 |
 |
 |
| |
Prevent Drug Craving and Relapse: A Memory Retrieval-Extinction Procedure - New Published Research
|
| |
| |
Download the PDF here
Download the PDF here
Download the PDF here
Timing Key in Rewriting Addict's Memory
By Kristina Fiore, Staff Writer, MedPage Today
Published: April 13, 2012
studies in both rats and heroin addicts suggest that memory reconsolidation performed immediately before an extinction procedure can diminish drug craving in the absence of the use of any pharmacologic agent. Using a type of exposure therapy to wipe out the emotional triggers that drive drug addiction may be most effective when the brain is in the process of recalling and re-storing memories, Chinese researchers found.
Calling up a drug-related memory just before having "extinction" therapy -- seeing drug-related cues without being able to use -- was associated with diminished cravings in rat and human models, Lin Lu, PhD, of Peking University in Beijing, and colleagues reported in Science. Performing this type of emotional-memory-scrambling as a memory is "reconsolidating" may eventually provide a means of treating addiction, according to David Epstein, PhD, of the National Institute on Drug Abuse (NIDA), a co-author on the paper. The idea behind memory reconsolidation is that once a patient consciously recalls a memory, it has to be stored in the brain as a new memory all over again. It can be altered, however, during that process.
Epstein explained that by itself, extinction therapy -- similar to exposure therapy, which tempers PTSD symptoms in veterans by having them relive the traumatic moment -- doesn't work all that well in drug addiction because it doesn't weaken the initial memory that drives the user to seek out more drugs.
"It's only new learning that's superimposed on top of an old memory that stubbornly sticks around," he said.
But if you perform extinction during memory reconsolidation and that memory is vulnerable to interference, Epstein said, it can be weakened.
"So when you do the extinction [during reconsolidation], you're doing more than just creating the usual new learning," he said. "You're overwriting the old memory."
Researchers have already tinkered with emotional-memory reconsolidation in addicts by using pharmacologics, mainly propranolol (Inderal), but also in animal models with other agents not approved for use in humans. So Lu and colleagues' aim was to be able to interfere with the process without using any drugs.
In a series of experiments in rats, they found that those addicted to cocaine or heroin had no interest in the drugs after having reconsolidation and extinction -- but only if the reminder occurred 10 minutes or an hour before extinction therapy.
Drug-seeking behaviors returned if 6 hours had elapsed between the memory trigger and the exposure therapy, the researchers reported.
"The timing is really crucial," Epstein said. "It reflects the limited time window during which memories are vulnerable to disruption."
In another model in which the rats were able to self-administer drugs, only those that had reconsolidation and extinction -- not extinction alone -- were less likely to resume drug-seeking behaviors a month later, they found.
Lu and colleagues then tested the treatment in Chinese heroin addicts who'd had medically supervised detoxification and were inpatients at a treatment facility.
Patients had either extinction therapy alone, or memory retrieval -- a 5-minute video of heroin use -- either 10 minutes or 6 hours before extinction therapy in which they were exposed to drug images and paraphernalia but not given any drugs.
Only those who had the 10-minute interval between the memory trigger and extinction therapy had significantly reduced cravings (P<0.01) and a trend toward decreases in blood pressure (P=0.09) that were sustained for about 6 months, the researchers reported, noting, however, that there were no changes in heart rate.
"That's kind of astonishing considering how nonintrusive and how seemingly inconsequential this procedure is," Epstein said.
But he noted that the trial reveals nothing about what would happen to the heroin addicts if they were exposed to the real-world environmental cues that got them hooked in the first place.
One of the next steps, he said, would be to conduct similar trials in an outpatient setting. No such U.S.-based trial is planned, but Epstein said NIDA is currently conducting an outpatient trial that uses propranolol to interfere with memory reconsolidation in addicts -- although it "would be nice to be able to do it completely behaviorally," he said.
