 |
 |
 |
| |
Sustained Virological Response (SVR) Rate of 67% in HCV Genotype 1-Infected Prior Null Responders Treated with Danoprevir/Ritonavir (DNVr) in Combination with Peginterferon Alfa-2a (40KD) plus Ribavirin (PegIFN Alfa-2a/RBV)
|
| |
| |
Reported by Jules Levin
AASLD Nov 9-13 2012 Boston
E.J. Gane,1 R. Rouzier,2 A. Wiercinska-Drapalo,3 D.G. Larrey,4 R. Bhardwaj,5 B.J. Brennan,5 M. Giraudon,6 S. Le Pogam,5 I. Nájera,5 R. Petric,5 J.Q. Tran,7 R. Kulkarni,8 Y. Zhang,8 P. Smith,5 E.S. Yetzer,8 N.S. Shulman8
1Auckland Clinical Studies, Auckland, New Zealand; 2Phase I/II Center, CAP Center, Montpellier, France; 3Department of Hepatology and Immunodeficiencies, Warsaw Medical University, Poland; 4Liver Unit, Saint Eloi Hospital, Montpellier, France;
5Hoffmann-La Roche Inc, Nutley, NJ, USA; 6F. Hoffmann-La Roche Ltd, Basel, Switzerland; 7Hoffmann-La Roche Inc, Palo Alto, CA, USA; 8Genentech, South San Francisco, CA, USA

from Jules: Roche has ongoing the Annapurna Study which looks at 4 orals: PI, non-nuc polymerase, nuc, rbv
http://www.natap.org/2012/HCV/090412_01.htm
AASLD: Up to 100% SVR4 rates with ritonavir-boosted danoprevir (DNVr), mericitabine and ribavirin with or without peginterferon alfa-2a (40KD) in HCV genotype 1-infected partial and null responders: results from the MATTERHORN study - (11/13/12) Feld study
AASLD: Safety and efficacy of ritonavir-boosted danoprevir (DNVr), peginterferon alfa-2a (40KD), and ribavirin with or without mericitabine in HCV genotype 1-infected treatment-experienced patients with advanced hepatic fibrosis: the MATTERHORN study - (11/13/12)

