| |
Fracture Risk and Zoledronic Acid Therapy in Men with Osteoporosis
|
| |
| |
Download the PDF here
Download the PDF here
N Engl J Med Nov 1 2012
Osteoporosis is an important cause of morbidity and mortality among men.1,2 Among persons older than 50 years of age, approximately 40% of all osteoporotic fractures worldwide occur in men.3 Mortality after osteoporotic fracture is higher among men than among women. 2,4
Previous studies involving men with osteoporosis have focused on the surrogate outcomes of bone mineral density and bone-turnover markers,5-9 but data from double-blind, randomized studies assessing antifracture efficacy are lacking. In addition, given the low awareness of the disease,10 the development of guidelines for the detection and treatment of osteoporosis in men has been limited.11 Hence, there is a need for randomized trials of osteoporosis treatment in men, with fracture as a primary end point. Men at risk for fractures are commonly not identified or treated.12
Zoledronic acid (Reclast, Novartis Pharmaceuticals; Aclasta, Novartis Pharma) is a bisphosphonate administered intravenously. At a dose of 5 mg once a year, it has antifracture efficacy in postmenopausal women with osteoporosis and positive effects on bone mineral density in men.13,14 Our multicenter, randomized, prospective trial assessed the effect of zoledronic acid on the risk of vertebral fracture among men with osteoporosis.
Background
Fractures in men are a major health issue, and data on the antifracture efficacy of therapies for osteoporosis in men are limited. We studied the effect of zoledronic acid on fracture risk among men with osteoporosis.
Methods
In this multicenter, double-blind, placebo-controlled trial, we randomly assigned 1199 men with primary or hypogonadism-associated osteoporosis who were 50 to 85 years of age to receive an intravenous infusion of zoledronic acid (5 mg) or placebo at baseline and at 12 months. Participants received daily calcium and vitamin D supplementation. The primary end point was the proportion of participants with one or more new morphometric vertebral fractures over a period of 24 months.
Results
The rate of any new morphometric vertebral fracture was 1.6% in the zoledronic acid group and 4.9% in the placebo group over the 24-month period, representing a 67% risk reduction with zoledronic acid (relative risk, 0.33; 95% confidence interval, 0.16 to 0.70; P=0.002). As compared with men who received placebo, men who received zoledronic acid had fewer moderate-to-severe vertebral fractures (P=0.03) and less height loss (P=0.002). Fewer participants who received zoledronic acid had clinical vertebral or nonvertebral fractures, although this difference did not reach significance because of the small number of fractures. Bone mineral density was higher and bone-turnover markers were lower in the men who received zoledronic acid (P<0.05 for both comparisons). Results were similar in men with low serum levels of total testosterone. The zoledronic acid and placebo groups did not differ significantly with respect to the incidence of death (2.6% and 2.9%, respectively) or serious adverse events (25.3% and 25.2%).
Conclusions
Zoledronic acid treatment was associated with a significantly reduced risk of vertebral fracture among men with osteoporosis. (Funded by Novartis Pharma;
Discussion
Over a 2-year period, two annual infusions of zoledronic acid significantly reduced the risk of new morphometric vertebral fractures by 67% among men with osteoporosis. This reduction was similar to that reported in postmenopausal women with osteoporosis who received zoledronic acid (relative reduction in the risk of vertebral fracture, 71% at 2 years),13 suggesting that the antifracture effect of zoledronic acid is independent of sex. Zoledronic acid therapy had an acceptable safety profile. These results provide support for the value of antiresorptive therapy in men with osteoporosis.