In terms of potential neural mechanisms, reconsolidation and extinction have been shown to depend on activation of N-methyl-D-aspartate (NMDA) receptors, the researchers said. In their study, they also saw increased expression of protein kinase C in the infralimbic cortex and diminished expression in the basolateral amygdala.
This may lead to structural changes in neurons that stabilize memory, they wrote.
In an accompanying editorial, Amy Milton, PhD, and Barry Everitt, PhD, of the University of Cambridge in England, agreed that "targeting maladaptive memories that play an important role in the persistence of addictive behavior may provide a new avenue for treatment interventions."
------------------------------------
Wiping Drug Memories
Science 13 April 2012: Perspective
Neuroscience
Amy L. Milton and Barry J. Everitt
Behavioural and Clinical Neuroscience Institute, Department
of Experimental Psychology, University of Cambridge
The tendency to relapse in individuals who are trying to remain abstinent is a major, but as yet unrealized, treatment target for drug addiction. On page 241 in this issue, Xue et al. (1) suggest that the memories elicited by drug cues and contexts can be diminished, thereby reducing their impact on relapse in both animals and people addicted to drugs.
Memories persist through the process of reconsolidation (2). When retrieved, a previously consolidated memory can enter a labile state in which new information can be introduced, before the memory restabilizes to persist in the brain, in its new updated form (3). By reexposing addicts to drug cues, a process similar to "cue-exposure therapy" (4), Xue et al. have manipulated the memory-updating process to overwrite the original memory and thereby reduce the risk of relapse in the long term. Rather than solely inducing memory extinction-the formation of a new "cue-no drug" memory that competes with the original "cue-drug" memory-the authors have used "extinction within the reconsolidation window" to reduce drug-seeking. A similar procedure for cue-fear memories has been shown to reduce fear in rats and humans (5, 6).
The authors used two models of drug use in rats that allow associations to form between stimuli (cues) and contexts, and two addictive drugs, cocaine and heroin. In the acute model, rats received injections of either cocaine or heroin in a specific environment or control injections in another environment. Subsequently, their preference for the drug-paired environment, tested when the rats were drug-free, provided a measure of their drug memory. Animals were reexposed to the drug-paired environment to reactivate the cue-drug memory and after a delay of 10 min, 1 hour, or 6 hours, they were exposed continuously to the same drug environment (but given no drug) to engage memory extinction processes. Only in the 10-min and 1-hour groups was subsequent preference for the drug environment abolished, and it did not return even after "reminder" injections of the drug.
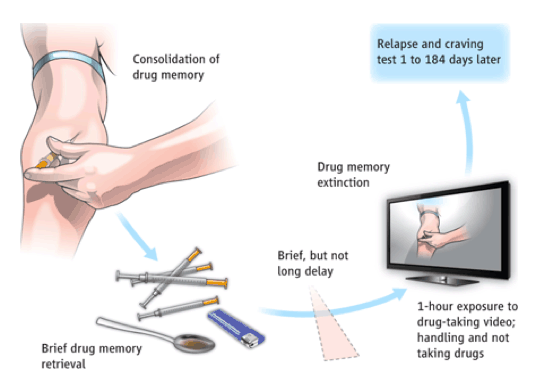
Decreasing craving and relapse.
A nonpharmacological method that weakens original cue-drug memories can decrease drug craving and relapse in abstinent heroin addicts (1). Heroin addicts associate drug cues and contexts with the addictive drug effect, establishing a drug memory. If a heroin addict (who has undergone detoxification) is shown images of drug-associated cues (such as drug paraphernalia) over an extended time period, this engages memory extinction processes, but this procedure does not erase the original cue-drug memory and the cravings can return. However, if the addict undergoes a brief "memory reactivation session" 10 min before extinction, this engages a memory reconsolidation process, which updates the old memory. The result is a greatly diminished "drug memory" later on, and drug cravings are reduced.
