 |
 |
 |
| |
DIFFERENT LIKELIHOOD OF ACHIEVING SVR ON A TELAPREVIR-CONTAINING
REGIMEN AMONG NULL RESPONDERS, PARTIAL RESPONDERS AND
RELAPSERS IRRESPECTIVE OF SIMILAR RESPONSESAFTER A
PEGINTERFERON/RIBAVIRIN 4-WEEK LEAD-IN PHASE: REALIZE STUDY
SUBANALYSIS: "When examining this population of TVR-treated patients
as a whole, there was a clear relationship between Week 4 response and
SVR rates"
|
| |
| |
Reported by Jules Levin
Presented at the 22nd Conference of the Asian Pacific Association for the
Study of the Liver (APASL), Taipei, Taiwan, February 16-19, 2012
Graham R Foster1, Stuart Roberts2, Stanislas Pol3, Pietro Andreone4, Eric J Lawitz5,
Zobair M Younossi6, Sandra De Meyer7, Don Luo8, Gaston Picchio8, Stefan Zeuzem9
1Queen Mary University of London, Institute of Cell and Molecular Science, London,
United Kingdom; 2Department of Gastroenterology, Alfred Hospital, Melbourne, VIC,
Australia; 3Assistance Publique-Hopitaux de Paris, Cochin Hospital, Paris, France; 4
Universita di Bologna, Bologna, Italy; 5Alamo Medical Research, San Antonio, TX, USA;
6Center for Liver Diseases and Department of Medicine, Inova Fairfax Hospital, Falls Church,
VA, USA; 7Janssen Infectious Diseases BVBA, Beerse, Belgium; 8Janssen Research &
Development, Titusville, NJ, USA; 9Johann Wolfgang Goethe University Medical Center,