Our study showed that zoledronic acid reduced the risk of height loss and moderate-to-severe vertebral fractures, which are associated with an increased risk of subsequent vertebral and nonvertebral fractures.20-23 Although the power of the study to detect a reduction in the risk of nonvertebral fracture was modest, rates of nonvertebral fracture were consistently lower among men who received zoledronic acid than among those who received placebo, and the point estimates were similar to the significant risk reductions in larger studies involving women.13
Zoledronic acid significantly improved bone mineral density and reduced bone-turnover markers, with changes from baseline that were similar to those reported for other bisphosphonates in men with osteoporosis6-9 and were consistent with those seen in postmenopausal women with osteoporosis receiving bisphosphonates (including zoledronic acid).13,24,25 Previous studies of bisphosphonates in men with osteoporosis have consistently revealed beneficial effects on bone mineral density and bone-turnover markers, but they were not primarily designed to assess the effects on fractures.6-9 For example, alendronate lowered the incidence of morphometric vertebral fractures (a secondary end point) in men in a 2-year double-blind trial involving 241 patients, but the number of fractures was small.7 Since clinical data showing a reduction in the risk of fracture among men with osteoporosis have been lacking, our study was designed to be placebo-controlled. We believed that clinical equipoise existed and that a positive result would probably improve care for men with osteoporosis. Recently, denosumab was shown to reduce the risk of vertebral fracture (a secondary end point) among men receiving androgen-deprivation therapy for nonmetastatic prostate cancer.26,27 Our data provide further support for the precept that antiresorptive treatments are effective in both men and women.
In our study, zoledronic acid had similar beneficial effects on fractures and bone mineral density in men with low testosterone levels and men with normal levels. However, few men had total testosterone levels that were low enough (<230 ng per deciliter) to benefit from testosterone treatment,19 making it difficult to draw conclusions about the effect of zoledronic acid in this population. Furthermore, because the randomization was not stratified according to total testosterone level, the numbers of patients with low levels in the two groups were different.
Despite the fact that current public health efforts to detect osteoporosis and prevent fractures in men are inadequate,28 the ability to establish detection and treatment recommendations has been limited because of the absence of unambiguous evidence of effective antifracture therapies in men.11 Although our findings with zoledronic acid do not imply that all data on drugs for osteoporosis in women can be extrapolated to men, our study should provide the confidence to proceed.
A key strength of this study was a study population that was sufficiently large to detect an effect of treatment on the risk of vertebral fracture. However, the study was not powered to address the effect of zoledronic acid on nonvertebral (including hip) fractures. For ethical reasons, men with multiple or severe vertebral fractures were not enrolled, and the patient population was relatively young. The significant difference that we observed in the incidence of myocardial infarction between the groups has not previously been observed with zoledronic acid,13,14 and any causality or association with zoledronic acid is unknown.
In conclusion, our prospective study that assessed fractures as the primary end point in men with osteoporosis showed that over a 2-year period, a once-yearly infusion of zoledronic acid at a dose of 5 mg was associated with a significant decrease in the risk of new vertebral fractures.
Results
Study Participants
In total, 588 men were randomly assigned to zoledronic acid, and 611 men were randomly assigned to placebo (Figure 1.); 58 men (9.9%) and 71 men (11.6%) in the two groups, respectively, discontinued the study. The modified intention-to-treat population comprised a total of 553 men who received zoledronic acid and 574 men who received placebo; these patients underwent baseline assessments and one or more post-baseline assessments of the primary efficacy variable. Fifty-two men who received zoledronic acid (8.8%) and 53 men who received placebo (8.7%) did not receive the second infusion.
Baseline characteristics were similar between the groups (Table 1.). Total testosterone measurements were available for 495 participants who received zoledronic acid and 516 participants who received placebo. A total of 116 men who received zoledronic acid (23.4%) and 149 men who received placebo (28.9%) had a total testosterone level of 350 ng per deciliter or less; a small proportion of men in the two groups combined (5.4%) had levels of 230 ng per deciliter or less. Baseline bone mineral density and bone-turnover marker levels were similar across all subgroups of total testosterone levels (Table S1 in the Supplementary Appendix); results with the use of 230 ng per deciliter as the threshold were similar to those with the use of 350 ng per deciliter (Tables S2, S3, and S4 in the Supplementary Appendix).