This apparent loss of drug memory was also observed in a more complex drug self-administration model, which more closely approximates addictive behavior in humans. Rats were trained to self-administer either cocaine or heroin, in the presence of drug-associated cues, over an extended period during which cue-drug memories were formed (7, 8). The animals then either underwent only extinction training or received a brief cue-drug memory-reactivation session 10 min before extinction training. When assessed 1 month later, only the group that had received the reminder and extinction showed a reduced tendency to resume drug-seeking behavior following exposure to either drug-associated cues or a reminder injection of cocaine or heroin. Furthermore, although extinction training occurred in a different environment to that of self-administration training, the resumption of drug seeking was reduced in the original self-administration environment. Thus, the usual context specificity of extinction (9) was overcome, suggesting that it may be possible to treat individuals in the clinic, with the beneficial effects of the treatment realized in the now-abstinent drug user's environment.
Remarkably, the authors successfully translated the approach of manipulating reconsolidation and extinction to a population of heroin addicts (see the figure). Three groups of patients, having undergone heroin detoxification, were briefly exposed to either a drug-relevant video or a control video followed, 10 min or 6 hours later, by 60 min of heroin cue extinction (they were exposed to imagery and drug paraphernalia, but given no drugs). Subjective craving and physiological responses (heart rate and blood pressure) were measured from 1 to 184 days later. Only the group that had the 10-min delay between the heroin video and extinction showed a marked reduction in craving and blood pressure after presentation of heroin-associated cues at every time point tested.
Although an increasing amount is known about the cellular and molecular mechanisms underlying reconsolidation (10, 11) and extinction (12)-e.g., both depend upon activation of N-methyl-d-aspartate (NMDA) receptors and intracellular signaling pathways-little is known about the nature of the interaction between reconsolidation and extinction that would explain why the extinction observed in this procedure is so profound and escapes its usual context specificity (9). Xue et al. point to a possible involvement of the atypical protein kinase C isoform PKM∫, a scaffold protein that maintains α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors in potentiated synapses (13) and so may reflect the structural changes in neurons that support a stabilized memory (14). Extinction training increased PKM∫ expression in the infralimbic cortex, which has long been implicated in the extinction of drug memories (12), and this increase was further enhanced by a memory reminder session before extinction training, perhaps strengthening extinction. Likewise, extinction reduced the expression of PKM∫ in the basolateral amygdala, an area critical for the reconsolidation of cue-drug memories (11), and this was further reduced by a memory reminder session before extinction training, perhaps suggesting impaired reconsolidation. How these changes occur, how they relate to each other, and whether they are causal in the apparent, subsequent amnesia remain to be investigated.
Perhaps the most remarkable aspect of these findings is that the effects of a purely behavioral manipulation in rats that in no sense were "addicted" translate so readily to patients who have been addicted to heroin for more than 9 years. This is especially so considering that even in the animal model with longer, 10-day drug self-administration histories, drug-taking behavior itself was explicitly extinguished, although no analogous process occurs either in the real-world environment of those addicted to drugs, or in the clinical study, in which there was only passive exposure to drug memory-eliciting stimuli. With these conundrums in mind and the obscure molecular mechanism, the findings nevertheless encourage the view that targeting maladaptive memories that play an important role in the persistence of addictive behavior may provide a new avenue for treatment interventions (15).
----------------------------
A Memory Retrieval-Extinction Procedure to Prevent Drug Craving and Relapse
Science 13 April 2012
Yan-Xue Xue,1* Yi-Xiao Luo,1* Ping Wu,1* Hai-Shui Shi,1,2 Li-Fen Xue,1 Chen Chen,1 Wei-Li Zhu,1
Zeng-Bo Ding,1 Yan-ping Bao,1 Jie Shi,1 David H. Epstein,3 Yavin Shaham,3 Lin Lu1
1National Institute on Drug Dependence, Peking University,
Beijing 100191, China. 2Department of Biochemistry and
Molecular Biology, Basic Medical College, Hebei Medical
University, Shijiazhuang 050017, China. 3Intramural Research
Program, National Institute on Drug Abuse, National Institutes
of Health, Baltimore, MD 21224, USA.
