 |
 |
 |
| |
Safety and Sustained Viral Response of MK-5172 for 12 Weeks in Combination With Pegylated Interferon
Alfa-2b and Ribavirin for 24 Weeks in HCV Genotype 1 Treatment-Naive Noncirrhotic Patients
|
| |
| |
Reported by Jules Levin
AASLD Nov 9-13 2012 Boston
Patrick Marcellin,1 John M Vierling,2 Bruce R Bacon,3 Michael Manns,4 Christine Fandozzi,5 Jacqueline Gress,5 Luzelena Caro,5 Christopher Gilbert,5 Peggy Hwang,5 Janice Wahl,5 Michael P Cooreman,5 Niloufar Mobashery5
1University Paris-Diderot, Clichy, France; 2Baylor College of Medicine, Houston, Texas, USA; 3Saint Louis University, St. Louis, Missouri, USA; 4Hannover Medical School, Hannover, Germany;
5Merck Sharp & Dohme Corp., Whitehouse Station, New Jersey, USA

BACKGROUND
MK-5172 is a once-daily, potent, next-generation NS3/4A protease inhibitor (PI)
-- Potent in vitro activity against a broad enzyme panel including all major hepatitis C virus (HCV) genotypes (G)1
-- In vitro activity against resistance-associated variants (RAVs) found in patients who failed therapy with first-generation PIs (boceprevir, telaprevir, and TMC-435)2
