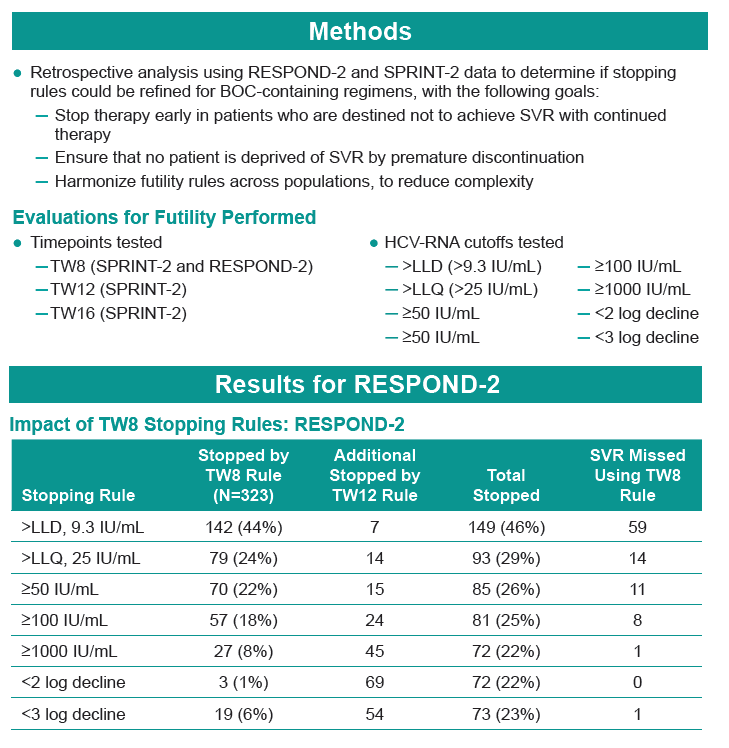
METHODS: Exploratory analysis using SPRINT-2 (treatment-naïve) and RESPOND-2 (prior treatment failures) data to determine if stopping rules (detectable HCV-RNA at TW24 for SPRINT-2 and TW12 for RESPOND-2) could be refined for BOC-containing regimens.
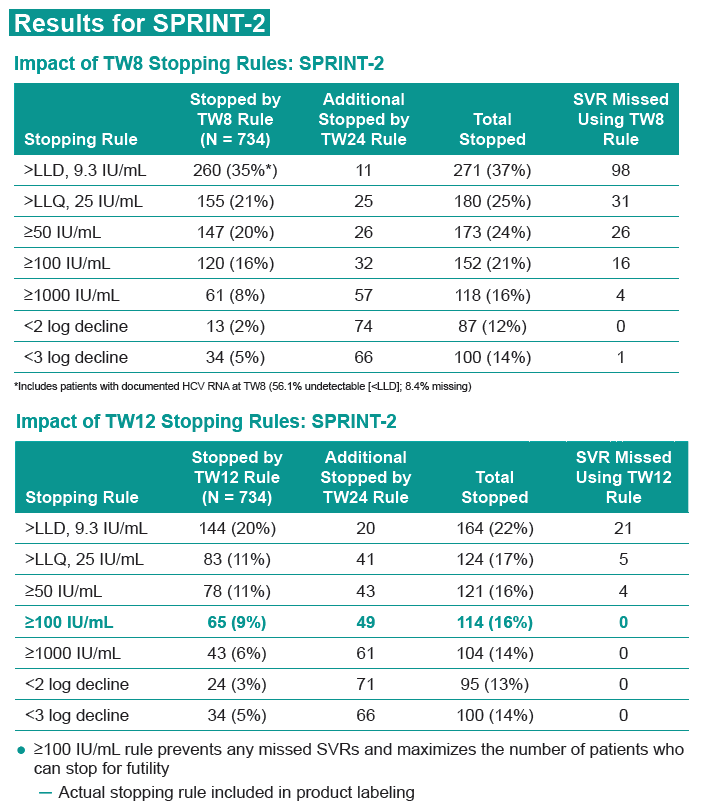
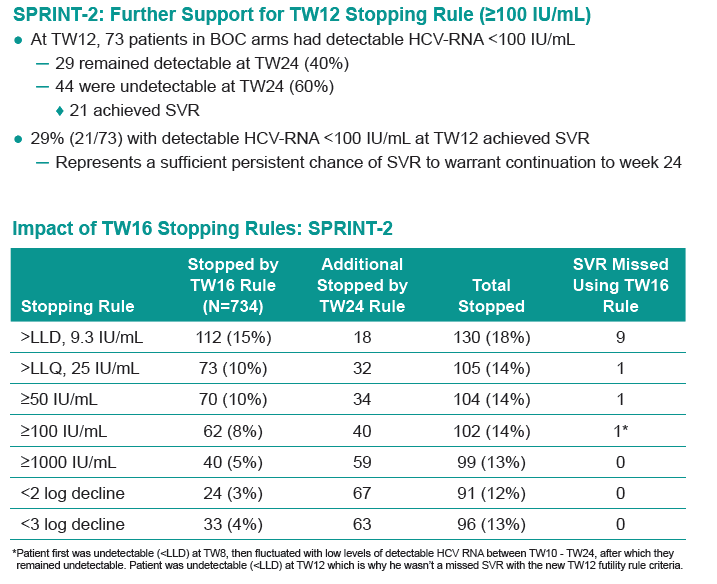
RESULTS: For SPRINT-2 BOC arms, earliest 100% negative predictive time point for SVR while sparing largest number patients from continued failing therapy was HCV-RNA ≥100 IU/ml at TW12 (65 [9%] patients, table): 4 HCV-RNA undetectable by TW24 but failed SVR, 30 detectable at TW24 (29 discontinued for treatment failure, 1 completed treatment [LLQ at TW24]), and 31 discontinued by TW24 (21 discontinued for treatment failure, 3 AE, 4 protocol non-compliance, 3 withdrew consent). 29 patients with detectable HCV-RNA <100 IU/ml at TW12 remained detectable at TW24. 