 |
 |
 |
| |
Clinically Significant Drug Interaction Between Colchicine and Ritonavir in Healthy Adults
|
| |
| |
Reported by Jules Levin
Presented at the 14th International Workshop on Co-morbidities and
Adverse Drug Reactions in HIV. 19-21 July 2012, Washington, DC.
S. Wason,1 J. L. DiGiacinto,2,3 and M. W. Davis1
1URL Pharma, Inc., Mutual Pharmaceutical Company, Inc., Philadelphia, PA; 2Salamandra, LLC, Bethesda, MD; 3Consultant, URL Pharma, Inc., Philadelphia, PA
Role of the Study Sponsor: This research and medical editorial
support was funded by URL Pharma, Inc., Philadelphia, PA.

ABSTRACT
Objectives: Concomitant administration of colchicine, a substrate for cytochrome 3A4 (CYP3A4) and the P-glycoprotein efflux transporter (P-gp), with a CYP3A4/P-gp inhibitor such as ritonavir has the potential to induce clinically significant increases in colchicine plasma concentrations and adverse events (AEs). A study was conducted to 1) determine the effect of multiple-dose ritonavir on the pharmacokinetics of a single dose of colchicine in healthy fasted adults and 2) assess the safety and tolerability of a single dose of colchicine administered alone and with multiple-dose ritonavir.
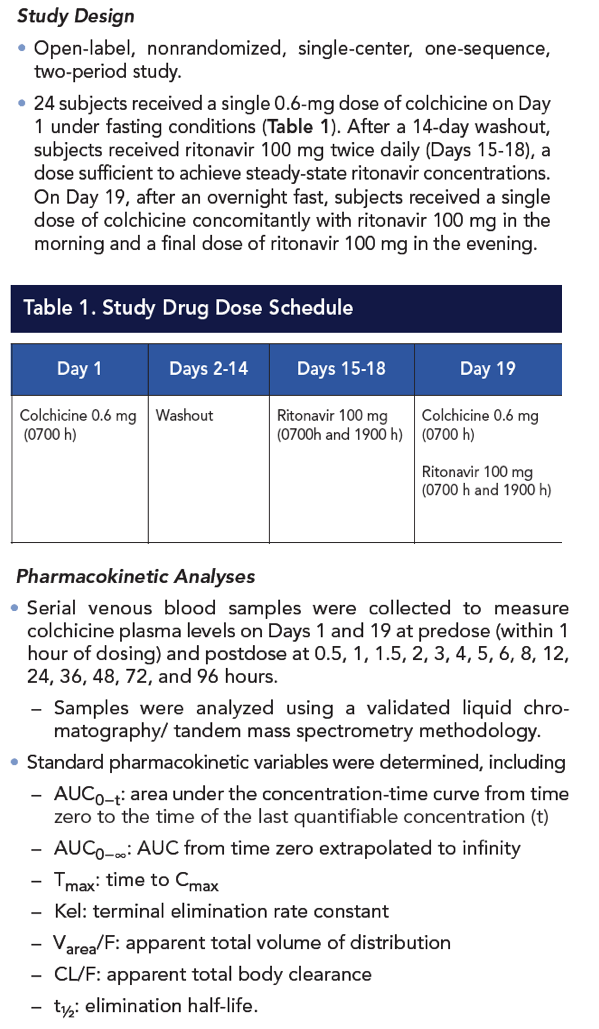
Methods: In an open-label, nonrandomized, one-sequence, two-period study, 24 healthy male and female volunteers received a single 0.6-mg dose of colchicine on Day 1 under fasting conditions. After a 14-day washout period, volunteers received ritonavir 100 mg twice daily on Days 15 to 18, a sufficient duration to achieve steady state. On Day 19, after an overnight fast, volunteers received another dose of colchicine 0.6 mg administered concomitantly with ritonavir 100 mg (morning dose) and then received the final 100-mg dose of ritonavir in the evening. Serial blood samples were collected on Days 1 and 19 to determine colchicine plasma concentrations at predose and postdose at 0.5, 1, 1.5, 2, 3, 4, 5, 6, 8, 12, 24, 36, 48, 72, and 96 hours. The following pharmacokinetic parameter values were calculated (n=18) for colchicine administered alone and with ritonavir steady-state concentrations: Cmax, Tmax, AUC0-t, AUC0-∞, t1/2, CL/F, and Varea/F.
