| |
Impact of erythropoietin on sustained virological response to peginterferon and ribavirin therapy for HCV infection: a systematic review and meta-analysis
|
| |
| |
Download the PDF here
Journal of Viral Hepatitis
February 2012
"Our meta-analysis determined that administration of EPO in patients who developed anaemia during anti-HCV combination therapy can considerably enhance SVR. No adverse event of EPO administration was reported among included subjects........Patients who developed anaemia and received EPO as adjuvant therapy had a significantly higher rate of SVR compared with those with anaemia who underwent RBV dose reduction as standard care with RR of 1.83 (95% CI 1.41-2.37). SVR occurred in 88 of 131 (67%) from the former group and 46 of 126 (37%) from the latter group."
Abstract
Summary. Anaemia is a common complication of antiviral therapy for chronic hepatitis C virus (HCV) infection that necessitates dose reductions or therapy discontinuation. Administration of erythropoietin (EPO) is an alternative to ribavirin (RBV) dose reduction, but its advantage in terms of sustained virological response (SVR) has not been determined yet. In a systematic way, randomized studies were identified that evaluated the effect of EPO administration vs RBV dose reduction on virological response in patients who developed anaemia during anti-HCV therapy. The random-effects model was employed to run meta-analysis. SVR was set as the end point of interest. Data were abstracted from four studies containing 257 patients who developed anaemia during therapy. One hundred and twenty six subjects underwent RBV dose reduction. Patients who received EPO in response to haemoglobin drop had a significantly higher probability of achieving SVR compared with those who underwent RBV dose reduction because of anaemia (relative risk = 1.83 95% CI; 1.41-2.37). No heterogeneity was observed across study results (I2 = 0). Publication bias assessment was nonsignificant. Our meta-analysis indicates that administration of EPO in patients who develop anaemia during anti-HCV therapy can considerably enhance SVR. Moreover, no adverse event of EPO administration was reported among included subjects.
Introduction
Hepatitis C virus (HCV) is an important cause of chronic liver disease. HCV accounts for 20% of acute hepatitis cases, 70% of all chronic hepatitis cases and 40% of all cases of liver cirrhosis [1]. As sustained virological response (SVR) to anti-HCV therapy avoids progression of liver fibrosis, decreases the risk of hepatocellular carcinoma and improves patients' survival, antiviral therapy is considered as a crucial option in the management of chronic HCV infection [2-5].
Current anti-HCV standard therapy is comprised of oral ribavirin (RBV) and peginterferon alpha-2a or alpha-2b. RBV dose during the treatment period and a patient's compliance with this drug are important factors in achieving SVR [6]. However, anaemia is the most challenging side-effect of RBV and is responsible for considerable numbers of dose reduction and treatment cessation [7]. Haemoglobin concentration has been reported to decrease to below 12 g/dL (mean decrease 3.7 g/dL) in 52% of patients receiving peginterferon alpha-2a and RBV combination therapy [Pegasys Package Insert, 2004]. Furthermore, significant anaemia (i.e. haemoglobin <10 g/dL) has been observed in 9-13% of these patients [8]. Although both interferon and RBV cause anaemia, RBV-induced haemolysis is generally identified as the main reason for dose reduction and treatment discontinuation [9].
Administration of recombinant human erythropoietin (EPO) is employed to maintain RBV dose, increase haemoglobin level and improve treatment compliance, whereas its impact in terms of SVR is not currently well elucidated [10-14]. Therefore, in this review, we aimed to draw a robust conclusion about the impact of EPO on SVR, by identifying, summarizing and pooling results of all available randomized clinical trials.
Discussion
Our meta-analysis suggests that administration of EPO agents to avoid further RBV dose reduction or treatment discontinuation significantly improves SVR compared with patients who underwent standard care. This finding is completely compatible with results of studies that evaluated the effect of RBV dose adjustment on virological response [27,28]. In spite of unclear risk of bias across included studies owing to lack of adequate methodological reporting, we believe that we can have high confidence in our findings because of the following reasons: (i) absence of statistical heterogeneity among study results despite different treatment protocols, (ii) statistically significant results despite very limited statistical power, (iii) absence of publication bias that indicates it is unlikely that unidentified studies in grey literature can alter the pooled result and, last but not least, (iv) a robust pooled estimate by exclusion sensitivity analysis that implies the final estimate was not dependent on any single study result. A higher rate of SVR among patients who developed anaemia despite EPO adjuvant therapy compared with other anaemia patients without EPO adjuvant therapy in Shiffman et al.'s study could be due to lower rate of treatment discontinuation, lower RBV dose reduction and tolerance for higher dose of RBV in the high-dose RBV group. Furthermore, in Bertoni et al.'s study, despite EPO usage, none of the patients who received standard care achieved haemoglobin increase. In addition, only in the adjuvant therapy group, the '80/80/80 rule' was respected (administration of at least 80% of the dose of peg-IFN and RBV for at least 80% of the treatment time). These patients had exactly the same rate of SVR as the control group (without anaemia and treated with full dose of RBV). These findings were in line with other included study results.
