 |
 |
 |
| |
Triple Therapy With Daclatasvir (DCV; BMS-790052), Peginterferon Alfa-2a and Ribavirin in HCV-Infected Prior Null and Partial Responders: 12-Week Results of Phase 2b COMMAND-2 Trial
|
| |
| |
Reported by Jules Levin
EASL 2012
Apr 18-22 Barcelona Spain
Ratziu V,1 Gadano A,2 Pol S,3 Hezode C,4 Ramji A,5 Cheng W,6 Sulkowski M,7 Everson GT,8 Diva U,9 McPhee F,9 Wind-Rotolo M,10 Hughes EA,10 Yin PD,9 Schnittman S,9 for the COMMAND-2 Investigators 1Hôpital Pitie Salpêtriere, Paris, France; 2Hospital Italiano de Buenos Aires, Buenos Aires, Argentina; 3Hôpital Cochin, Paris, France; 4 CHU Henri Mondor, Creteil, France; 5Gastrointestinal Research Institute, Vancouver, Canada; 6Royal Perth Hospital, Perth, Australia; 7Johns Hopkins University, Lutherville, MD, USA; 8University of Colorado-Denver, Aurora, CO, USA; 9Bristol-Myers Squibb, Wallingford, CT, USA; 10Bristol-Myers Squibb, Princeton, NJ, USA


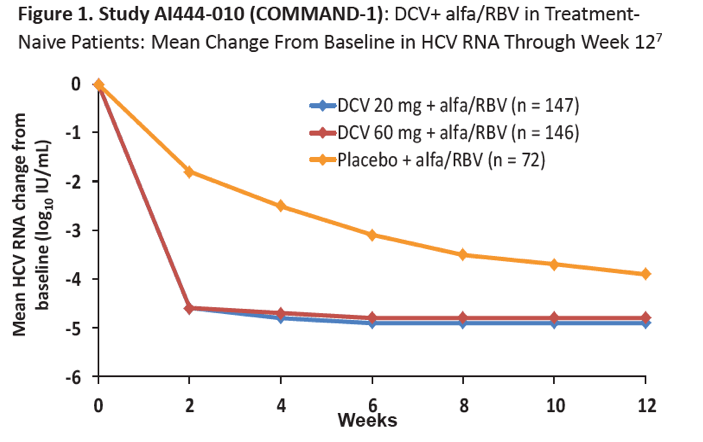

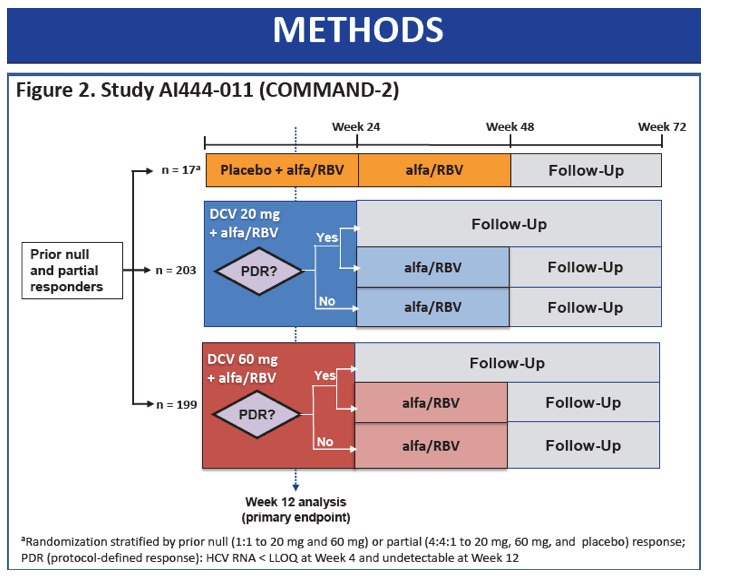

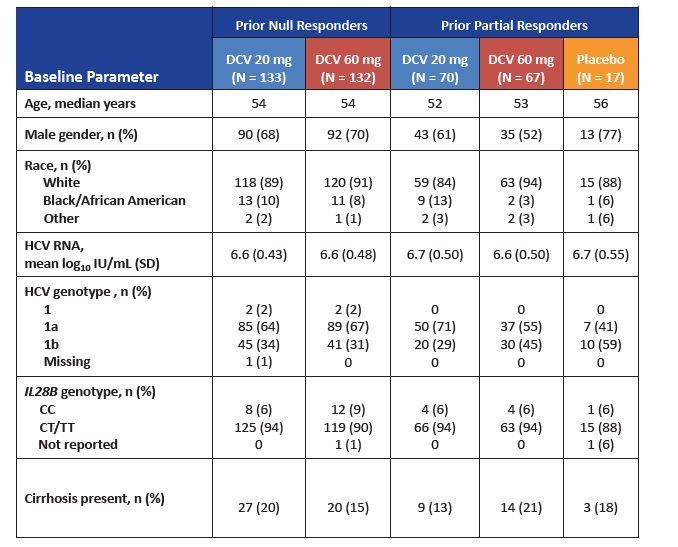
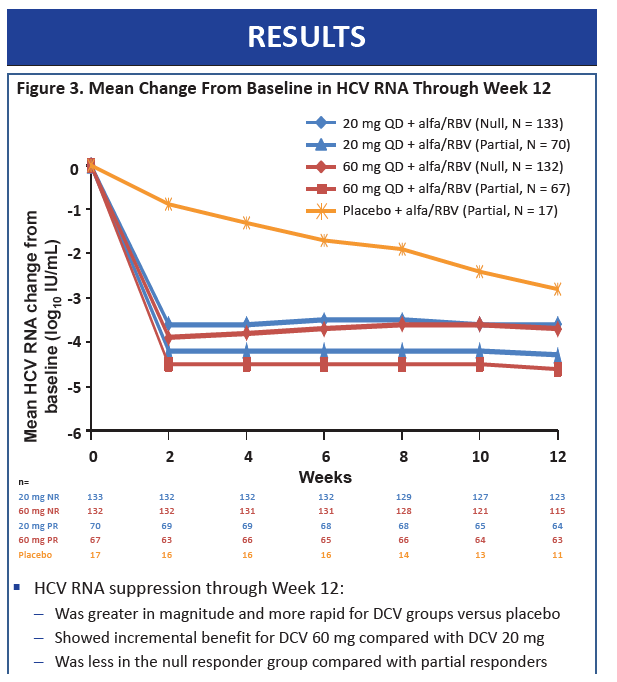
|
| |
|
 |
 |
|
|