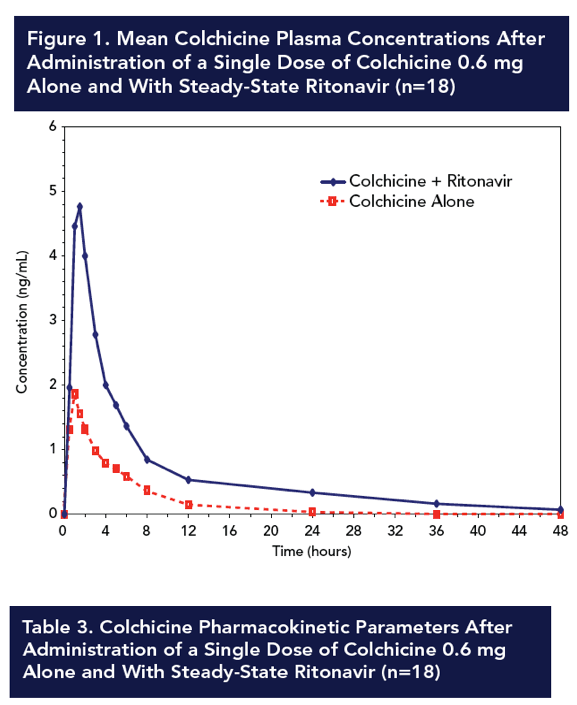
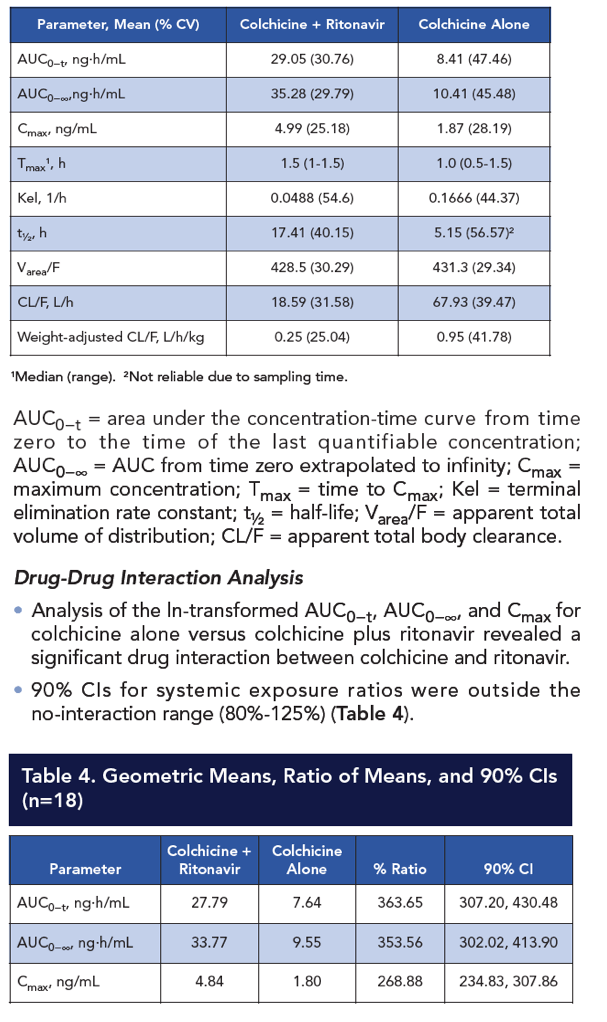
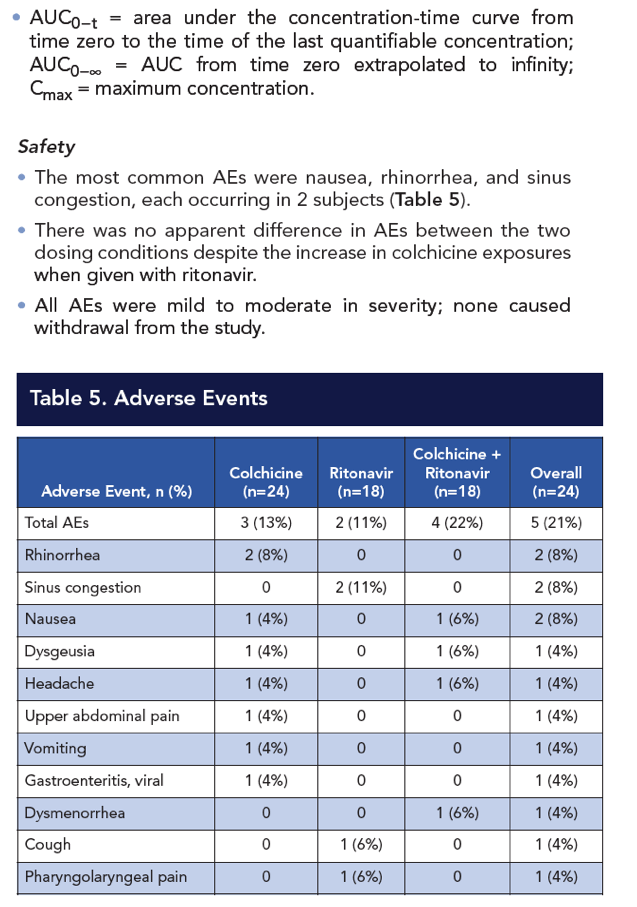
Results: Compared with colchicine alone, coadministration of a single 0.6-mg dose of colchicine with steady-state ritonavir resulted in a 170% increase in colchicine Cmax, a 245% increase in AUC0-t, and a 239% increase in AUC0-∞; Tmax was not affected. CL/F decreased by 70% following colchicine-ritonavir coadministration compared with colchicine alone. Colchicine 0.6 mg alone and with ritonavir was well tolerated, with no apparent increase in AEs during concomitant colchicine-ritonavir therapy. The most common AEs following administration of colchicine, with or without ritonavir, were nausea and rhinorrhea (n=2 for each). All AEs were mild to moderate in severity and did not result in withdrawal from the study.
Conclusion: A clinically significant drug-drug interaction occurs with the concomitant administration of single-dose colchicine and multiple-dose ritonavir that results in significant increases in systemic colchicine exposures. In this study, concomitant colchicine-ritonavir administration did not significantly increase the AE profile compared to colchicine alone. Nevertheless, colchicine dose adjustment is recommended when colchicine is coadministered with ritonavir or other strong CYP3A4/P-gp inhibitors to minimize the risk of colchicine-related toxicities.



|
| |
|
 |
 |
|
|