 |
 |
 |
| |
Hepatitis B Surface Antigen (HBsAg) Loss in HBeAg+ Patients with Chronic Hepatitis B (CHB) Treated with Tenofovir Disoproxil Fumarate (TDF)
|
| |
| |
Reported by Jules Levin
22nd Conference of the Asian Pacifi c
Association for the Study of the Liver (APASL)
February 16-19, 2012
Taipei, Taiwan
P Marcellin1, E Heathcote2, M Buti3, Z Krastev4, I M Jacobson5, R A De Man6, G M Dusheiko7, S Zeuzem8, C N Barnes9, L Lou9 1Hopital Beaujon, University of Paris, Clichy, France; 2Toronto Western Hospital, University of Toronto, Toronto, ON, Canada; 3Servicio de Medicina Interna Hepatologia, Hospital General Universitari Vall d'Hebron and Ciberehd, Barcelona, Spain; 4University Hospital St. Ivan Rilsky, Sofi a, Bulgaria; 5Weill Medical College of Cornell University, New York, NY, USA; 6Erasmus MC Universitiy Medial Center , Rotterdam, The Netherlands; 7Royal Free Hospital, London, UK; 8Medizinische Klinik I, Frankfurt, Germany; 9Gilead Sciences, Foster City, CA
Conclusions
· Continued treatment with TDF increased the proportion of both HBeAg(+) and HBeAg(-)patients with reduced HBsAg level
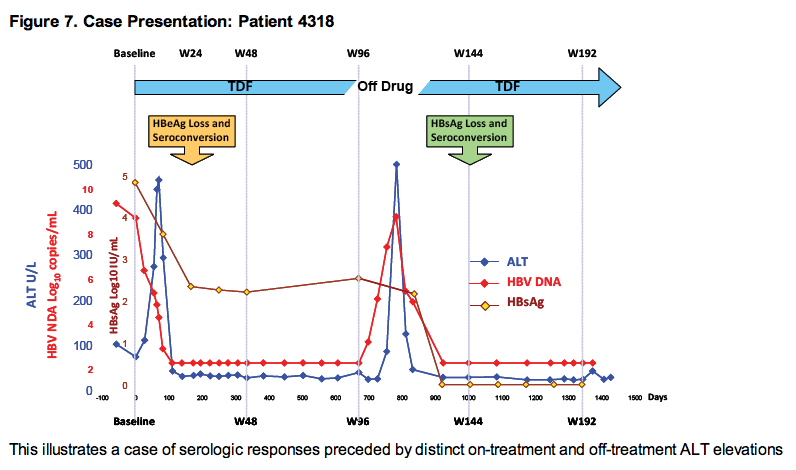
· Loss of HBsAg is nearly always preceded or accompanied by HBeAg seroconversion and frequently heralded by ALT elevations
Background
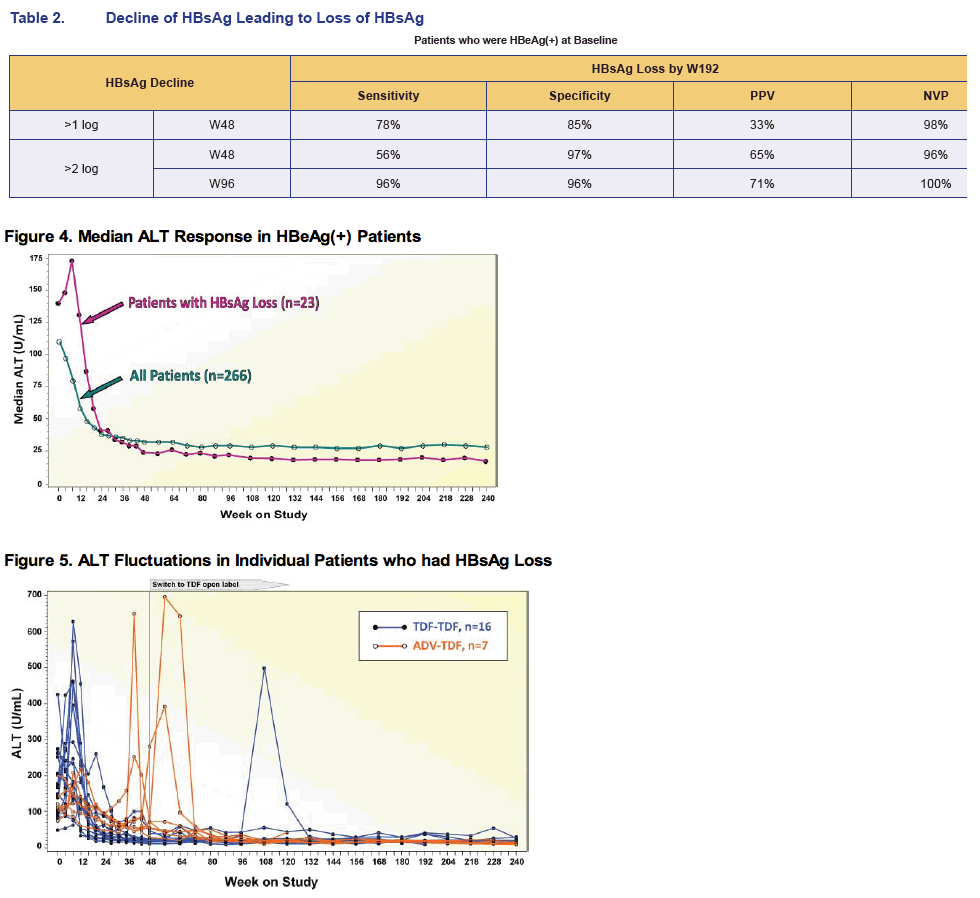
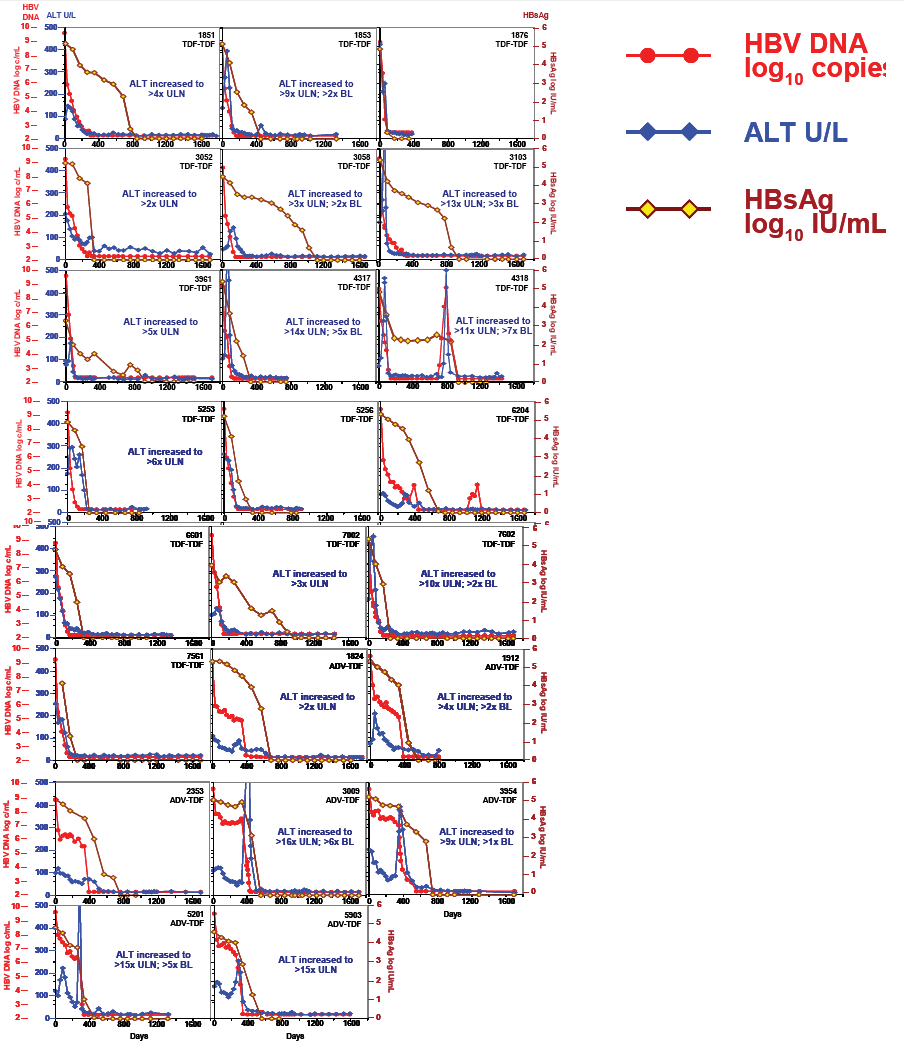
· In TDF pivotal study 103, 23 (11%) patients who were HBeAg(+) at baseline had HBsAg loss with up to 5 years of TDF treatment. All 23 patients achieved HBsAg loss by Year 41.
