 |
 |
 |
| |
Telaprevir French Cohort Authorization for Temporary Use in Genotype 1 Hepatitis C Cirrhotic Patients with Prior Partial Response or Relapse
|
| |
| |
Reported by jules Levin
EASL April 2012 Barcelona, Spain
Blanca Hadacek,1 Anne-Charlotte Danton,1 Nicole Barbé,1 Florence Masson,1 Delphine Quelard,1 Dominik Naessens,2 Gaston Picchio,3 James Witek,3 Isabelle Lonjon-Domanec,1 Alain Miara1
1Janssen-Cilag, Paris, France; 2Janssen Infectious Diseases BVBA, Beerse, Belgium; 3Janssen Research and Development, Titusville, NJ, USA
----------------------------
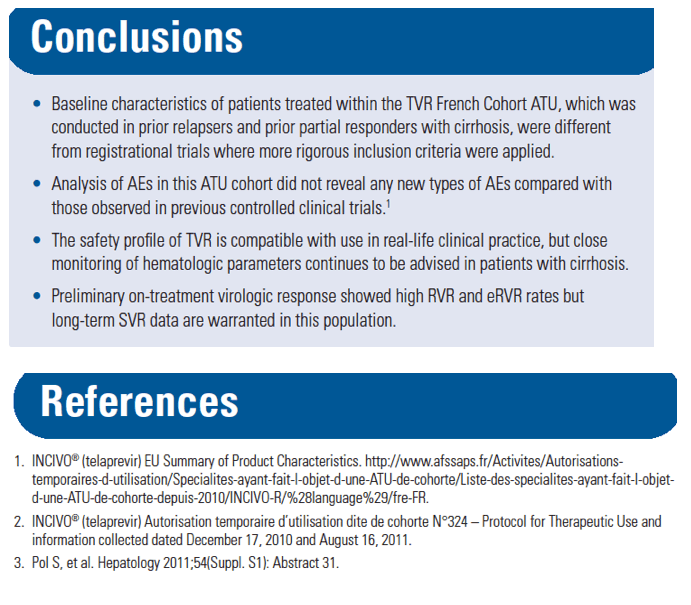
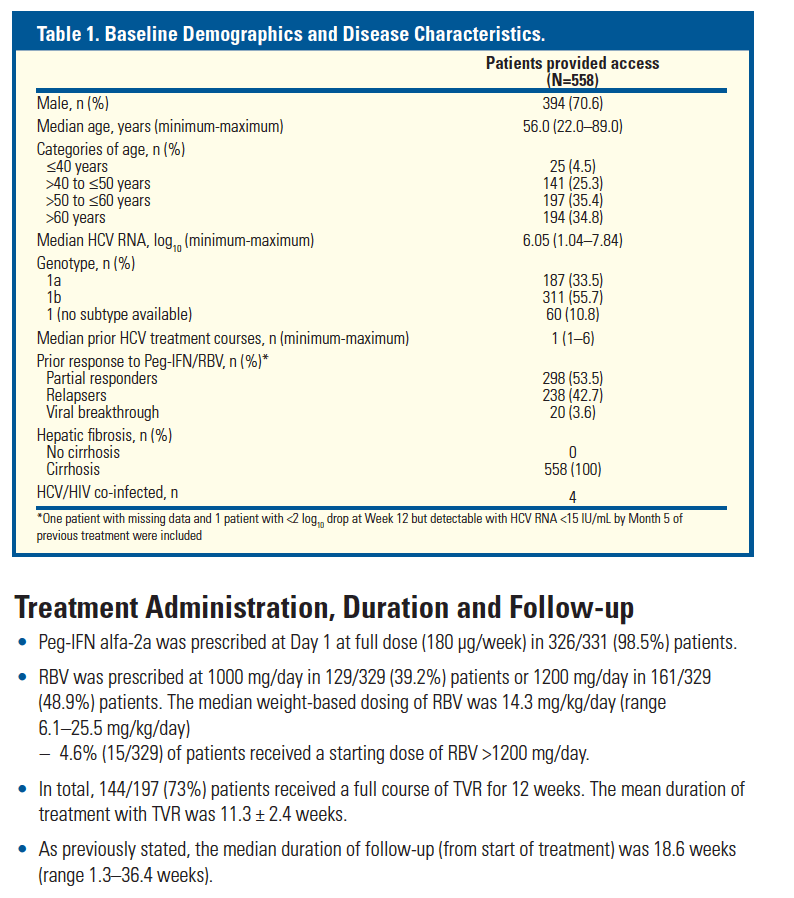
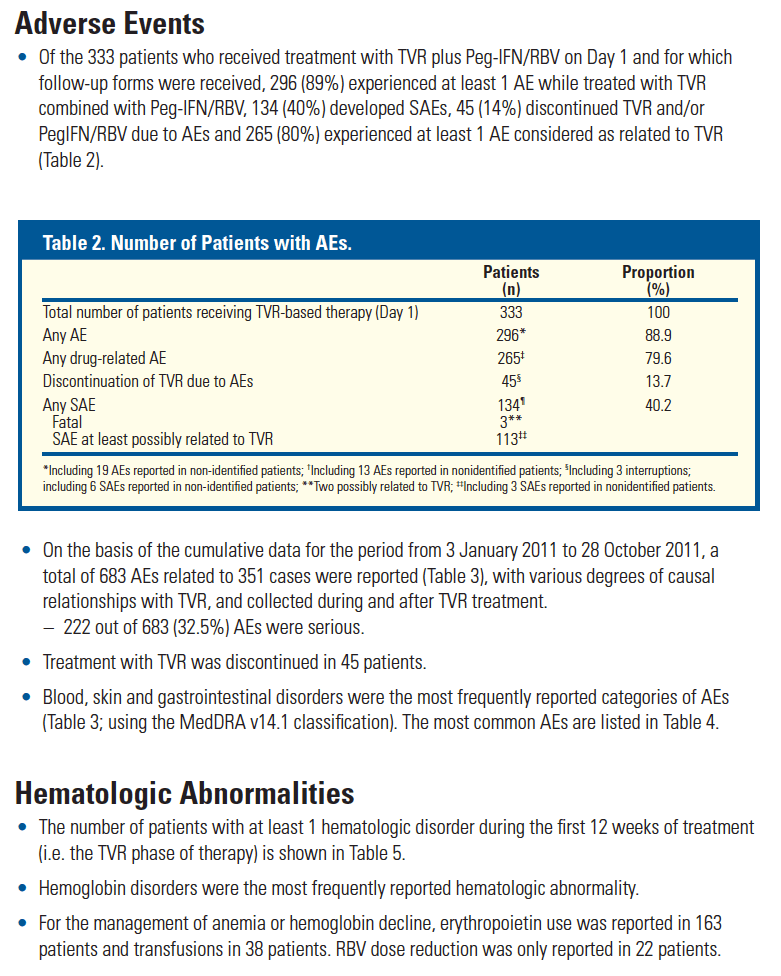
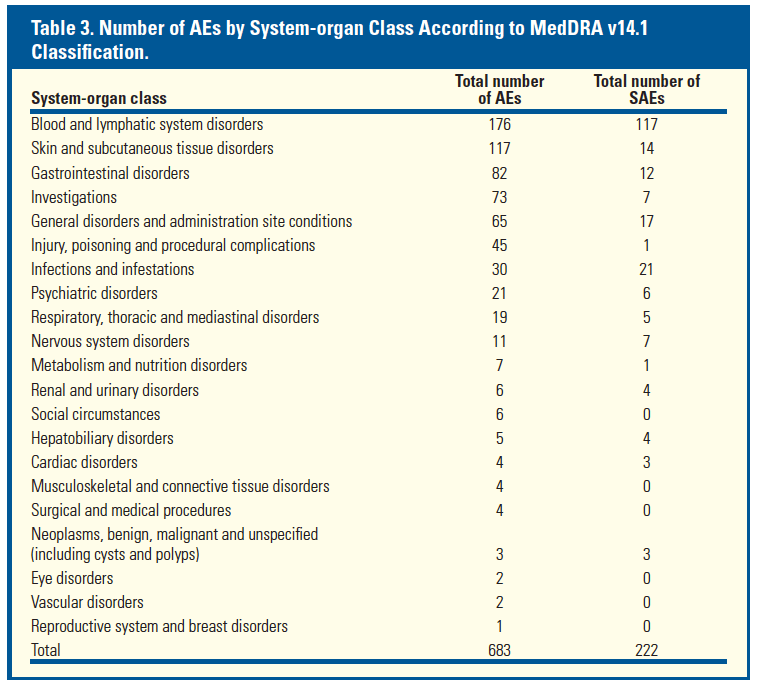
from Jules: However, if you look at this previously reported data just below the safety data reported in this poster from studies previous to approval show there were previous indications of safety concerns in cirrhotics, so my reading is that these data show there were phase 3 data suggesting safety concerns in cirrhotics, unless these data which were reported in Dec 2011 at HepDart were never previously reported to investigators or anyone......
---------------------------------------------------
Review Activity/Safety: Telaprevir in Combination with Peginterferon Alfa-2a and Ribavirin Increased Sustained Virologic Response in Genotype 1 Chronic HCV
Reported by Jules Levin
HEP DART 2011, Koloa, Hawaii, USA, December 4-8, 2011
RS Kauffman*1
http://www.natap.org/2011/hepDART/hepDART_03.htm
--------------------------------------------------






|
| |
|
 |
 |
|
|