Abstract
Drug use and relapse involve learned associations between drug-associated environmental cues and drug effects. Extinction procedures in the clinic can suppress conditioned responses to drug cues, but the extinguished responses typically reemerge after exposure to the drug itself (reinstatement), the drug-associated environment (renewal), or the passage of time (spontaneous recovery). We describe a memory retrieval-extinction procedure that decreases conditioned drug effects and drug seeking in rat models of relapse, and drug craving in abstinent heroin addicts. In rats, daily retrieval of drug-associated memories 10 minutes or 1 hour but not 6 hours before extinction sessions attenuated drug-induced reinstatement, spontaneous recovery, and renewal of conditioned drug effects and drug seeking. In heroin addicts, retrieval of drug-associated memories 10 minutes before extinction sessions attenuated cue-induced heroin craving 1, 30, and 180 days later. The memory retrieval-extinction procedure is a promising nonpharmacological method for decreasing drug craving and relapse during abstinence.
Conditioning plays a major role in drug addiction, and responses to drug-associated cues persist during prolonged abstinence (1, 2). These findings led to the development of cue-exposure therapies to extinguish the craving- and relapse-provoking effects of drug cues (1, 3). However, cue-exposure therapy in clinical settings does not usually prevent relapse when former drug addicts return to their previous drug environments (4). Animal learning studies predict that extinction responding is susceptible to renewal, reinstatement, and spontaneous recovery. Respectively, these terms refer to resumption of original learned responses after change of environmental context, acute exposure to the unconditioned stimulus (such as food or drug), or passage of time (5).
More recently, preclinical investigators have been able to decrease behavioral effects of drug-associated cues by pharmacologically interfering with reconsolidation of drug-cue memories (6-9). Reconsolidation refers to a time-dependent process in which consolidated memory items are rendered transiently unstable shortly after their reactivation (10-12). However, with the exception of the beta-adrenoceptor antagonist propranolol (13, 14), which is approved for human use, the other pharmacological compounds used in these studies are not suitable for human use (15-18). Consequently, the results from rat reconsolidation studies have not yet "translated" to clinical use in addiction treatment.
A nonpharmacological alternative may be possible: the "memory retrieval-extinction" behavioral procedure to interfere with reconsolidation of fear cues in rats and humans (19, 20). Reinstatement, renewal, and spontaneous recovery of fear responding are prevented by acute exposure to cues previously paired with footshock (a retrieval manipulation) if that exposure is followed 10 min or 1 hour later (but not 6 hours later) by repeated exposure to the same cues in extinction sessions. Thus, extinction experience within the timeframe of the "reconsolidation window" after cued retrieval of the fear memories mimicked the behavioral effect of a pharmacological manipulation on suppression of fear conditioning (19, 20).
We used an extinction-reinstatement procedure in rats [an animal model of drug relapse (21)] and a cue-induced-craving procedure in humans (1) to assess whether the memory retrieval-extinction procedure can decrease drug- and cue-induced drug preference and relapse in rats and cue-induced drug craving in humans.
We first assessed the effect of the memory retrieval-extinction manipulation on drug-priming-induced reinstatement of drug conditioned place preference (CPP) and spontaneous recovery of drug CPP (details are available in the supplementary material). In the Pavlovian CPP version of the reinstatement model, CPP is induced by a drug, extinguished, and then reinstated by priming injections of the drug (21); like other conditioned responses (22), the extinguished CPP response can reemerge after the passage of time (spontaneous recovery).