- 14 (23%) genotype A, 7 (8%) genotype D, 1 (3%) genotype B, 1 (12%) genotype F
· Multivariate analysis of HBeAg(+) patients treated with TDF for 4 years revealed that HBsAg loss is strongly associated with2:
- HBeAg loss during the first 24 weeks
- Rapid HBsAg decline during the first 12 weeks
- Shorter known duration of infection
· One patient in study 102, who was HBeAg(-) at baseline had HBsAg loss with up to 5 years of TDF treatment3
Objective
· To examine the dynamics of serum HBsAg levels in relation to the virologic, serologic and biochemical outcomes associated with up to 240 weeks of TDF treatment
· To empirically identify patient characteristics associated with HBsAg decline and loss
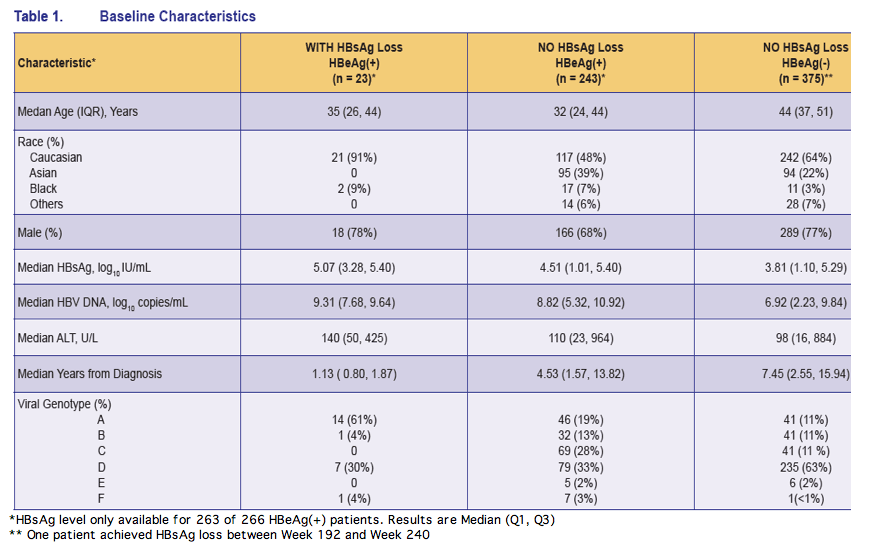
*HBsAg level only available for 263 of 266 HBeAg(+) patients. Results are Median (Q1, Q3)
** One patient achieved HBsAg loss between Week 192 and Week 240
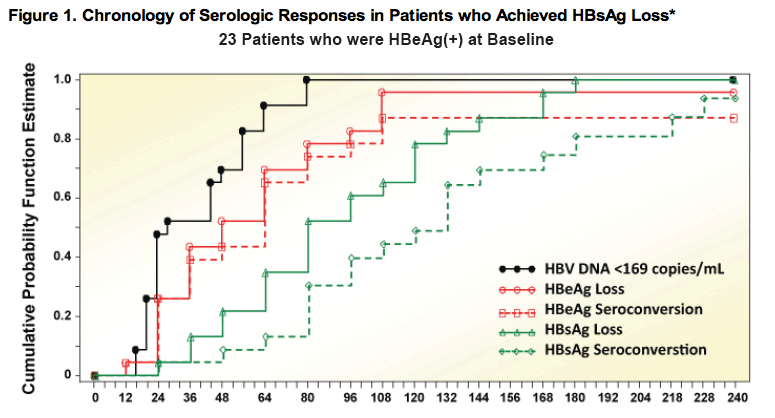
*Kaplan-Meier Analysis reports confi rmed fi rst time-to-event for all data in this figure. A confirmed event is defined as an event observed in two or more consecutive visits. Reversions are not captured by this methodology
