 |
 |
 |
| |
Pharmacokinetic Evaluation of the Interaction Between the HCV Protease Inhibitor Boceprevir and the
HMG-CoA Reductase Inhibitors Atorvastatin and Pravastatin
|
| |
| |
Reported by Jules Levin
HEP DART, December 4-8, 2011, Koloa, Hawaii
EGJ Hulskotte,1 S Gupta,2* F Xuan,2 MGJA van Zutven,1 E O'Mara,2* L Galitz,3 JA Wagner,2 JR Butterton2
1MSD, The Netherlands; 2Merck Sharp & Dohme Corp., Whitehouse Station, NJ, USA; 3Cetero Research, Miami, FL, USA
*Former employee of Merck Sharp & Dohme Corp., Whitehouse Station, NJ, USA
ABSTRACT
Background: Boceprevir is a potent, orally administered, ketoamide inhibitor targeting the active site of the NS3
protease. It has recently been approved for the treatment of chronic hepatitis C genotype 1 infection in both the
United States and the European Union. Patients with hepatitis C often also suffer from hyperlipidemia. Atorvastatin and pravastatin are among the most commonly prescribed statins used to lower high cholesterol levels and reduce the risk of heart attack and stroke. Atorvastatin is metabolized extensively by CYP3A4, is a substrate of the OATP1B1 transporter, and is possibly also a substrate of P-gp. As boceprevir is an inhibitor of both CYP3A4 and OATP1B1, co-administration may increase the plasma concentrations of atorvastatin. Pravastatin is not metabolized to a significant extent by the CYP450 system. Pravastatin is a substrate of OATP1B1 and OATP2B1, but not of P-gp. An effect of boceprevir on pravastatin via these transporters cannot be excluded.
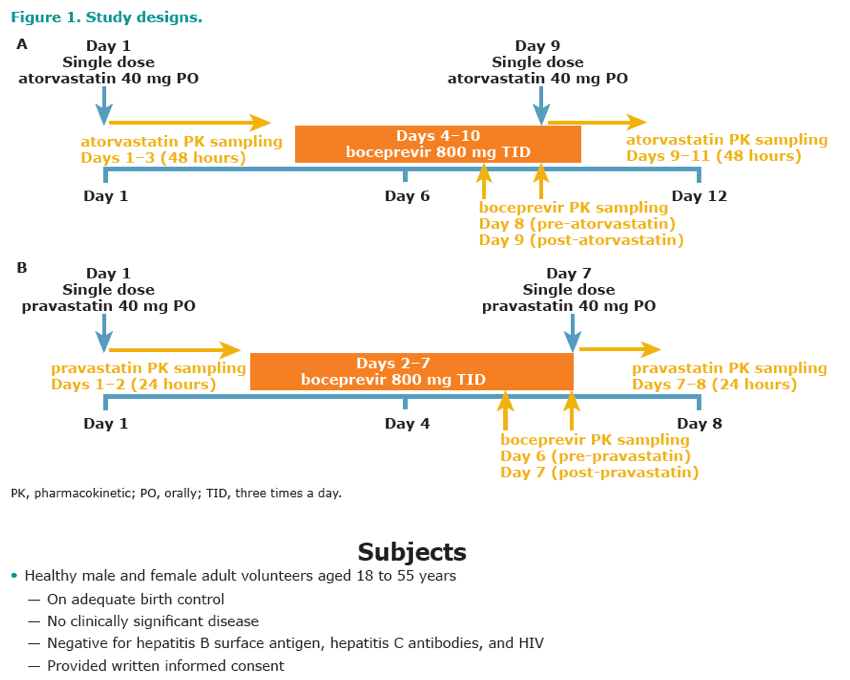
Methods: This study was an open-label, fixed-sequence study in healthy volunteers. Ten subjects received a single 40-mg dose of atorvastatin followed, after a washout of 3 days, by 800 mg boceprevir TID for 7 days. On day 6, subjects concomitantly received a single 40-mg dose of atorvastatin. Ten different subjects received a single 40-mg dose of pravastatin; 24 hours later, they received 800 mg boceprevir TID for 6 days. On day 6, subjects concomitantly received a single 40-mg dose of pravastatin. Blood samples were collected for determination of pharmacokinetics of atorvastatin, pravastatin, and boceprevir. Safety assessments included electrocardiograms, vital signs, clinical laboratory tests, physical examination, and adverse event monitoring.
