 |
 |
 |
| |
GS-7977 & HIV ARTs PK - No Clinically Significant Pharmacokinetic Interactions Between Sofosbuvir (GS-7977) and HIV Antiretrovirals Atripla, Rilpivirine, Darunavir/Ritonavir, or Raltegravir in Healthy Volunteers
|
| |
| |
Reported by Jules Levin
AASLD - 63rd Annual Meeting of the American Association for the Study of Liver Diseases
November 9-13, 2012, Boston, Massachusetts, United States
Brian Kirby, Anita Mathias, Stephen Rossi, Candra Moyer, Gong Shen, Brian P. Kearney
Gilead Sciences, Inc., Foster City, CA
"PK parameters for SOF, GS-566500 (data not shown), GS-331007 (major circulating
metabolite of SOF), FTC, TFV, EFV, DRV, RTV, RAL, and RPV were estimated using
noncompartmental methods (WinNonlin 6.2)
- Geometric least-squares means ratios (GMRs) and 90% confidence intervals (CIs)
were calculated (test/reference) for SOF and metabolites (AUC0-last, AUC0-inf, and Cmax)
and ARVs (AUCtau, Cmax and Ctau) using analysis of variance (ANOVA) with a
lack-of-PK-alteration bounds of 70%-143%"

Program Abstract
Background: Recently approved HCV protease inhibitors (PIs) have drug interaction liabilities with key HIV antiretrovirals (ARVs), specifically, ritonavir-boosted PIs. Sofosbuvir (SOF, formerly GS-7977), a NS5B nucleotide inhibitor, is in Phase 3 clinical development for the treatment of chronic HCV infection. We conducted a study to evaluate potential interactions between SOF and common HIV ARVs of varying 3rd agent classes (NNRTI, PI/r and integrase inhibitor-based).
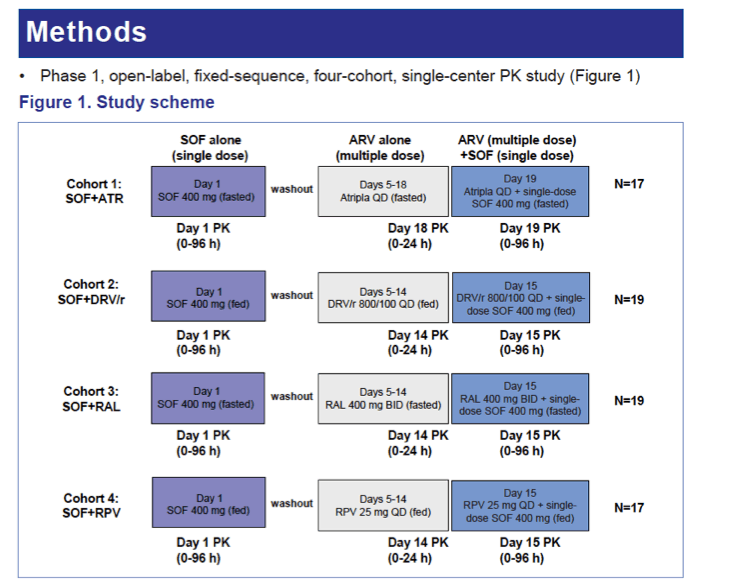
Methods: In a Phase 1, fixed sequence, 4-cohort study, healthy volunteers received a single dose of SOF 400 mg before and after 14 days of Atripla (ATR, EFV/FTC/TDF 600/200/300 mg QD; n=17, fasted), or 10 days of rilpivirine (RPV, 25 mg QD, n=17, fed), darunavir/ritonavir (DRV/r, 800/100 mg QD, n=19, fed), or raltegravir (RAL, 400 mg BID, n=19, fasted). Geometric means ratio % (GMR%, combination vs alone) of PK parameters AUCinf and Cmax for SOF and GS-331007 (circulating nucleoside metabolite of SOF), or AUCtau, Cmax, and Ctau for TFV, FTC, EFV, RPV, DRV/r, or RAL were evaluated against a predetermined 70-143% equivalence boundary. Safety assessments were conducted throughout the study.