Summary of Serologic Response
· 23 patients who were HBeAg(+) at baseline and achieved HBsAg loss and 19
had HBsAg seroconversion up to 240 weeks of TDF treatment; 2 had reversion
back to HBsAg(+) (1 not confirmed)
· 23 of the 23 patients achieved HBV DNA < 169 copies/mL prior to HBsAg loss
· 22 of the 23 had confirmed HBeAg loss; 20 had HBeAg seroconversion
- 19 patients had HBeAg loss prior to or at the same visit as HBsAg loss
- 3 patients had HBeAg loss after HBsAg loss but prior to or at the same visit as HBsAg seroconversion
- 1 patient who had HBsAg loss/seroconversion did not have confi rmed HBeAg loss
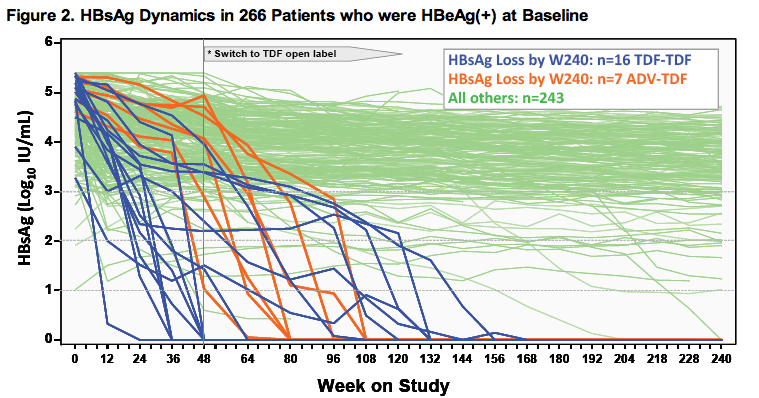
*TDF-TDF and ADV-TDF patients were randomized to receive TDF and ADV, respectively, for the first 48-weeks; both continued with open-label TDF after
Week 48.
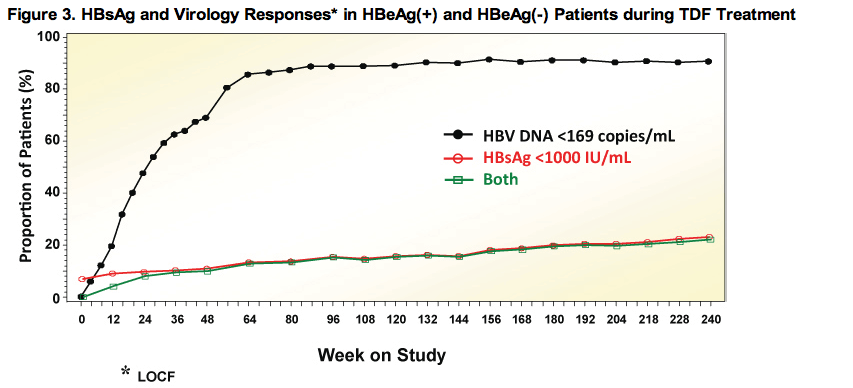
A Subset of Patients with HBsAg and Virologic Responses
· Increasing number of patients reached HBV DNA < 169 copies/mL and HBsAg < 1000 IU/mL with continuous TDF treatment
· 120 patients had the above at Week 240, or reached the above and had HBsAg loss prior to Week 240; 91 of 120 had HBsAg >1000 IU/mL at baseline

|
| |
|
 |
 |
|
|