-- 5 log10 drop in HCV RNA levels in patients with HCV G1 following monotherapy of ≥30 mg once daily (qd) for 7 days3
AIM
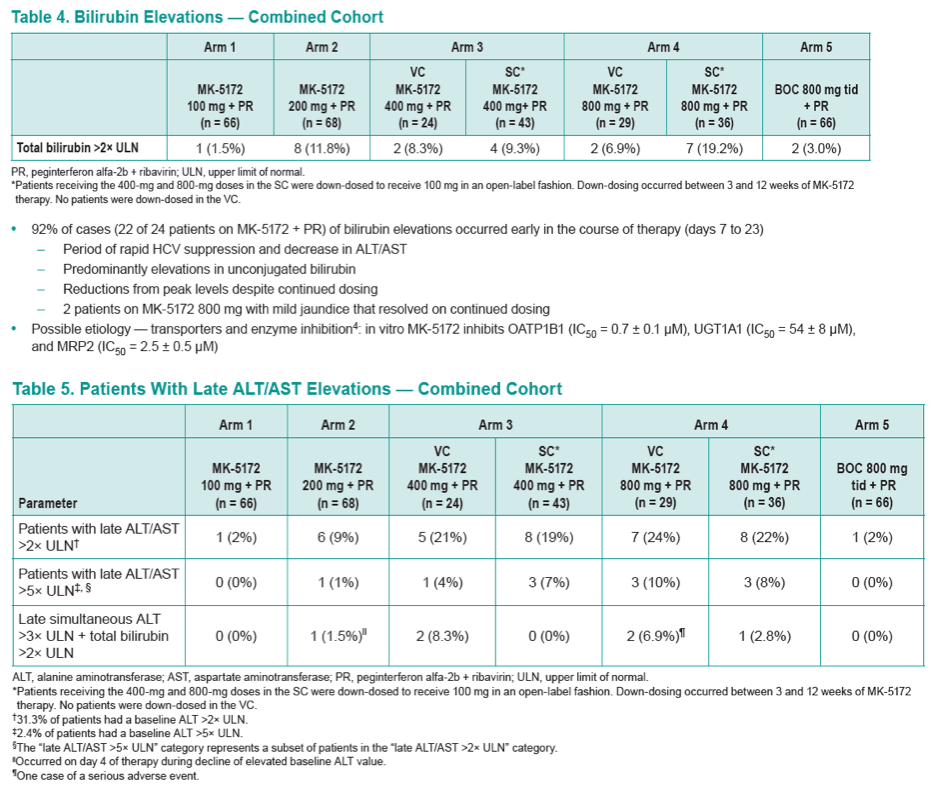
To examine the safety and efficacy of MK-5172 administered with pegylated interferon alfa-2b (1.5 μg/kg/week) and ribavirin (PR) (600-1400 mg/day) in HCV G1 treatment-naive (TN) patients