44 with detectable HCV-RNA <100 IU/ml at TW12 were undetectable at TW24; of these 21 had SVR. In RESPOND-2, 39/323 BOC patients had detectable HCV-RNA <100 IU/ ml at TW12; of these 31 were detectable but ≤25 IU/ mL (lower limit of quantification [LLQ]) of assay, 6 continued therapy, and 5 had SVR.
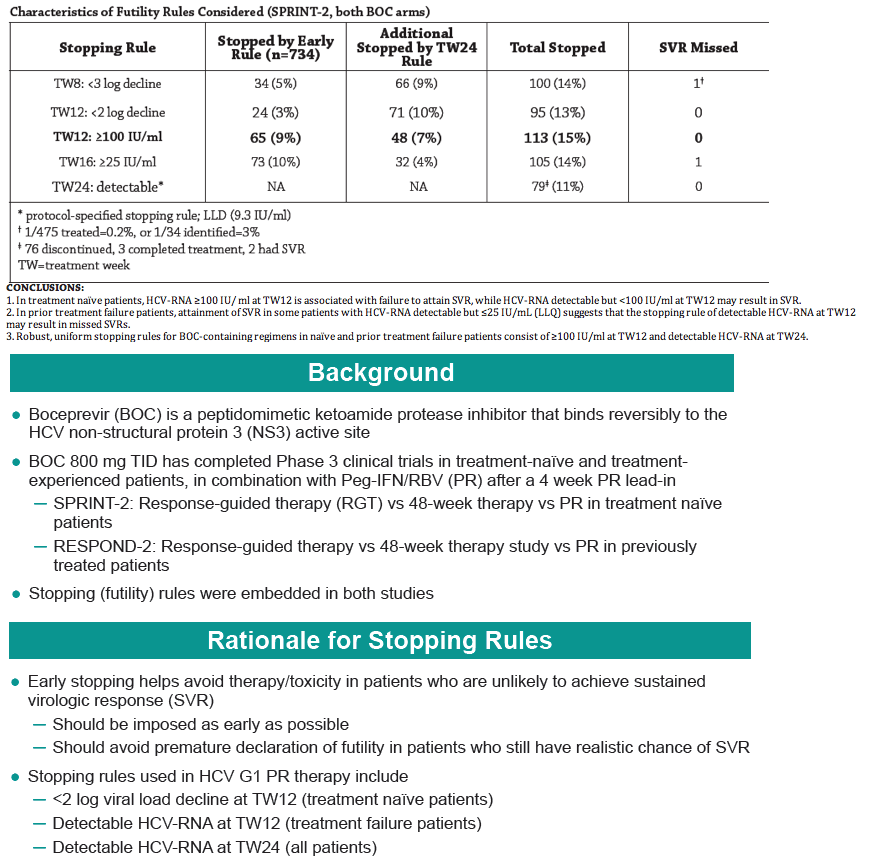
CONCLUSIONS:
1. In treatment naïve patients, HCV-RNA ≥100 IU/ ml at TW12 is associated with failure to attain SVR, while HCV-RNA detectable but <100 IU/ml at TW12 may result in SVR.
2. In prior treatment failure patients, attainment of SVR in some patients with HCV-RNA detectable but ≤25 IU/mL (LLQ) suggests that the stopping rule of detectable HCV-RNA at TW12 may result in missed SVRs.
3. Robust, uniform stopping rules for BOC-containing regimens in naïve and prior treatment failure patients consist of ≥100 IU/ml at TW12 and detectable HCV-RNA at TW24.


|
| |
|
 |
 |
|
|