Erythropoietic agents might not be completely safe. Hypertension, headache, reaction at injection site, increased numbers of platelets in the blood, severe thrombocytopenia and antibody-mediated pure red cell aplasia during anti-HCV therapy are rare complications of EPO adjuvant therapy [29-32]. Although none of the included studies reported any adverse events attributable to EPO among included patients, owing to the small number of included subjects, the full safety of EPO use during anti-HCV therapy could not be assured in this review.
Conclusion
Our meta-analysis determined that administration of EPO in patients who developed anaemia during anti-HCV combination therapy can considerably enhance SVR. No adverse event of EPO administration was reported among included subjects.
Results
Our search strategy yielded 58 unique citations that included four randomized clinical trials [20-23], three prospective [12,24,25] and two retrospective studies [25,26] that evaluated the effect of EPO on SVR. Nonrandomized studies were excluded. Three studies by Falasca, Sharvadze and Bertino et al. randomized patients who developed anaemia to EPO recipients and patients with RBV dose reduction as standard care, whereas Shiffman et al. randomized patients to EPO recipients and placebo recipients from the beginning of study. To compare EPO vs RBV dose reduction, from Shiffman et al.'s study, only data of patients who developed anaemia in either the erythropoietic support group or control group were included in the analyses. Overall, 257 unique patients were included of whom 131 patients had received EPO and 126 subjects had undergone RBV dose reduction.
Study characteristics
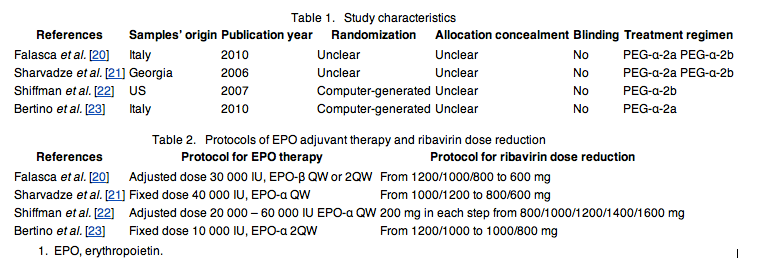
Table 1 shows the characteristics of included studies. All studies were published as full text in peer-review journals between 2006 and 2010. One study was from United States, two from Italy and one from Georgia. In all studies, patients received weight-based RBV accompanying peginterferon alpha-2a or 2b. None of the randomized trials were double-blinded, and the method of concealment was unclear in all RCTs. Bertino and Shiffman had used computer-generated random numbers; however, the method of random number sequence generation was unknown in Scharvadze and Falasca et al.'s study. Table 2 shows predefined protocols for EPO adjuvant therapy and RBV dose reduction in control groups.
Patient characteristics

Table 3 shows baseline characteristics of included patients. Approximately two-thirds of included patients had genotype 1 infection. Sharvadze et al. had not reported history of anti-HCV therapy in their subjects; however, in other studies, only treatment-na•ve patients were eligible for inclusion. Baseline characteristics of included patients were available in Falasca and Bertino et al.'s studies. In Sharvadze and Bertino et al.'s study, all patients who received EPO normalized haemoglobin level and did not need further RBV dose reduction. In Shiffman et al.'s study, patients who dropped haemoglobin despite EPO therapy vs those who developed anaemia without haematologic support are compared.
Comparative sustained virological response rate of anaemia patients who received erythropoietin vs standard care
Patients who developed anaemia and received EPO as adjuvant therapy had a significantly higher rate of SVR compared with those with anaemia who underwent RBV dose reduction as standard care with RR of 1.83 (95% CI 1.41-2.37). SVR occurred in 88 of 131 (67%) from the former group and 46 of 126 (37%) from the latter group. No heterogeneity was observed between study results (P = 0.88, I2 = 0). Figure 1 shows the summary estimates with 95% CI of included studies and pooled RR for likelihood of SVR in patients who received EPO vs those who received standard care. As presented in Fig. 2, the pooled estimate was not dependent on any single study. Publication bias assessment was nonsignificant for this analysis (P = 0.29) (Fig. 3).
|
|
| |
| |
|
|
|