In experiment 1 (cocaine CPP) (fig. S1A) and experiment 2 (morphine CPP) (Fig. 1A), we used four groups of rats: (i) no memory retrieval + extinction; (ii) memory retrieval + 10-min delay + extinction; (iii) memory retrieval + 1-hour delay + extinction; and (iv) memory retrieval + 6-hour delay + extinction. We analyzed the data with the between-subjects factor of group and the within-subjects factor of CPP test (test 2, after extinction; test 3, drug-induced reinstatement or spontaneous recovery test). Brief (10 min) cued retrieval of the drug memories 10 min or 1 hour but not 6 hours before the longer 45-min daily extinction sessions impaired drug-priming-induced reinstatement of drug CPP for cocaine (table S1, statistical results, and fig. S1) and morphine (group x test interaction, F3,25 = 9.5, P < 0.01) (Fig. 1). In experiment 3, we used an identical experimental procedure to demonstrate that the memory retrieval-extinction manipulation also impaired spontaneous recovery of cocaine CPP (fig. S2).
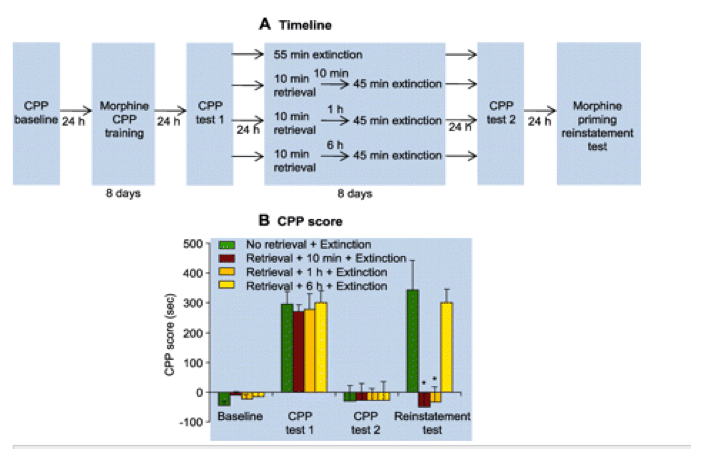
Fig. 1
In rats, retrieval of drug-cue memories 10 min or 1 hour before extinction sessions prevented drug-priming-induced reinstatement of morphine CPP. (A) During CPP training, rats learned to associate one environmental context with the effect of morphine injections (10 mg/kg, subcutaneous) and to associate another context with saline injections. Next, all rats were tested for their place preference (CPP test 1). Twenty-four hours later, rats were divided into four groups and given different memory retrieval-extinction manipulations: 55-min extinction training (in one group), or 10-min memory retrieval + 45 min extinction training (in the other three groups-with either a 10-min, 1-hour, or 6-hour delay between memory retrieval and extinction training). All rats were tested for reinstatement of morphine CPP induced by a priming injection of morphine (5 mg/kg, subcutaneous). (B) Effect of the experimental manipulations on the CPP score. Data are mean � SEM of preference score in seconds (time spent in the morphine-paired chamber minus time spent in the saline-paired chamber) during the CPP tests. Asterisk indicates different from the "no memory retrieval" condition; P < 0.05; n = 9 to 11 rats per experimental condition.
We next assessed the effect of the memory retrieval-extinction manipulation on drug-priming-induced reinstatement of the drug self-administration behavior, spontaneous recovery, and context-induced reinstatement of drug seeking [a renewal manipulation (23)]. These experiments used the operant self-administration version of the reinstatement model, in which animals are trained to respond for drug infusions, given daily extinction sessions until operant responding ceases, and then tested for reinstatement of (nonreinforced) pressing on the drug-associated device (such as a lever or nosepoke operandum) after acute noncontingent exposure to drug-priming injections or exposure to drug-associated cues (24, 25). A selective increase in nonreinforced responding on the device previously associated with drug infusions (but not on the inactive device) is interpreted to indicate relapse to drug seeking (26). Like drug CPP, the extinguished drug-reinforced conditioned response undergoes spontaneous recovery after the completion of extinction training (27).
