| |
Bristol-Myers Squibb to Present New Data Demonstrating Company's Commitment to Research and Development in Liver Disease at The International Liver CongressTM / European Association for the Study of the Liver (EASL) Annual Meeting
|
| |
| |
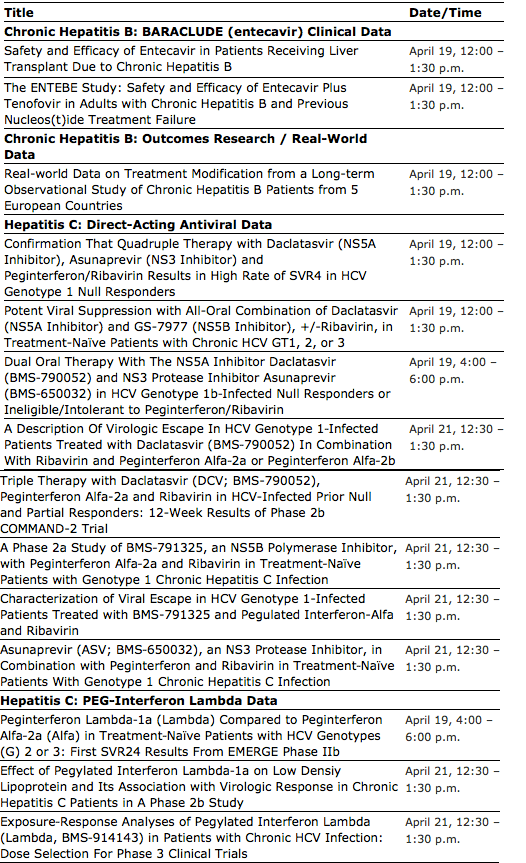
First Report of SVR4 Data from the study of daclatasvir (BMS-790052) plus GS-7977 +/-ribavirin in treatment-naïve patients with chronic hepatitis C genotype 1, 2, or 3, during a late breaker poster presentation
Oral presentations on hepatitis C investigational compounds daclatasvir (BMS-790052), asunaprevir (BMS-650032), and peginterferon lambda-1a (Lambda) demonstrate advancement of robust pipeline
Breadth of data highlights Company's commitment to pursuing research that aims to improve the management of liver disease
PRINCETON, N.J.--(BUSINESS WIRE)--Bristol-Myers Squibb Company (NYSE:BMY) announced today that 20 abstracts on the Company's research in liver disease have been accepted for presentation at The International Liver CongressTM 2012, the 47th annual meeting of the European Association for the Study of the Liver (EASL), in Barcelona, April 18 - 22. Bristol-Myers Squibb is studying a portfolio of compounds that has the potential to address unmet medical needs for patients with liver disease, including the investigational compounds daclatasvir, asunaprevir, Lambda, BMS-791325, and BMS-986094 (INX-189) for hepatitis C (HCV); brivanib for hepatocellular carcinoma (HCC); and BARACLUDE® (entecavir). BARACLUDE is currently indicated for the treatment of chronic hepatitis B (CHB) in adults with evidence of active viral replication and either evidence of persistent elevations in aminotransferases (ALT or AST), or histologically active disease.
Key presentations include one late breaker poster presentation and two oral presentations of Phase II data on the Company's investigational HCV direct-acting antivirals (DAAs) and Lambda:
· A late breaker presentation on the first report of SVR4 results from a Phase II study of the direct-acting antiviral daclatasvir (NS5A Inhibitor) and GS-7977 (formerly PSI-7977; an NS5B inhibitor), +/-ribavirin, in treatment-naïve patients with chronic HCV genotype 1, 2, or 3
· An oral presentation on dual oral therapy with daclatasvir plus asunaprevir in HCV genotype 1b-infected null responders or ineligible/intolerant to alfa/ribavirin
· The first report of SVR24 results from the EMERGE Phase IIb study peginterferon Lambda-1a (Lambda) compared to peginterferon alfa-2a (alfa) in treatment-naïve patients with HCV genotypes 2 or 3
"Bristol-Myers Squibb is at the forefront of discovering, developing, and delivering potential treatments for diseases of the liver where there remains considerable unmet medical needs," said Brian Daniels, MD, senior vice president, Global Development and Medical Affairs, Research and Development, Bristol-Myers Squibb. "For example, our goal in hepatitis C is to expand treatment options for patients by developing our portfolio of investigational compounds through multiple treatment approaches. The data we are presenting at the International Liver Congress help to expand our understanding of the potential efficacy and safety profiles of these investigational compounds and support our ongoing Phase III development program in HCV."
The Company will also present a late-breaker oral presentation on data from the BRISK-PS study of investigational compound brivanib in patients with HCC who failed or were intolerant to sorafenib and six presentations of outcomes research/real-world data that add to the understanding of the prevalence of and current treatment patterns in HBV, HCV and HCC.
The complete list of Bristol-Myers Squibb data presentations is below. Abstracts can be accessed on the ILC/EASL website at
http://www.easl.eu/_the-international-liver-congress/general-information.

|
|
| |
| |
|
|
|