 |
 |
 |
| |
REAL-LIFE SAFETY OF TELAPREVIR OR BOCEPREVIR IN COMBINATION WITH PEGINTERFERON ALFA/RIBAVIRIN, IN CIRRHOTIC NON RESPONDERS. FIRST RESULTS OF THE FRENCH EARLY ACCESS PROGRAM (ANRS CO20-CUPIC)
|
| |
| |
Reported by Jules Levin
HepDart Dec 408 2011 Hawaii
C Hezode1, C Dorival2, F Zoulim3, V de Ledinghen4, T Poynard5, P Mathurin6, D Larrey7, M Bourliere8, S Pol9, P Cacoub5, PH Bernard10, D Lucidarme11, Y Barthe2, H Fontaine9, F Carrat2, JP. Bronowicki12 pour le groupe CUPIC (ANRS CO 20)
Hopital Henri Mondor, Creteil1, UMR-S 707, Paris2, INSERM U871, Lyon3, Hopital Haut-Leveque, Pessac4, Hopital de la Pitie-Salpetriere, Paris5, Hopital Claude Huriez, Lille6, Hopital Saint-Eloi, Montpellier7, Fondation Hopital Saint Joseph, Marseille8, Hopital Cochin, Paris9, Hopital Saint Andre, Bordeaux10, Hopital Saint Philibert, Lomme11, Hopital de Brabois, Nancy12

ABSTRACT: CONCLUSIONS: In this large cohort of cirrhotic patients, the safety profile of TVR or BOC in combination with PEG-IFN and RBV was poor and was associated with increased rates of SAE (40% to 57%) compared to those reported in phase III clinical trials (9% to 14%). These data strongly suggest that triple combination therapy must be administered cautiously with intensive safety monitoring in these patients.
BACKGROUND: Phase III trials have shown that Telaprevir (TVR) administration is associated with frequent dermatological side effects (pruritus, rash), and anemia, whereas Boceprevir (BOC) administration is associated with frequent anemia. However, few