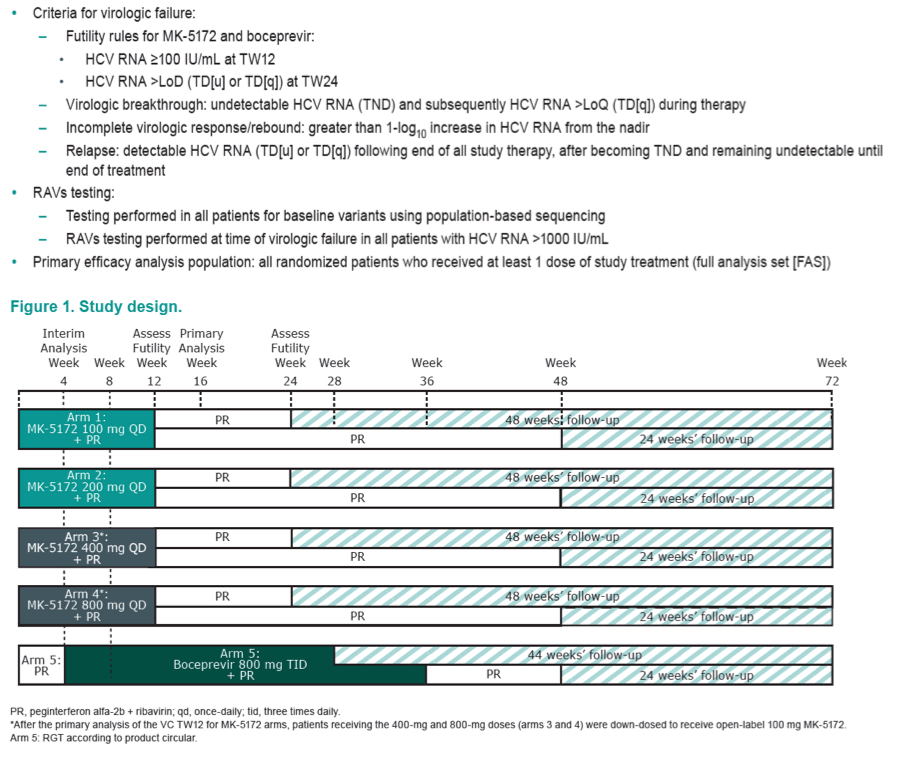

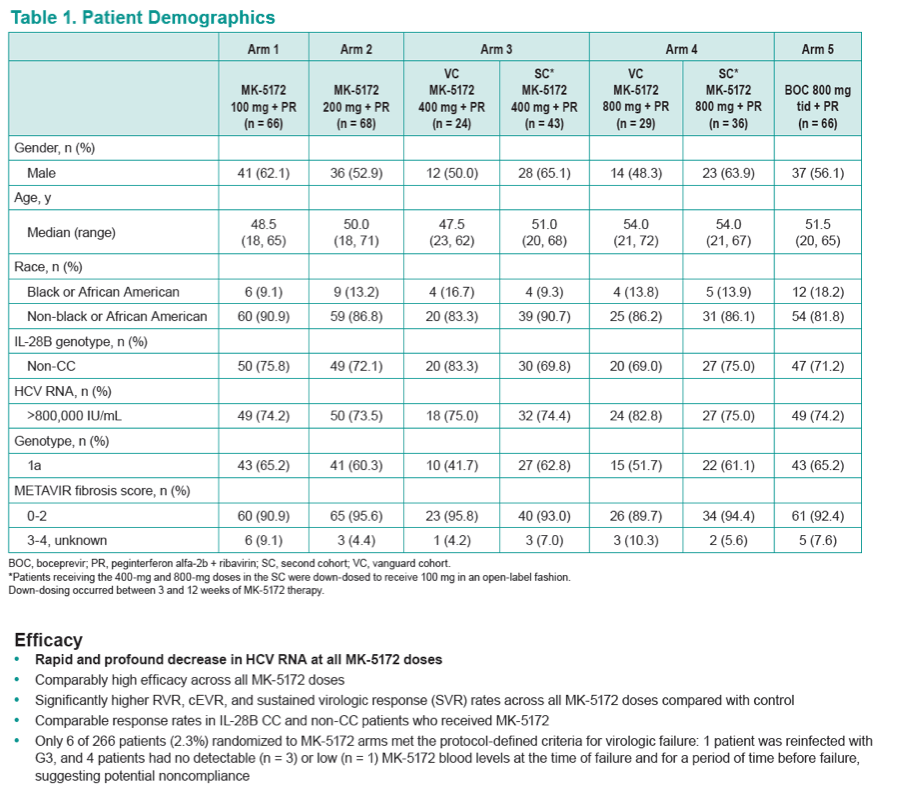
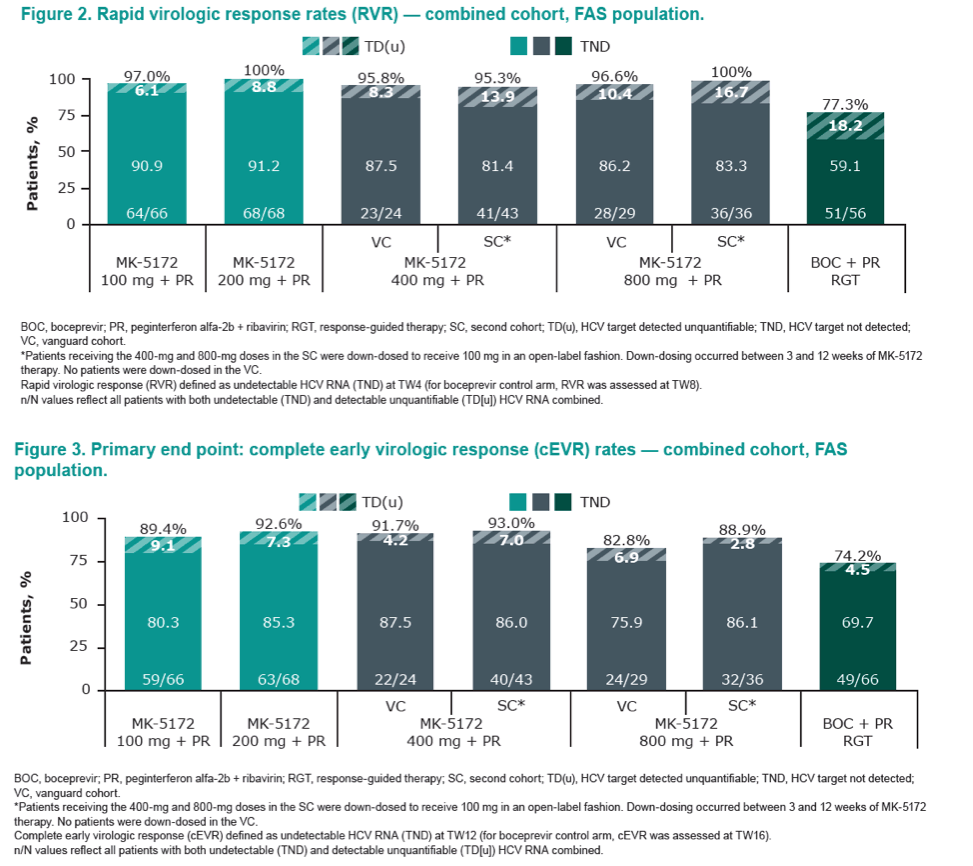
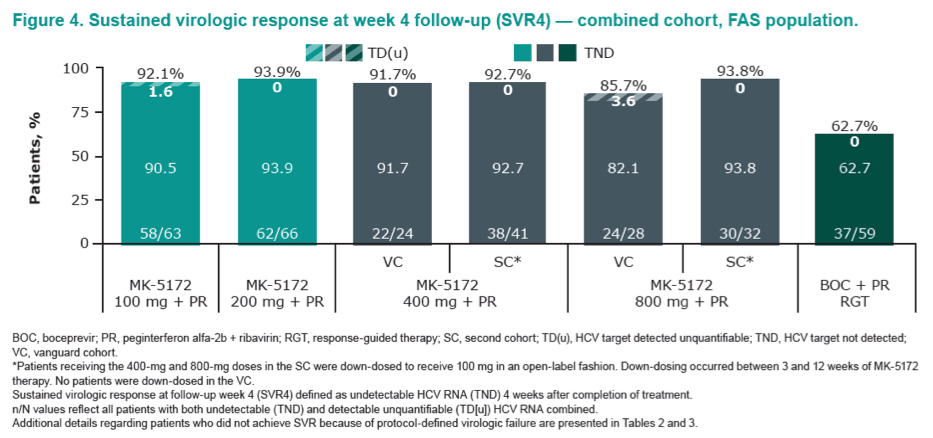
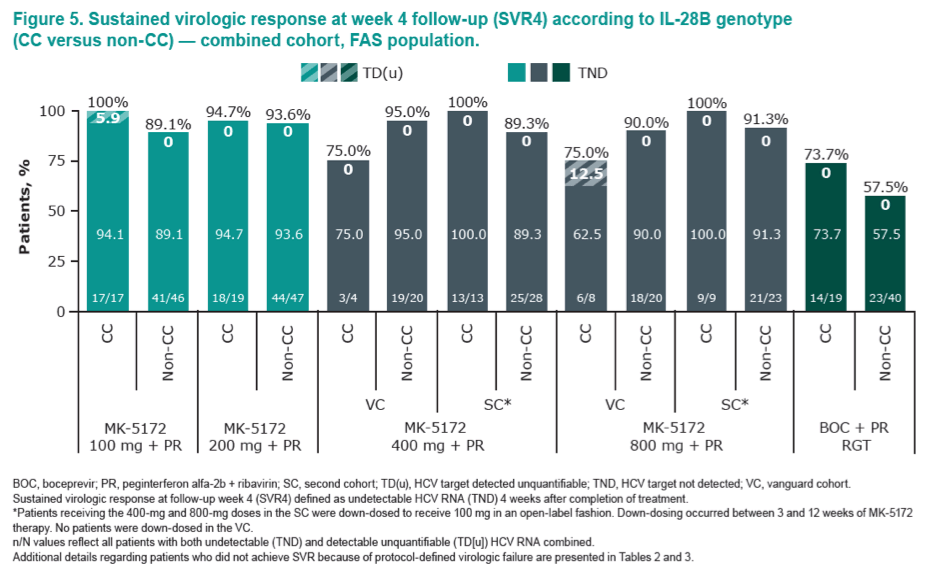
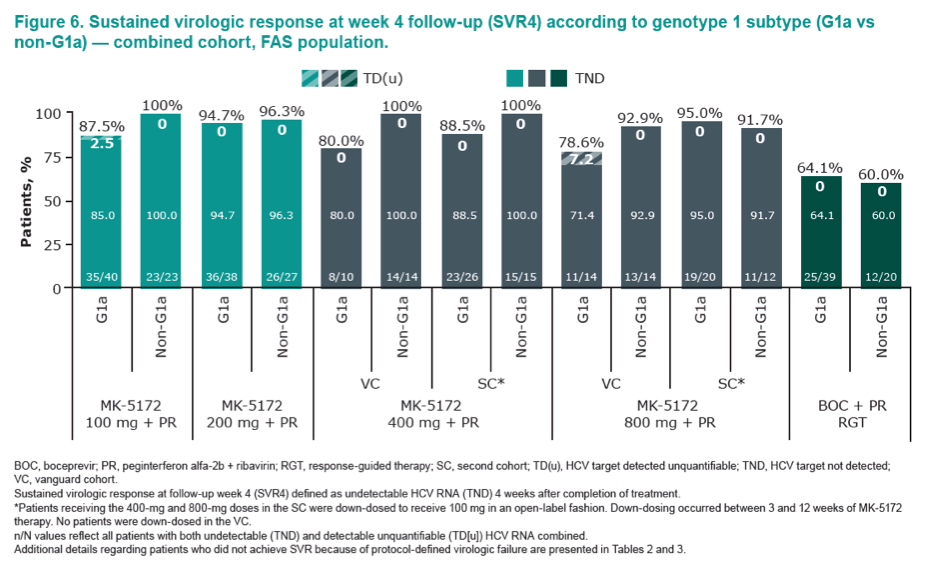
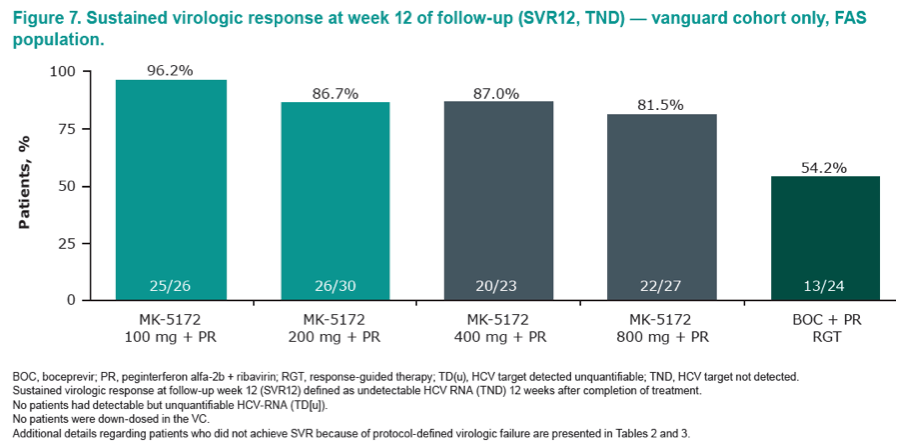
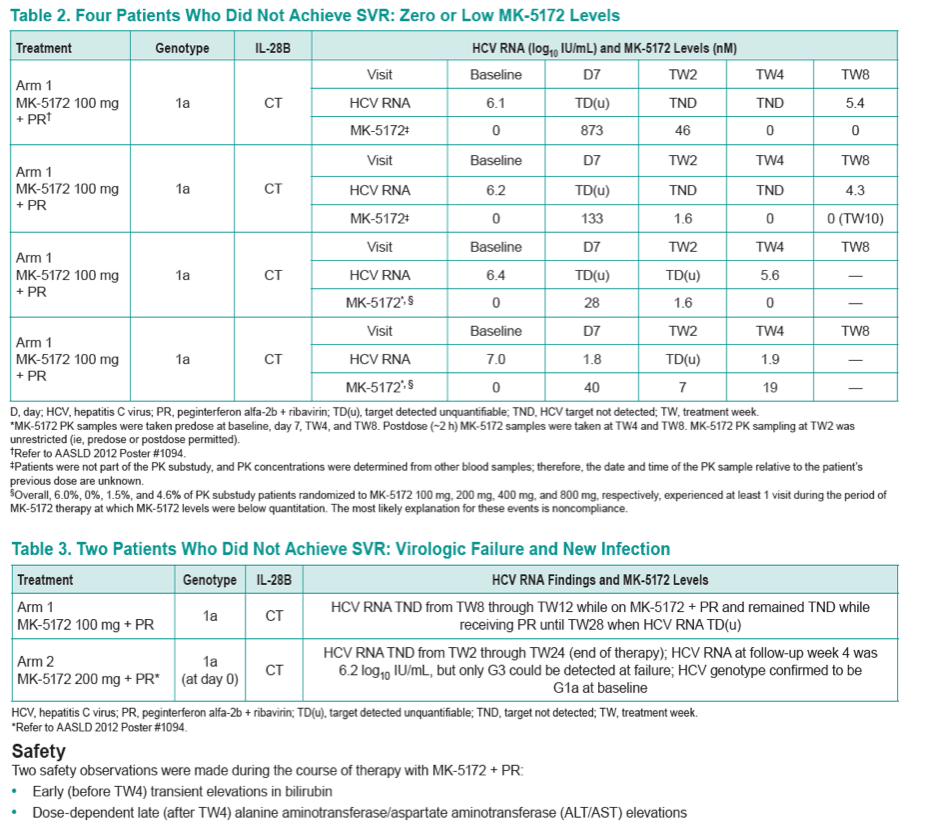

|
| |
|
 |
 |
|
|