 |
 |
 |
| |
Effect of Hepatic Impairment on the Pharmacokinetics of Asunaprevir (BMS-650032, ASV)
|
| |
| |
Reported by Jules Levin
AASLD 2012 Nov 9-12 Boston MA
Eley T,1 He B,1 Li J,1 Colston E,1 Child M,1 Bedford BR,1 Kandoussi H,1 Wind-Rotolo M,1
Pasquinelli C,1 DeMicco MP,2 Marbury TC,3 Bertz RJ1
1Bristol-Myers Squibb Research and Development, Princeton, NJ; 2Advanced Clinical Research Institute, Anaheim, CA; 3Orlando Clinical Research Center, Orlando, FL
AASLD: Phase 2b Study of the Interferon-Free and Ribavirin-Free Combination of Daclatasvir, Asunaprevir, and BMS-791325 for 12 Weeks in Treatment-Naive Patients With Chronic HCV Genotype 1 Infection - (11/05/13)
AASLD: BMS at AASLD - (11/18/13)

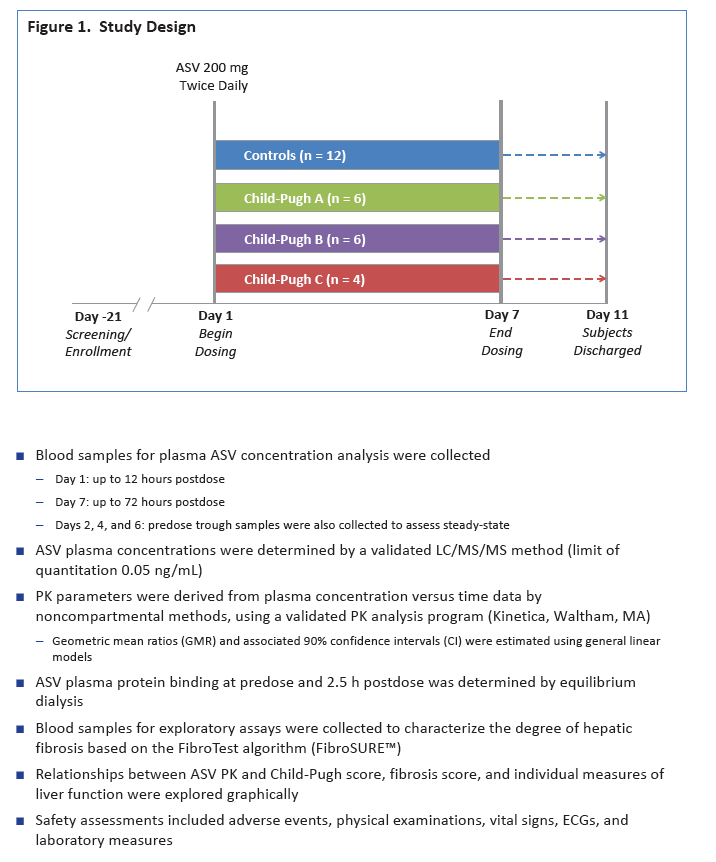
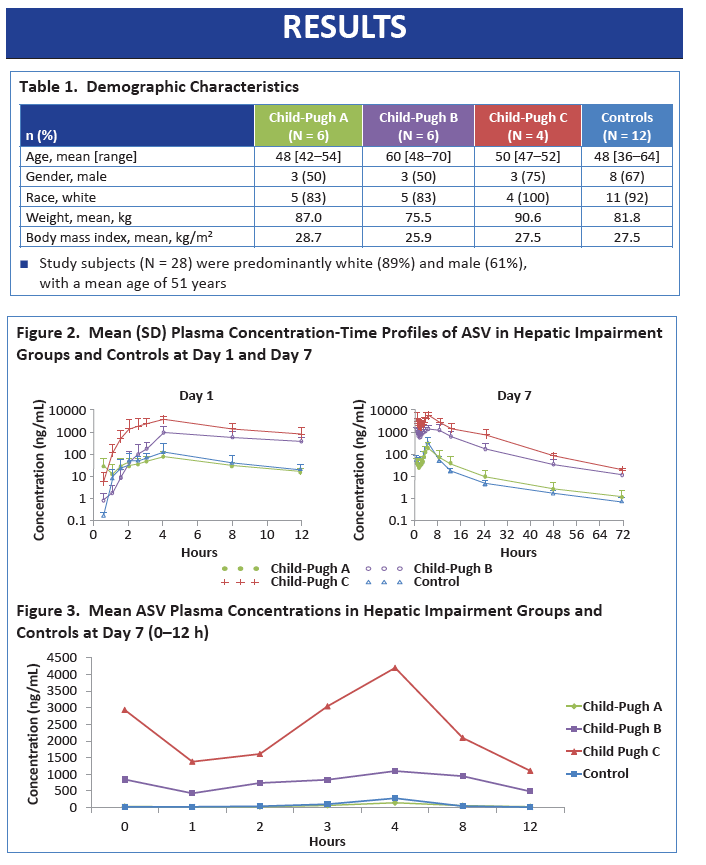
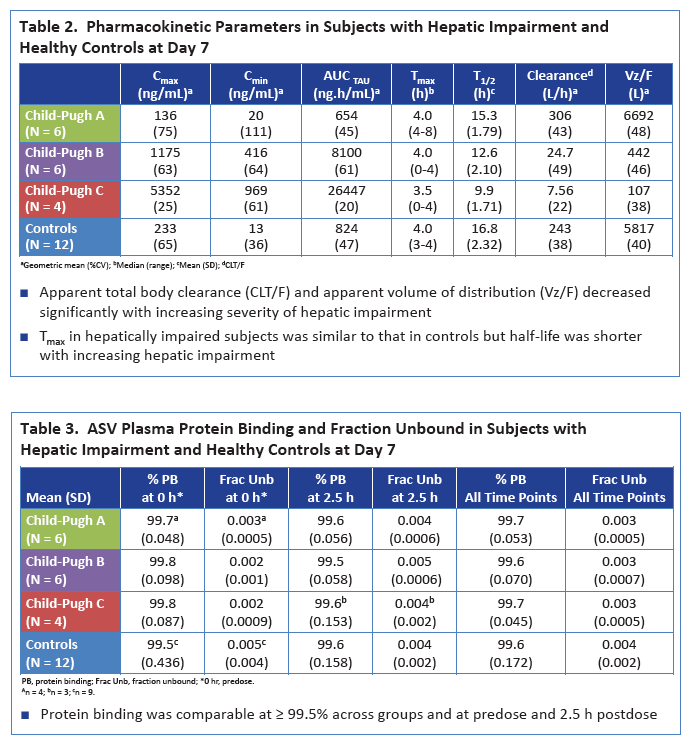
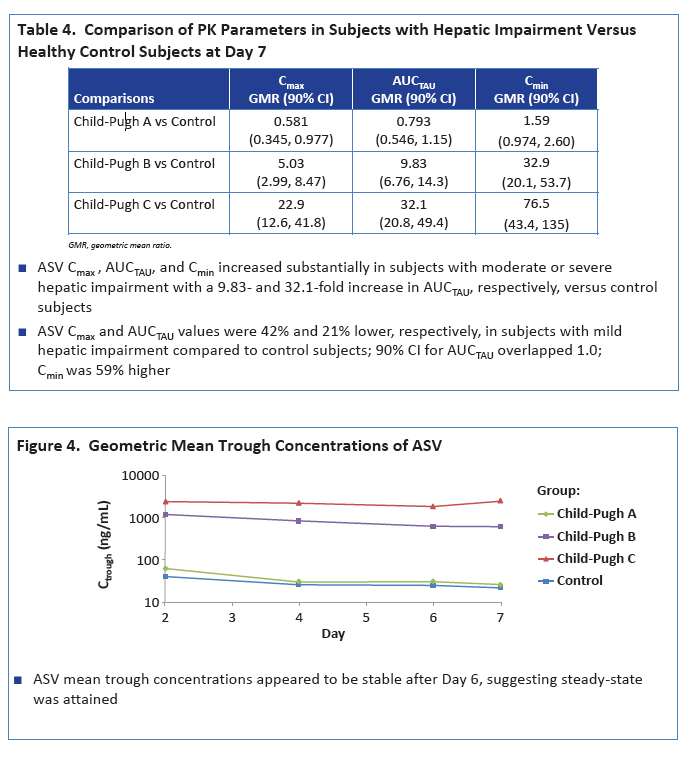
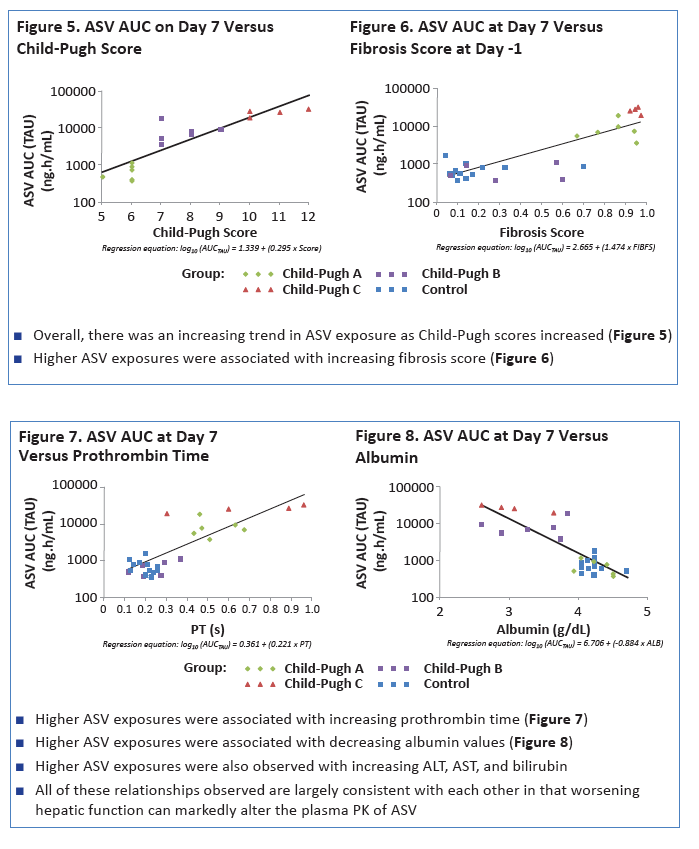

|
| |
|
 |
 |
|
|