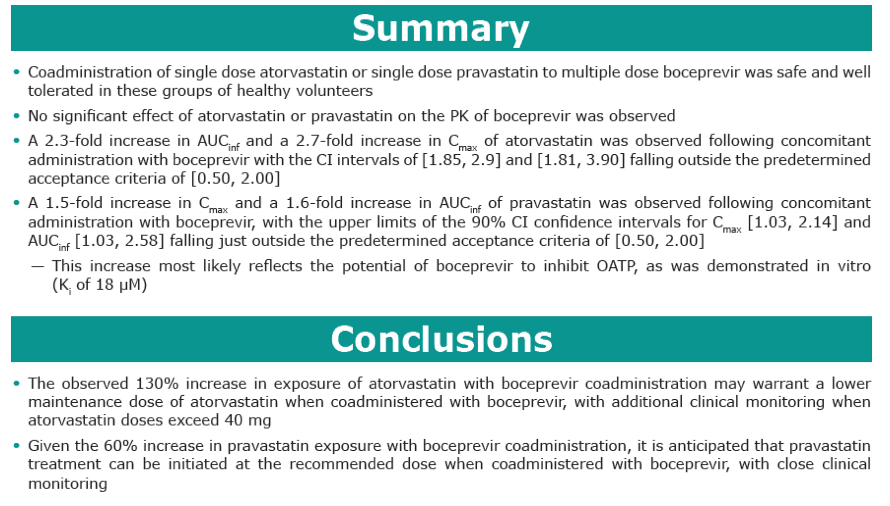
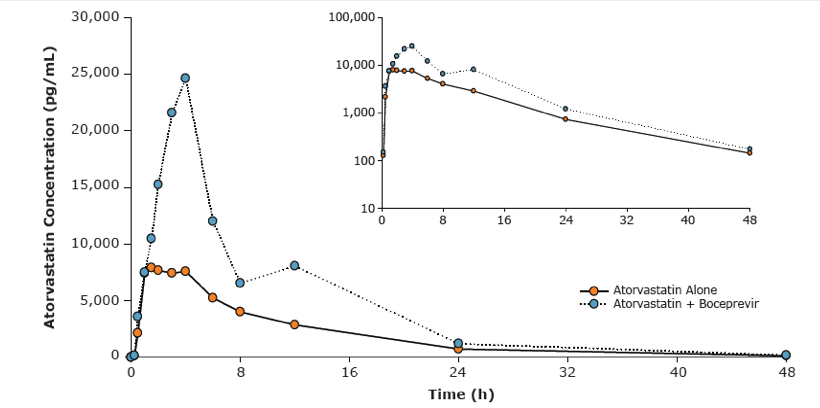
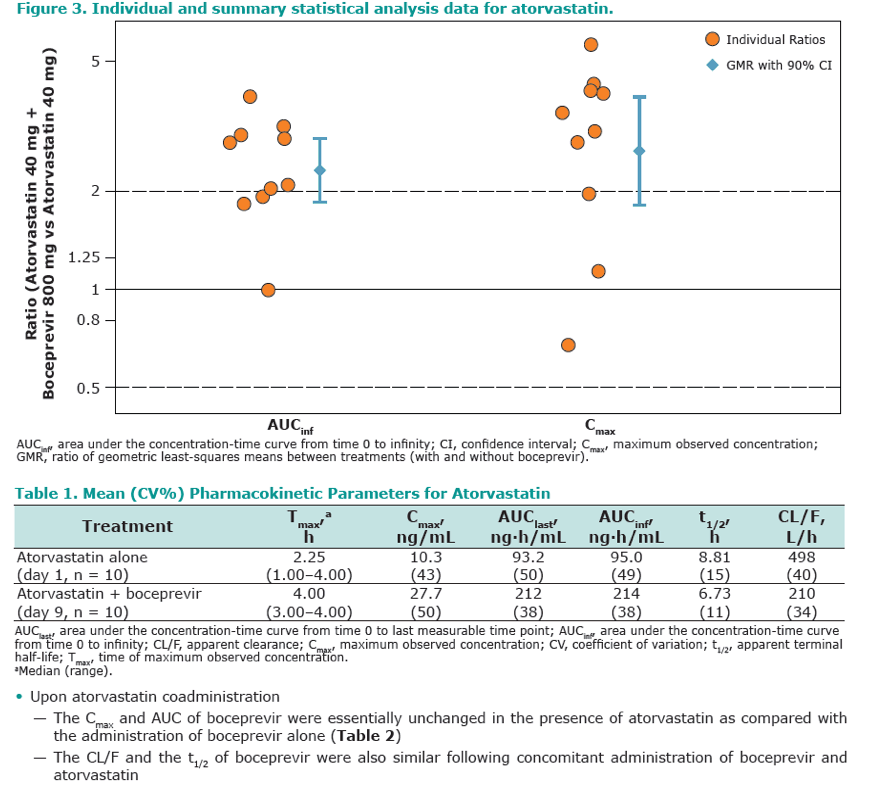
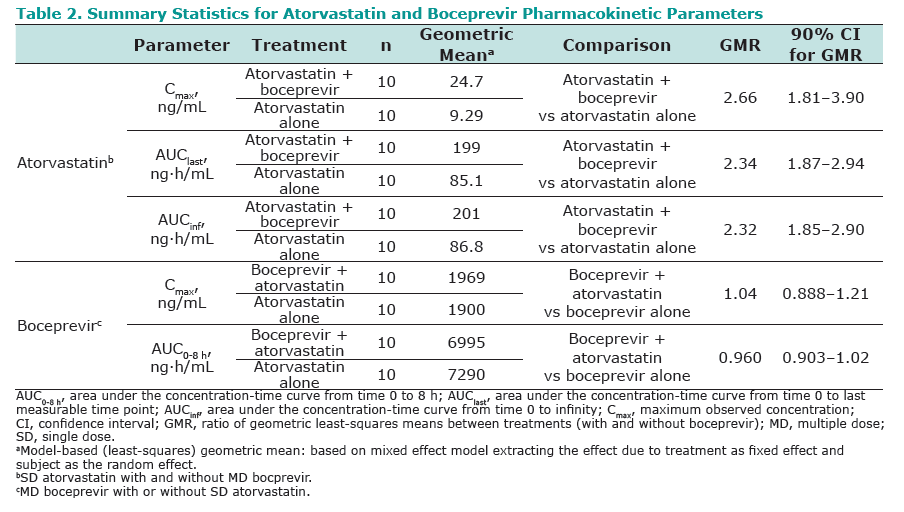
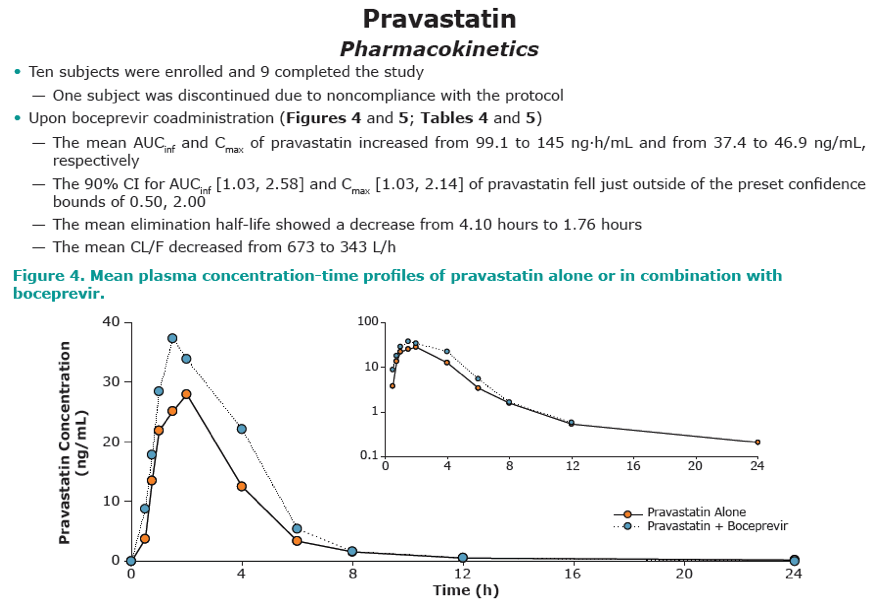
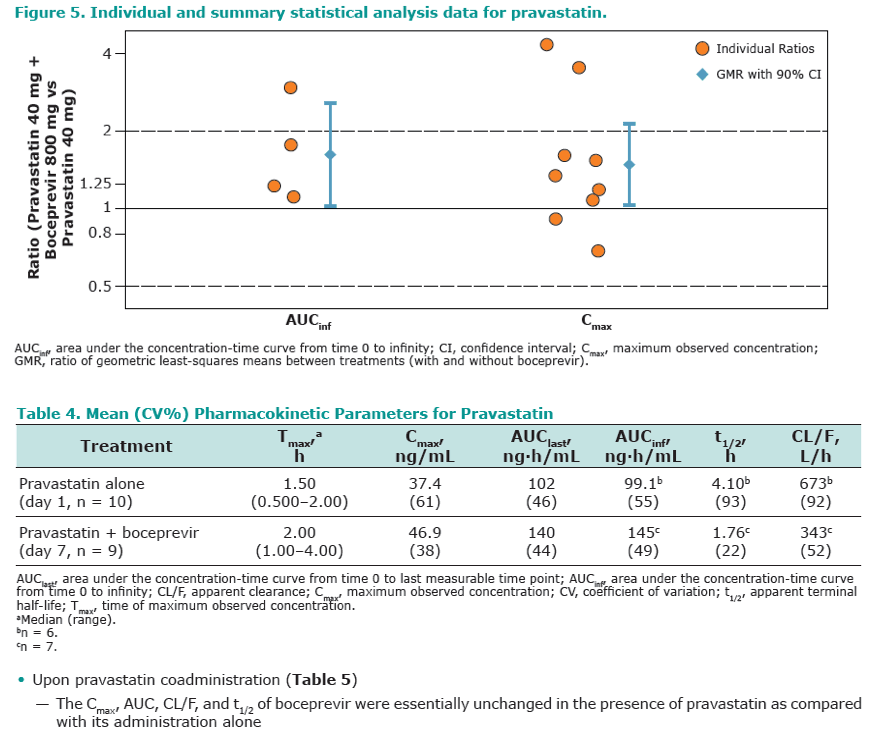
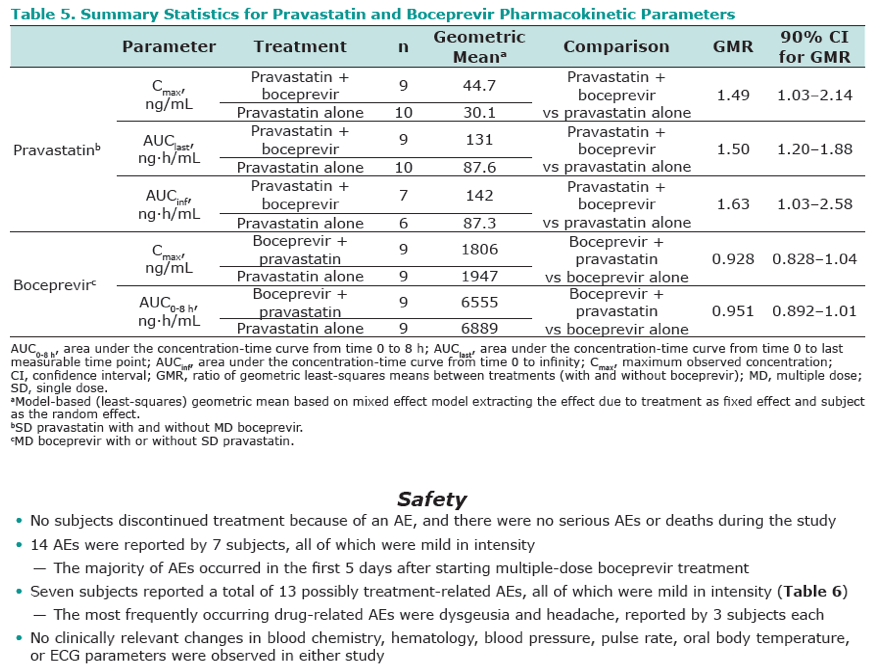
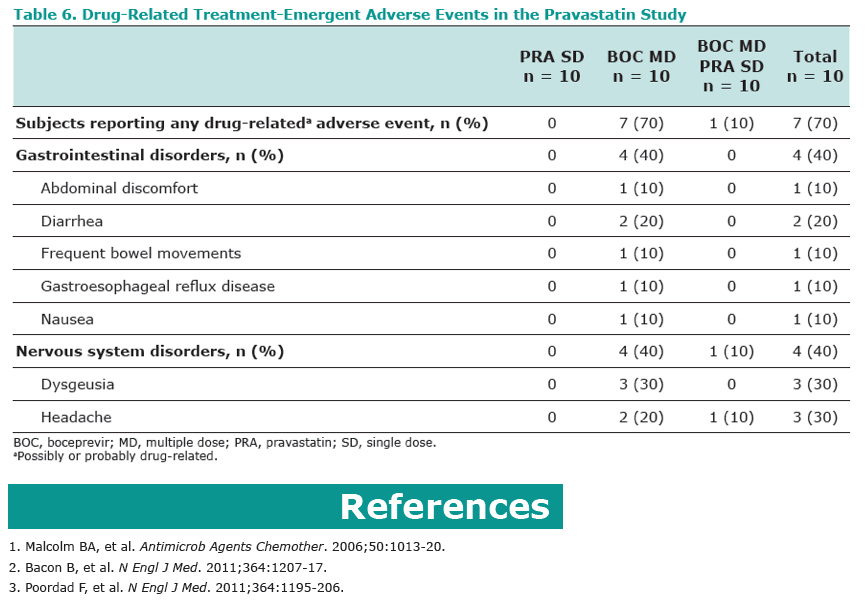
Results: Co-administration of boceprevir with atorvastatin or pravastatin was safe and well