 |
 |
 |
| |
ABT-267 Combined With Pegylated Interferon Alpha-2a/ribavirin in Genotype 1 (GT1) HCV-Infected Treatment-Naïve
Subjects: 12-Week Antiviral and Safety Analysis
|
| |
| |
Reported by Jules Levin
EASL 2012 Apr 18-22 Barcelona Spain
J Greg Sullivan1, Maribel Rodriguez-Torres2, Eric Lawitz3, Fred Poordad4, Lino Rodrigues5, Andrew Campbell5, Carolyn Setze5, Wangang Xie5, Amit Khatri5, Emily Dumas5, Preethi Krishnan5, Tami Pilot-Matias5,
Laura Williams5, Barry Bernstein5
1Parkway Medical Center, Birmingham, Alabama, United States; 2Fundacion de Investigacion de Diego, San Juan, Puerto Rico; 3Alamo Medical Research, San Antonio, Texas, United States; 4Cedars-Sinai Medical Center, Los Angeles, California, United States, 5Abbott, Abbott Park, Illinois, United States
CONCLUSIONS
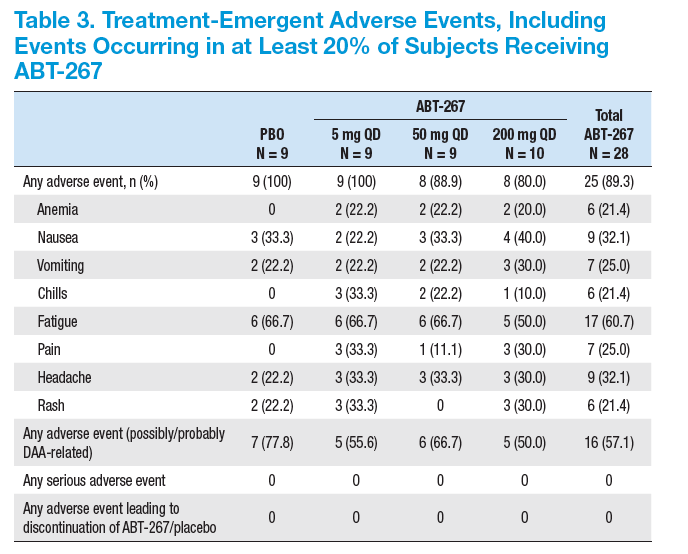
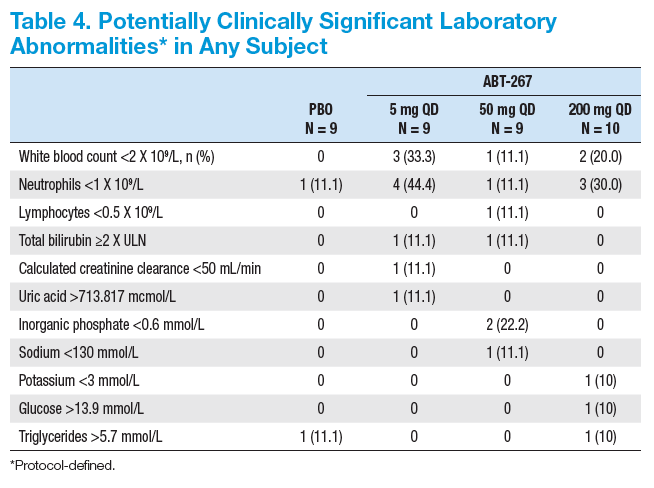
· ABT-267 + P/R was well tolerated with a safety profi le similar to P/R alone
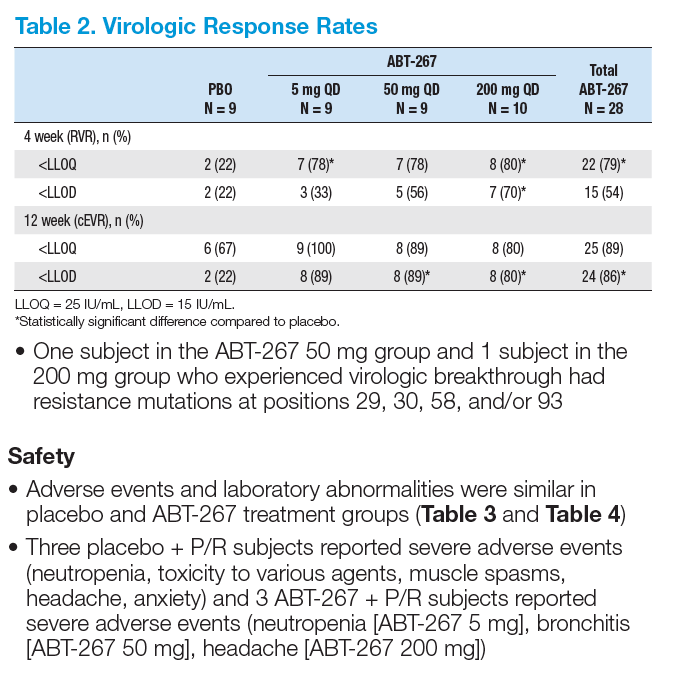
· ABT-267 + P/R demonstrated potent antiviral activity with 86% of subjects achieving cEVR
(HCV RNA < LLOD) in GT1 HCV-infected treatment-naïve subjects
· These data support additional studies with ABT-267 in combination with other DAAs for the
treatment of chronic GT1 HCV infection



|
| |
|
 |
 |
|
|