In experiments 4 and 5, we used rats that had been trained to self-administer cocaine or heroin to demonstrate the inhibitory effect of the memory retrieval-extinction manipulation on drug-priming-induced reinstatement (Fig. 2 and fig. S3). The statistical analysis included the between-subjects factor of group (no memory retrieval + extinction, memory retrieval + 10-min delay + extinction, and memory retrieval + 6-hour delay + extinction for cocaine; or no memory retrieval + extinction and memory retrieval + 10-min delay + extinction for heroin) and the within-subjects factor of reinstatement condition (last extinction session, reinstatement test session). Brief (15 min) cued retrieval of the drug memories 10 min but not 6 hours before the long 180-min daily extinction sessions impaired drug-priming-induced reinstatement of cocaine (fig. S3) or heroin (Fig. 2) seeking. There were significant group x reinstatement condition interactions for both cocaine (table S2) and heroin (F1,10 = 6.9, P < 0.05). No group differences were seen in responding on the inactive nosepoke operandum (P > 0.1). Additionally, the memory retrieval-extinction manipulation accelerated extinction responding in the cocaine-trained rats (table S2 and fig. S3) but not the heroin-trained rats (P > 0.05) (Fig. 2).
In experiments 6 and 7, we used rats that had been trained to self-administer cocaine to demonstrate the inhibitory effect of the memory retrieval-extinction manipulation on spontaneous recovery (experiment 6) and renewal (context-induced reinstatement; experiment 7) of cocaine seeking (figs. S4 and S5). In the renewal experiment, the rats were trained to self-administer cocaine in a distinct context (context A). Then, the operant responding was extinguished in a different, nondrug context (context B). During the subsequent tests, reinstatement of cocaine seeking was assessed after exposure to context A (28). Brief (15 min) cued retrieval of the cocaine memories 10 min before the long 180-min daily extinction sessions impaired spontaneous recovery (fig. S4) and renewal (fig. S5) of cocaine seeking. The statistical analyses, which included the between-subjects factor of group (no memory retrieval + extinction and memory retrieval + 10-min delay + extinction) and the within-subjects factor of test condition (last extinction session, spontaneous recovery, or renewal test session), showed significant interactions between group x test condition (table S2). No group differences were seen in responding on the inactive nosepoke operandum (P > 0.1). Additionally, the memory retrieval-extinction manipulation modestly accelerated extinction responding in experiment 6 (fig. S4) but not experiment 7 (fig. S5).
In experiment 8, we assessed the effect of the memory retrieval-extinction manipulation on the protein expression of protein kinase M∫ (PKM∫) in medial prefrontal cortex (mPFC; infralimbic and prelimbic subregions) and amygdala (basolateral and central subregions). PKM∫ is a constitutively active atypical isoenzyme of protein kinase C that mediates long-term maintenance of aversive and appetitive memories (29, 30), including drug-associated memories (31, 32). We found that extinction training alone increased PKM∫ expression in infralimbic (but not prelimbic) cortex and decreased PKM∫ expression in basolateral (but not central) amygdala (fig. S6). Furthermore, the memory retrieval-extinction manipulation with a 10-min but not 6-hour delay potentiated extinction-induced increases in PKM∫ expression in infralimbic cortex and extinction-induced decreases in PKM∫ expression in basolateral amygdala (table S2 and fig. S6). There was no group effect on the levels of ß-actin (a control protein).
Next, we assessed the clinical relevance of the memory retrieval-extinction procedure in inpatient detoxified heroin addicts. Heroin craving was assessed by using a visual analog scale (VAS) on which the participants had to rate their current craving for heroin, before and immediately after exposure to a neutral cue and a heroin cue. Neutral and heroin cues were both 5-min videotapes. Heart rate (HR) and blood pressure were monitored before and after cue exposure as additional measures of cue reactivity. The heroin addicts were assigned to three groups: (i) no memory retrieval + extinction, (ii) memory retrieval + 10-min delay + extinction, or (iii) memory retrieval + 6-hour delay + extinction. The memory retrieval-extinction manipulation with a 10-min but not 6-hour delay inhibited both cue-induced craving (Fig. 3) and cue-induced increases in blood pressure, but not heart rate (Fig. 4). Craving reactivity to cues was assessed by using change scores from preexposure baseline (33).