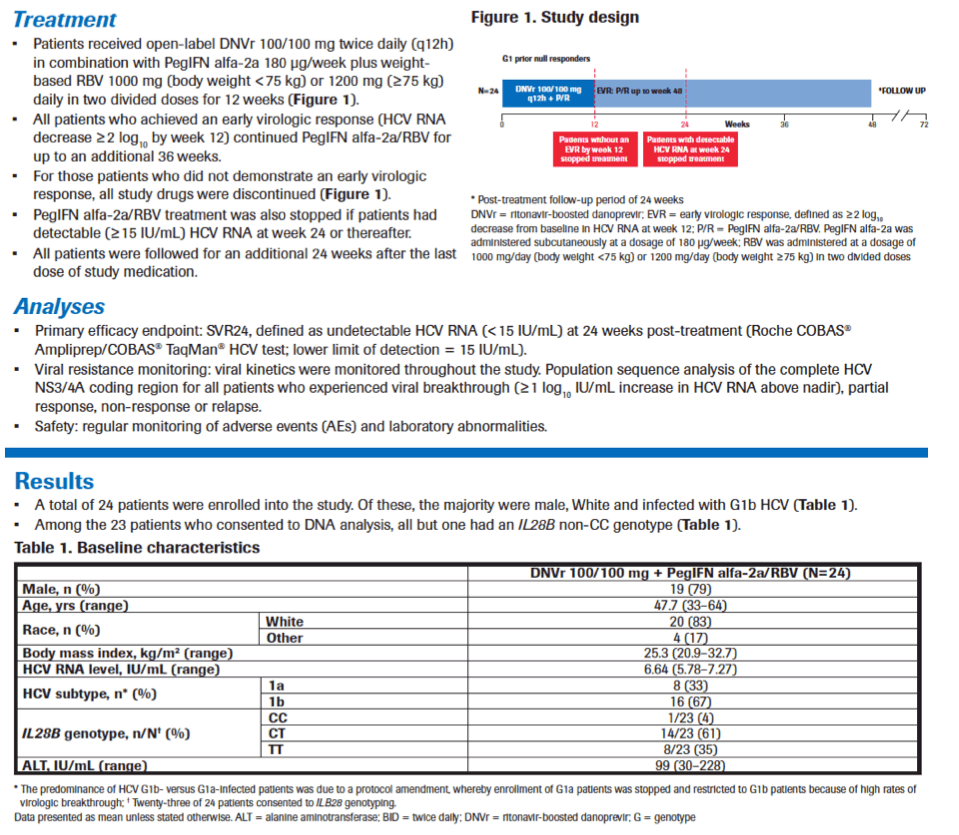
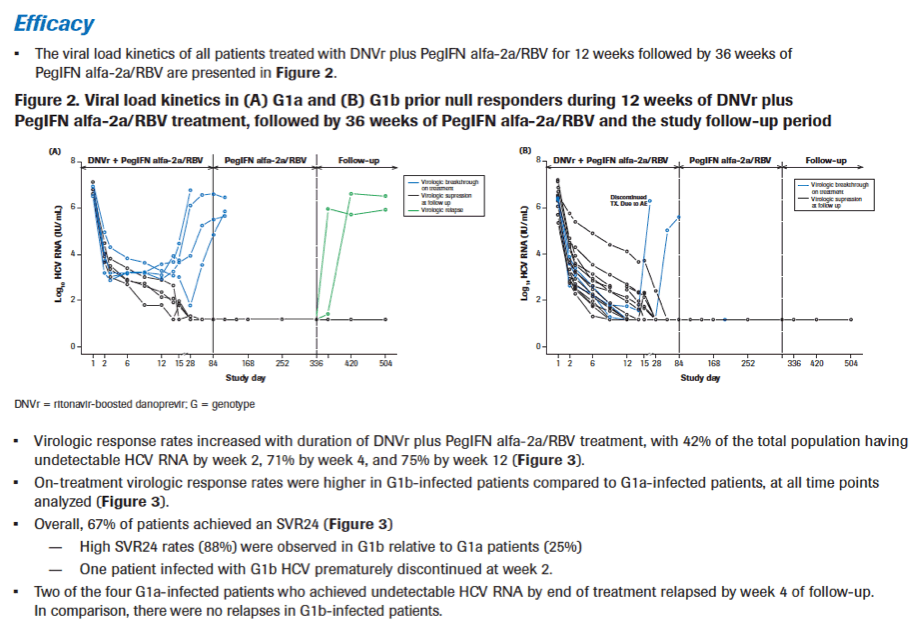
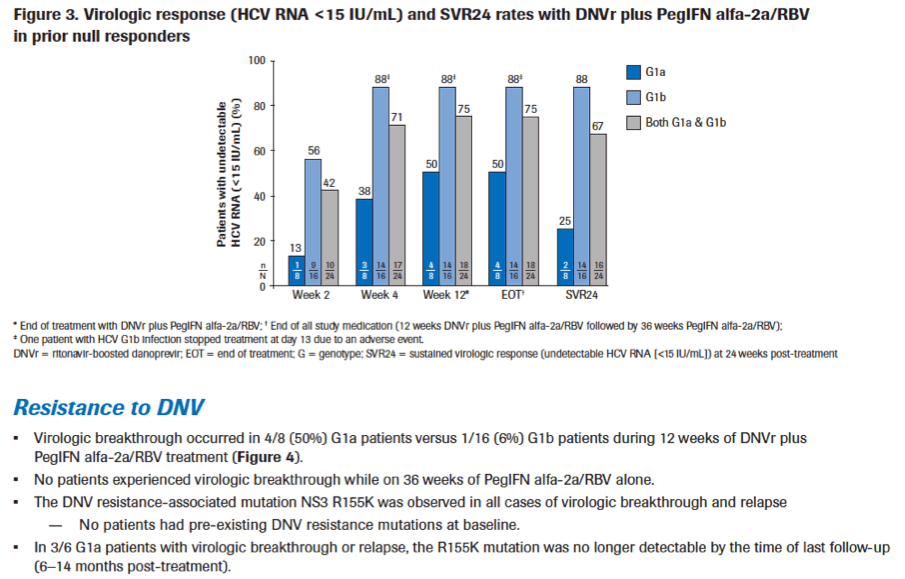
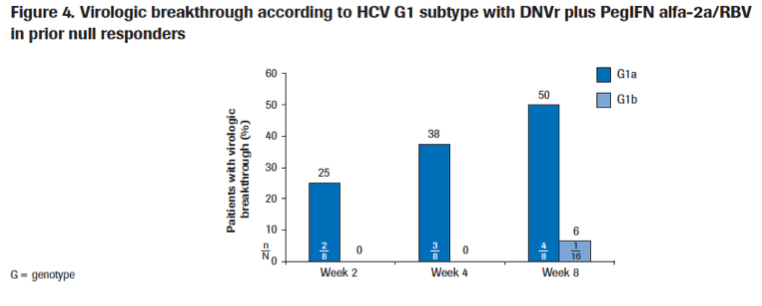
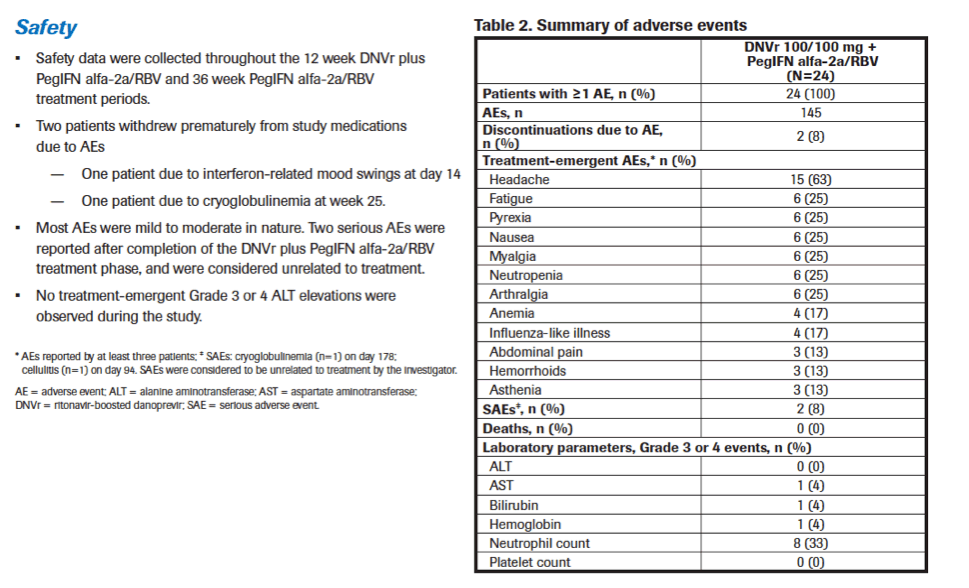

|
| |
|
 |
 |
|
|