Frankfurt am Main, Germany
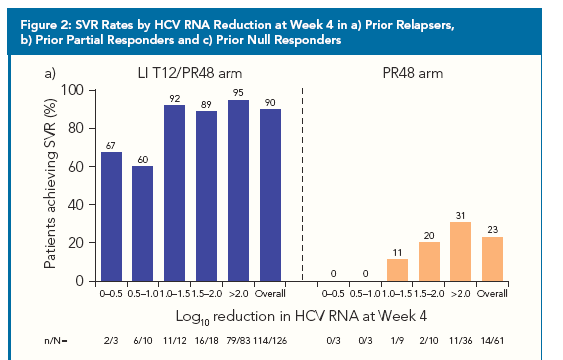
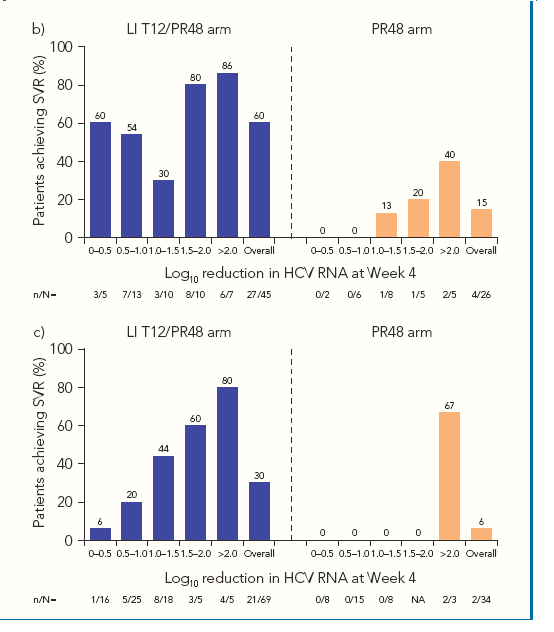
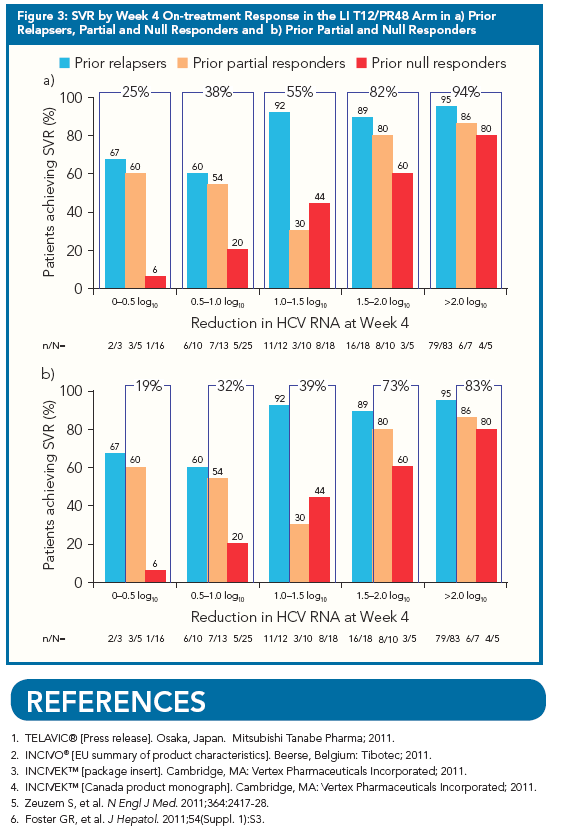
"Since prior response data may not be available for all patients in clinical practice,
we also calculated overall SVR rates according to Week 4 response in the
LI T12/PR48 arm (Figure 3a). When examining this population of TVR-treated
patients as a whole, there was a clear relationship between
Week 4 response and SVR rates"
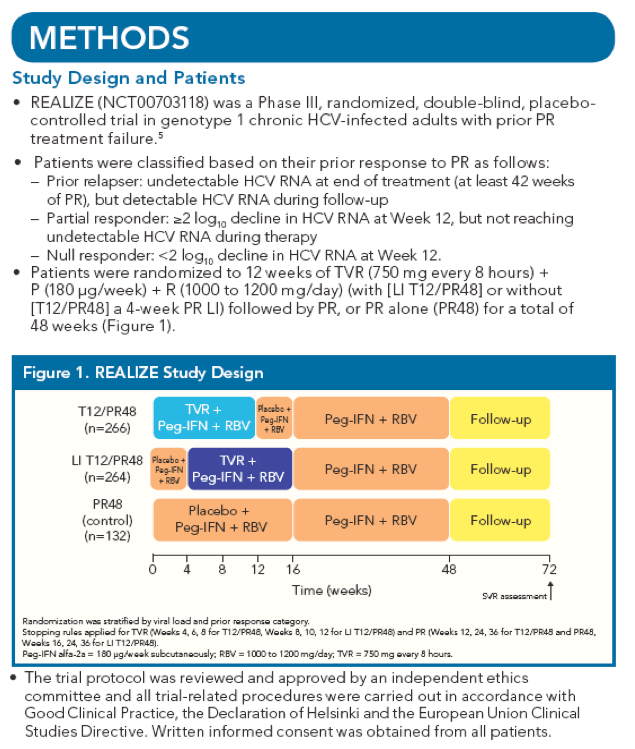
In the present analysis, we aimed to determine if refining the Week 4 < or ≥1 log10 HCV
RNA response categories impacts SVR with TVR-based treatment in previously treated
patients. Therefore we conducted an analysis using smaller intervals of HCV RNA
response at Week 4 in the PR and the LI TVR plus PR arms.
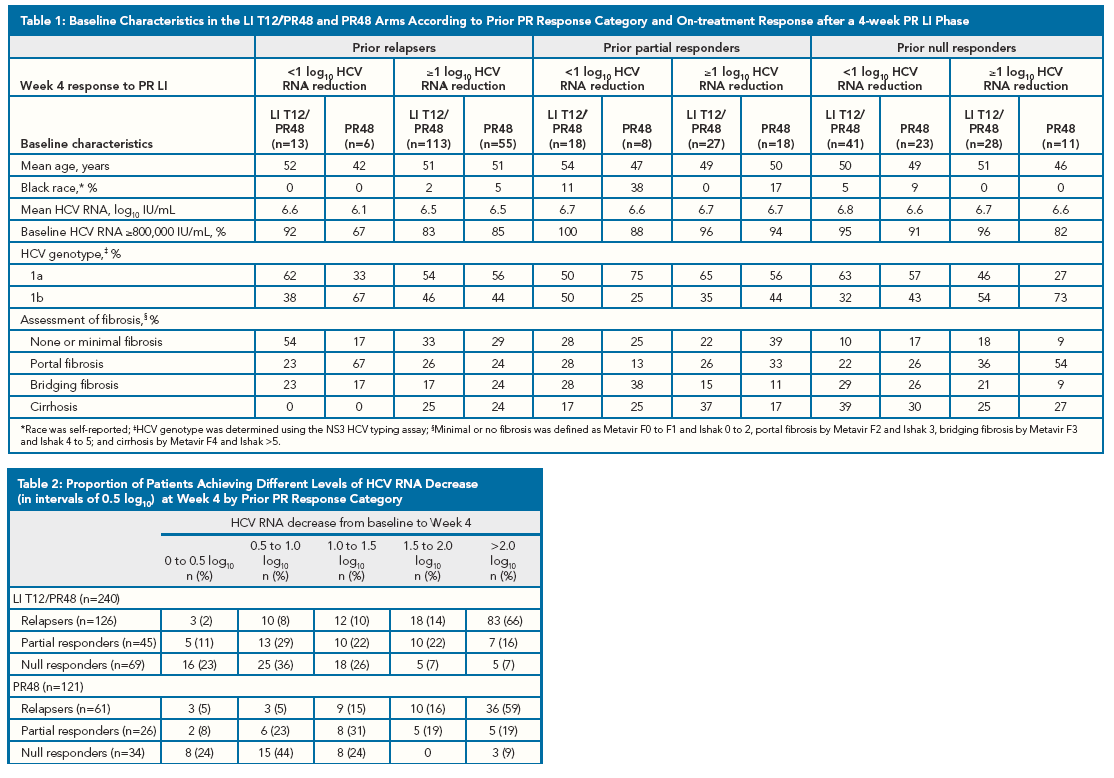
Patients in the LI T12/PR48 and PR48 arms were categorized according to their HCV RNA
reduction after 4 weeks of PR. Week 4 response groups were: 0 to <0.5, ≥0.5 to <1.0, ≥1.0 to
<1.5, ≥1.5 to <2.0, or ≥2.0 log10 decrease in HCV RNA. These 0.5 log10 intervals are greater
than the assays' coefficient of variation (0.2 log10).
· SVR rates by Week 4 HCV RNA response were analyzed for patients in each prior
response category.




|
| |
|
 |
 |
|
|