patients with cirrhosis were included in these trials. We present here the first report of the safety profile of TVR and BOC, used in combination with peginterferon (PEG-IFN) and ribavirin (RBV) in cirrhotic patients infected with HCV genotype 1, relapsers or partial-responders during a first course of standard-of-care therapy included in the French Early Access Program for the use of protease inhibitors.
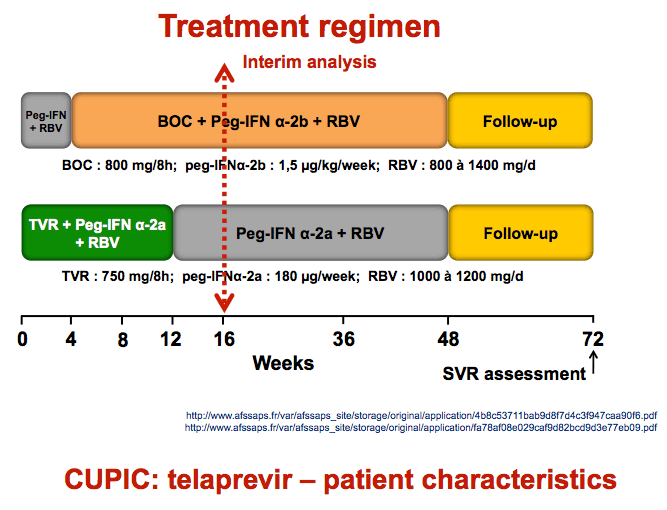
METHODS: In the cohort, 359 patients with HCV genotype 1 infection and compensated cirrhosis (Child A) were prospectively included. They received either 12 weeks of TVR, PEG-IFN-alfa2a and RBV followed by 36 weeks of PEG-IFN/RBV, or 4 weeks of PEG-IFN-2b/RBV (lead-in phase) followed by 44 weeks of BOC, PEG-IFN-2b and RBV. The choice of therapy was made by the treating physician and the patients were not randomized to receive either treatment. Thus, the following results for the two molecules stand for themselves and cannot be compared. Safety analysis is restricted to patients with at least 8 weeks of follow-up
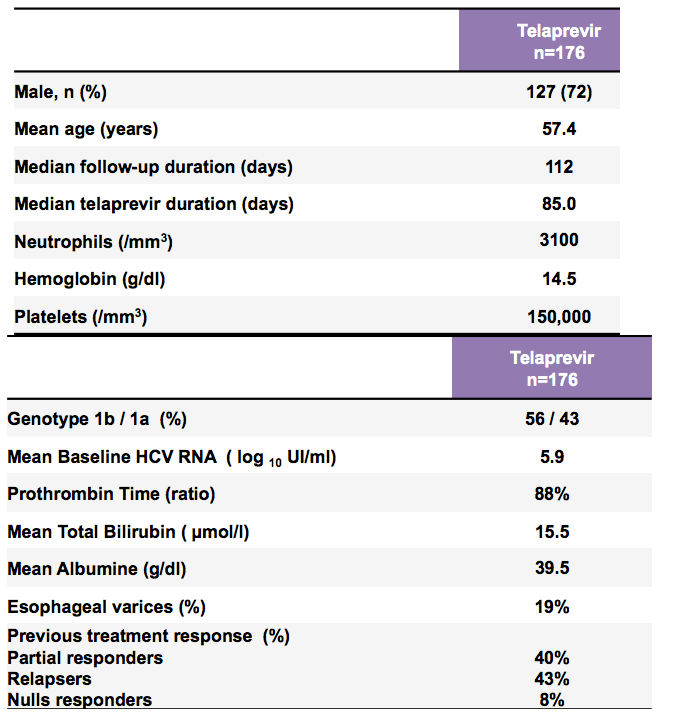
RESULTS: Patients treated with TVR included 81 men (76%) and 26 women (24%) with a mean age of 57.0, The frequencies of serious adverse events, laboratory abnormalities and events of special interest are summarized in the table below:
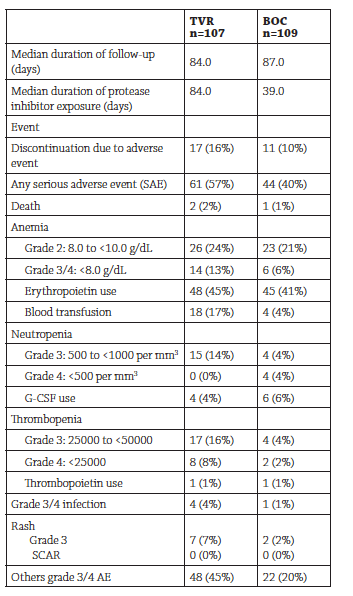


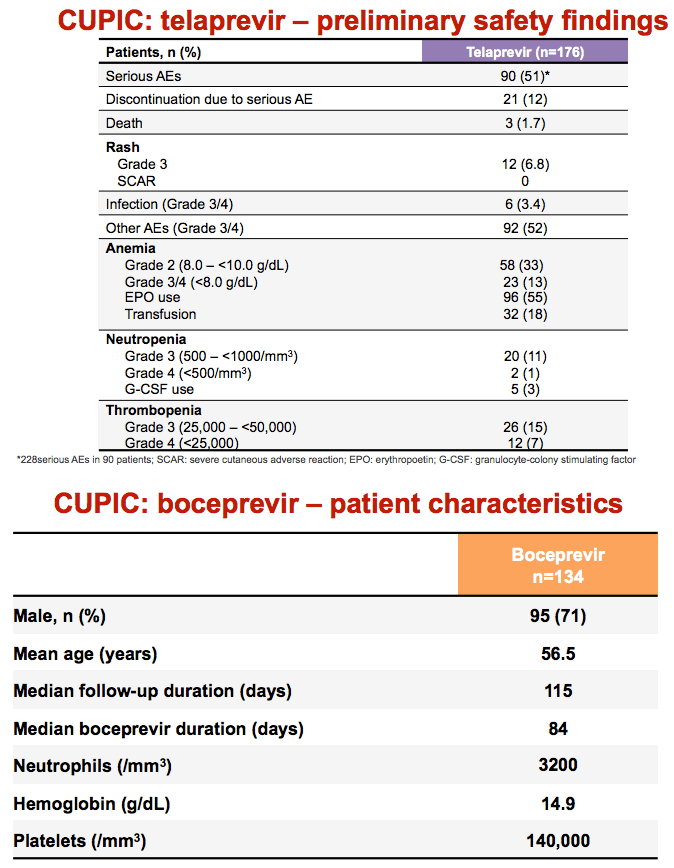
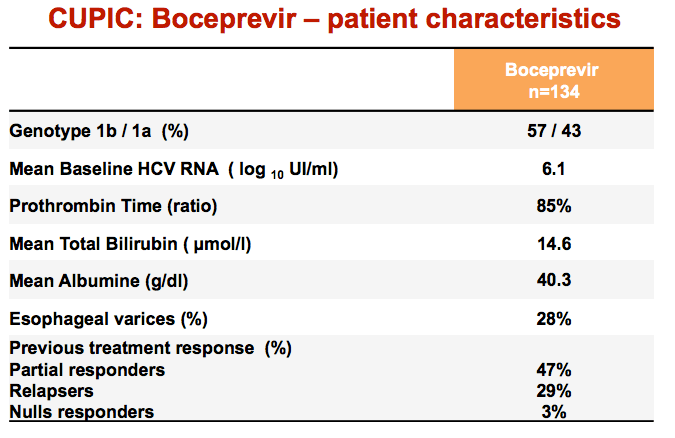
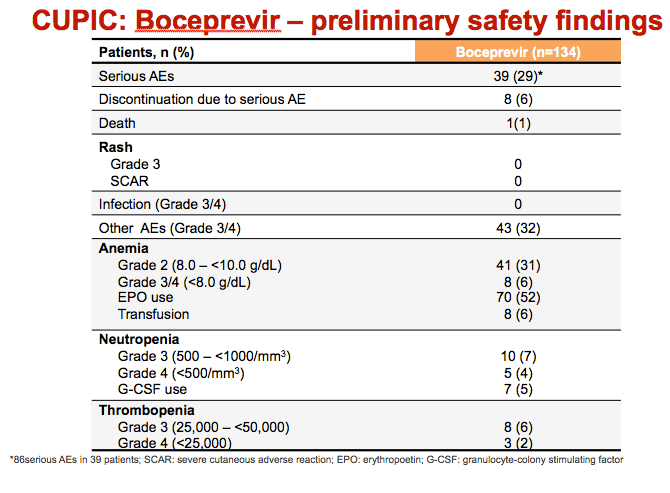
|
| |
|
 |
 |
|
|