For cue-induced craving, the statistical analysis [SAS PROC MIXED, Satterthwaite method for denominator degrees of freedom that takes into account missing cells in repeated-measures analysis of variance (ANOVA)], which included baseline (day 1) as the covariate, the between-subjects factor of group, and the within-subjects factors of test day (posttreatment tests on days 4, 34, and 184) and cue type (neutral cue and heroin cue), showed a significant interaction between group x cue type (F2,23.75 = 9.0, P < 0.01). For systolic and diastolic blood pressure, the analyses showed a trend toward an interaction between group x cue type (F2,343.25 = 2.38, P = 0.094 and F2,374.46 = 2.42, P = 0.09, respectively). No group differences were observed for heart rate.
In 2009, Monfils and colleagues introduced a memory retrieval-extinction procedure whose application to both rats (19) and humans (20) led to long-lasting blockade of shock-conditioned fear responses. Here, we introduce an appetitive-conditioning version of the memory retrieval-extinction procedure whose application caused long-lasting attenuation of conditioned drug effects and drug seeking (in rats) and drug craving (in detoxified heroin users). The behavioral effects of our procedure were also associated with changes in the expression of the memory-maintenance-related molecule PKM∫ in infralimbic cortex and basolateral amygdala. As in the fear-conditioning studies, a key determinant of effectiveness was the interval between the shorter memory-retrieval sessions and the longer extinction sessions or the interval between short reexposure to the drug-associated cues (memory retrieval) and subsequent longer nonreinforced reexposure to the same cues (extinction).
The development of the memory retrieval-extinction fear-conditioning procedure was inspired by theoretical accounts of memory retrieval and reconsolidation (34) and studies of pharmacological manipulations of reconsolidation of fear memories (11, 35). The latter body of work has since been extended to appetitive memories (6), including memories of drug-associated cues (7, 36). In those studies, investigators inferred that memory reconsolidation was disrupted on the basis of findings that post-retrieval systemic or intracranial injections of pharmacological agents within a specific time interval (up to 2 hours after retrieval)-often termed a "reconsolidation window"-disrupted the expression of responses to aversive or appetitive cues (6, 12, 37). Accordingly, results from studies of memory retrieval-extinction manipulations have been taken to reflect interference with reconsolidation (19, 20, 38). The findings that the memory retrieval-extinction manipulation is ineffective when the extinction sessions are given at delays that are longer than the reconsolidation window (19, 20, 38) supports this interpretation. The retrieval-extinction manipulation also blocks shock-induced reinstatement, spontaneous recovery, and renewal of conditioned-fear expression [phenomena that are otherwise reliably observed after extinction training (5)] in some studies (19, 20, 38), but not others (39, 40). On the basis of the above findings, a plausible interpretation of our data is that the memory retrieval-extinction manipulation interfered with reconsolidation of memories for drug cues. This hypothesis is supported by two sets of observations. First, across the different experiments in both rats and humans, the memory retrieval-extinction manipulation was effective only within the time window of reconsolidation. Second, in the CPP experiments the memory retrieval-extinction manipulation completely blocked drug-priming-induced reinstatement and spontaneous recovery.
However, a reconsolidation account of the data should be made with some caution in the case of the drug self-administration experiments. In those experiments, nosepoke responding was significantly lower during the last extinction session than during the tests for drug-priming-induced reinstatement, spontaneous recovery, and renewal in the 10-min or 1-hour memory retrieval-extinction condition. Additionally, the effect of the memory retrieval-extinction manipulation on nosepoke responding during extinction training-a behavior induced in part by exposure to the drug-associated cues (26)-was modest and inconsistent across experiments. Together, these observations suggest that our memory retrieval-extinction manipulation only weakened the memories of the drug cues (or decreased their motivational effects) rather than completely preventing the expression of the conditioned response, as would have been predicted by a reconsolidation account of the data.