tolerated in these groups of healthy volunteers. In the atorvastatin group all enrolled subjects completed the trial, in the pravastatin group one subject discontinued due to noncompliance. In the presence of boceprevir, atorvastatin AUCinf and Cmax increased with geometric mean ratios (GMR)[90% CI] of 2.3 [1.9, 2.9] and 2.7 [1.8, 3.9], respectively. Concomitant boceprevir administration increased the AUCinf and Cmax of pravastatin with GMRs [90% CI] of 1.6 [1.0, 2.6] and 1.5 [1.0, 2.1], respectively. This slight increase may reflect the potential of boceprevir to inhibit OATP, as was demonstrated in vitro (Ki of 18 μM). Neither atorvastatin nor pravastatin had a meaningful effect on the pharmacokinetics of boceprevir.
Conclusion: The observed 130% increase in exposure of atorvastatin with boceprevirco-administration may warrant a lower maintenance dose of atorvastatin when co-administered with boceprevir, with additional clinical monitoring when atorvastatin doses exceed 40 mg. Given the minimal increase of 60% in pravastatin exposure with boceprevir co-administration, it is anticipated that pravastatin treatment can be initiated at the recommended dose when co-administered with boceprevir, with close clinical monitoring.



|
| |
|
 |
 |
|
|