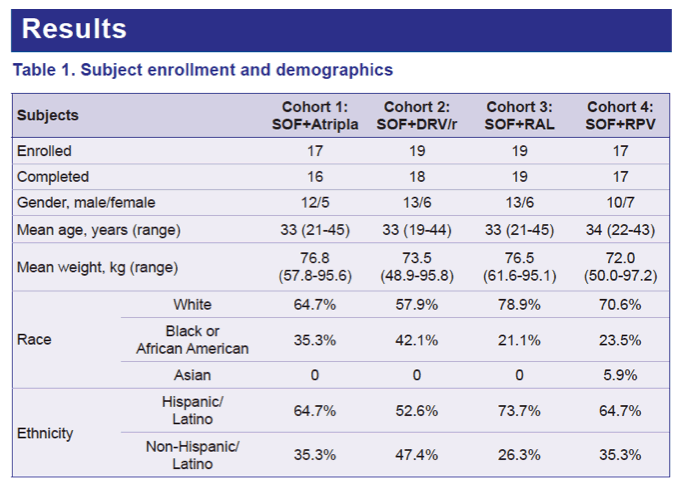
Results: 16 and 17 subjects completed the ATR and RPV cohorts, respectively, and 18 subjects completed the DRV/r and RAL cohorts. The most frequent adverse events were dizziness, headache, diarrhea, nausea and constipation, predominantly during ATR dosing. All but 1 event were mild and most were consistent with known EFV adverse events. Overall, SOF was well tolerated with very few treatment-emergent events during the administration of SOF with ARVs.
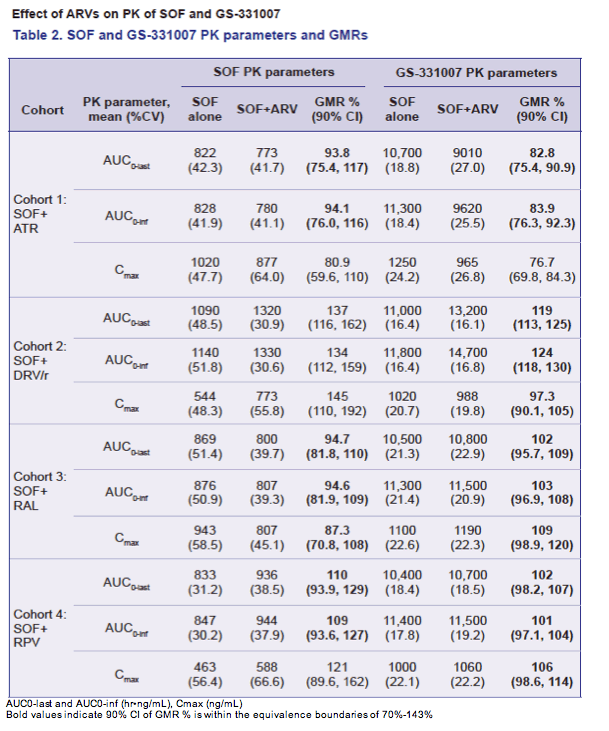
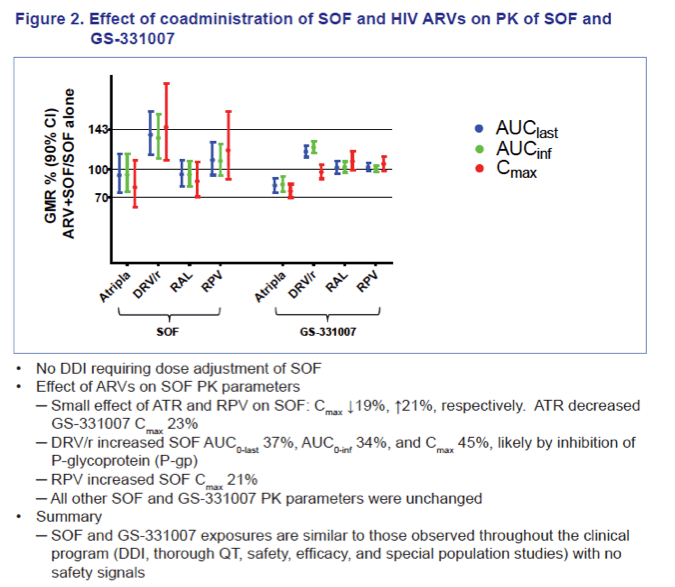
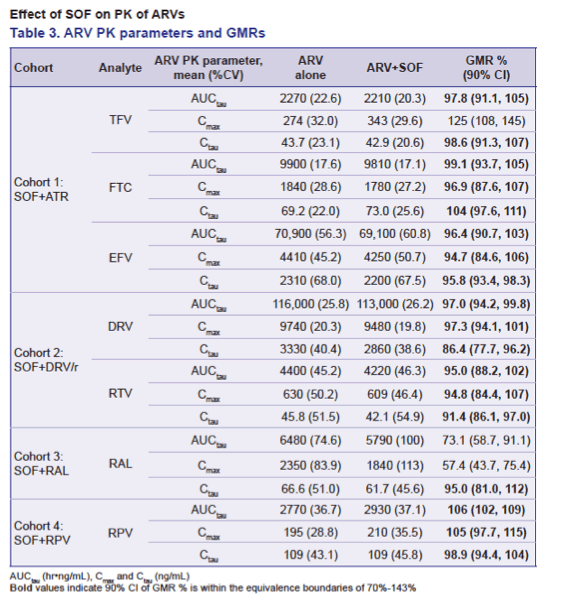
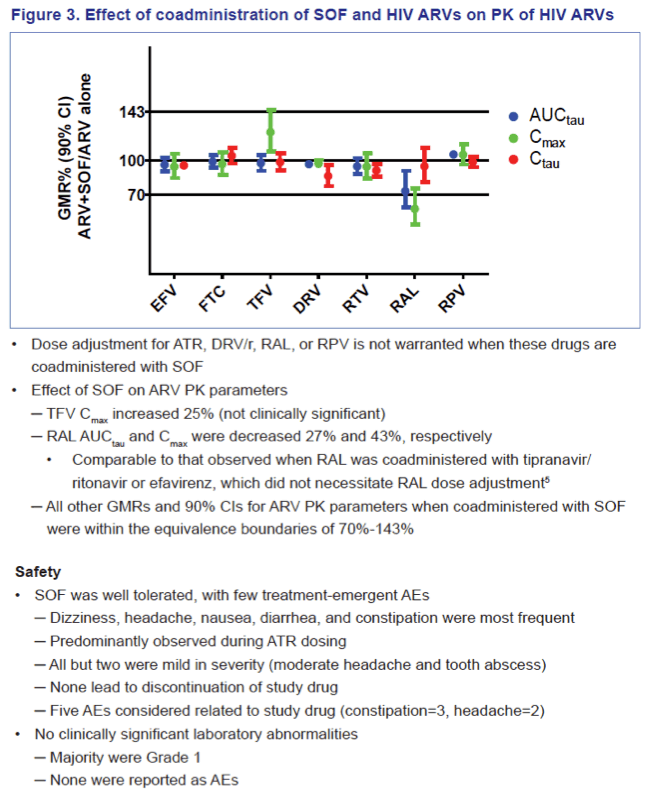
Co-administration of HIV ARVs and SOF did not result in clinically significant changes in SOF and/or GS-331007 exposure. ATR slightly decreased SOF and GS-331007 Cmax (~20-23%), RPV, DRV/r, and RAL modestly increased SOF exposure by 21 to 45% with no effect on GS-331007. SOF slightly increased TFV Cmax (~25%), modestly decreased RAL AUCtau (~27%) and Cmax (~43%), but did not affect RAL Ctau, and had no other effect on PK parameters of FTC, TFV, EFV, RPV, or DRV/r.
Conclusions: No clinically significant DDIs were observed between SOF and EFV, RPV, DRV/r, RAL or the standard-of-care backbone of FTC/TDF. These data support potential use of SOF with a variety of ARV regimens in the HIV/HCV co-infected population.


|
| |
|
 |
 |
|
|