What might account for the attenuation but not blockade of drug seeking in the self-administration experiments? One possibility is that the memory retrieval-extinction manipulation preferentially disrupted reconsolidation of stimulus-response Pavlovian-based memories that mediate drug CPP in rats and cue-induced drug craving in humans, while having less impact on reconsolidation of response-outcome operant-based memories that play a role in reinstatement of drug seeking in the drug self-administration procedure. Operant drug seeking is controlled by a complex interplay between operant and Pavlovian conditioning processes (7, 26), and there is evidence that reconsolidation of operant memories is more difficult to disrupt than reconsolidation of Pavlovian memories (41).
Another issue to consider in interpreting the present data is that the memory-retrieval manipulations were performed under extinction conditions, and therefore, a given manipulation could have affected reconsolidation of cue memories, consolidation of extinction memory, or both (6). Our retrieval manipulation in the self-administration experiments was 15 min of daily non-reinforced operant responding in the presence of the drug-associated cues. Thus, an alternative interpretation could be that intermittent exposure to extinction training within the consolidation window of extinction memory may have strengthened the extinction memory, rendering the original appetitive memory less susceptible to reinstatement, spontaneous recovery, or renewal. Indeed, results from fear-conditioning studies demonstrate that pharmacological manipulations that promote consolidation of extinction memory, decrease reinstatement, spontaneous recovery, and renewal of fear memories (42-44).
It is also possible that our memory retrieval-extinction manipulation both facilitated extinction consolidation and disrupted reconsolidation. Two lines of evidence from published reports support this hypothesis. The first is our recent finding that post-training PKM∫ activity in basolateral amygdala is critical for memories of morphine reward and morphine withdrawal aversion but not extinction memory, whereas PKM∫ activity in infralimbic cortex is critical for extinction memory but not reward or withdrawal memories (31). The second is that plasticity in basolateral amygdala is critical for reconsolidation of memories for both aversive and appetitive cues (6, 7) and for the effects of the retrieval-extinction manipulation on fear memories (38), whereas infralimbic plasticity is critical for maintenance of aversive and appetitive extinction memories (44, 45). In the experiments reported here, we found that repeated cocaine-cue retrieval 10 min before daily extinction sessions potentiated the opposite effects of extinction training alone on PKM∫ in the infralimbic cortex (increased expression) versus basolateral amygdala (decreased expression) (fig. S6). These findings are consistent with a "dual" effect of the memory retrieval-extinction manipulation on both consolidation of extinction memory and reconsolidation of cue memories.
Investigators have identified several ways to disrupt cue-memory reconsolidation or strengthen extinction learning (7, 46). However, their potential as preventive treatments for addiction is limited because they often rely on pharmacological agents that are either not approved for human use or that can cause problematic side effects. We used established animal models of drug relapse and a standard human laboratory procedure for drug-induced craving to assess a purely behavioral procedure to decrease the motivational effects of drug cues during abstinence. The memory retrieval-extinction procedure decreased cue-induced drug craving and (extrapolating from our rat data) perhaps could reduce the likelihood of cue-induced relapse during prolonged abstinence periods. If our procedure weakens the original drug-cue memories rather than solely facilitating extinction, it may overcome the contextual renewal problems that have limited the clinical effectiveness of traditional extinction procedures (4), although this possibility needs empirical evaluation in human addicts. Last, although the cellular mechanisms and brain circuits underlying the long-lasting effects of the retrieval-extinction procedure on drug relapse and craving remain to be elucidated, our data point to a role for PKM∫ activity in the infralimbic cortex and basolateral amygdala.
|
| |
|
 |
 |
|
|