Fractures
A total of 30 of 553 men in the zoledronic acid group (5.4%) and 40 of 574 men in the placebo group (7.0%) had radiographs that could be evaluated at month 12 but not at month 24; 4 men (0.7%) and 3 men (0.5%), respectively, had radiographs that could be evaluated at month 24 only; the remaining patients had radiographs that could be evaluated at both time points. A significantly lower proportion of men in the zoledronic acid group (1.6%) had one or more new morphometric vertebral fractures over 24 months, as compared with men in the placebo group (4.9%) (Figure 2.), corresponding to an absolute risk reduction of 3.3 percentage points and a relative risk reduction of 67% (P=0.002). Sensitivity analyses with the use of data on patients for whom results of radiography at month 24 were available, single imputation, and multiple imputation had similar results (Table S5 in the Supplementary Appendix). A 68% reduction in the relative risk of new morphometric vertebral fractures with zoledronic acid was apparent at month 12 (P=0.02) (Figure 2, and Table S6A in the Supplementary Appendix). The total testosterone level did not affect the antifracture efficacy of zoledronic acid (P>0.80 for interaction). Among men with serum total testosterone levels of 350 ng per deciliter or less, zoledronic acid was associated with a nonsignificant 67% reduction in the relative risk of new morphometric vertebral fractures (P=0.13) (Table S2 in the Supplementary Appendix).
Significantly fewer men who received zoledronic acid than men who received placebo had one or more new moderate-to-severe morphometric vertebral fractures, both at month 12 (relative risk reduction, 81%; P=0.01) and at month 24 (relative risk reduction, 63%; P=0.03). Similar results were seen for new or worsening morphometric vertebral fractures at month 12 (relative risk reduction, 55%; P=0.07) and month 24 (relative risk reduction, 59%; P=0.007). Changes in height (least-squares mean) from baseline were -0.8 and -2.5 mm at month 12 (P=0.008) and -2.2 and -4.5 mm at month 24 (P=0.002) in the zoledronic acid and placebo groups, respectively.
Six men who received zoledronic acid (1.0%) and 11 men who received placebo (1.8%) had one or more clinical vertebral or nonvertebral fractures during the study (Table S6B in the Supplementary Appendix), but the between-group difference was not significant. With the use of the observed effects in the HORIZON studies,13,14 Bayesian analyses suggested that zoledronic acid may reduce the risk of clinical fractures among men (Table S6C in the Supplementary Appendix).
Bone Density
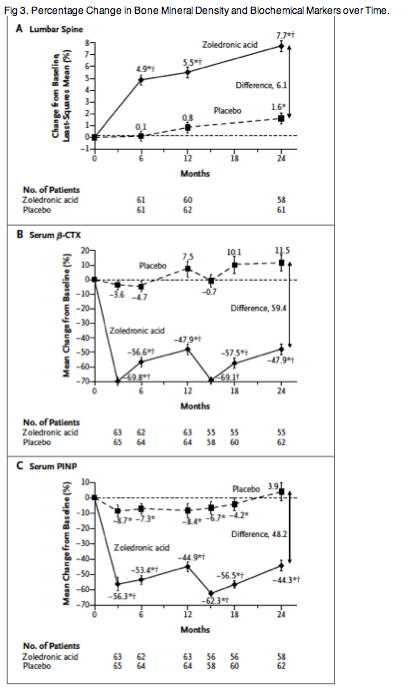
As compared with placebo, zoledronic acid was associated with significant and sustained increases in bone mineral density at the lumbar spine, total hip, and femoral neck over a 24-month period (P<0.05 for all comparisons) (Figure 3, and Fig. S1A and S1B in the Supplementary Appendix). The effect of zoledronic acid on bone mineral density was similar in men with total testosterone levels of more than 350 ng per deciliter and in men with levels of 350 ng per deciliter or less (P>0.40 for interaction) (Table S3 in the Supplementary Appendix).
Biochemical Markers
Serum ß-CTX, PINP, and BSAP levels were lower in men who received zoledronic acid than in men who received placebo at all time points measured (P<0.001 for all comparisons) (Figure 3, and Fig. S1C in the Supplementary Appendix). A similar pattern was seen in urinary N-terminal telopeptide levels (P<0.001 for all comparisons). The effect of zoledronic acid on bone-turnover markers was generally similar in men with total testosterone levels of more than 350 ng per deciliter and those with levels of 350 ng per deciliter or less (P>0.10 for interaction at months 12 and 24) (Table S4 in the Supplementary Appendix), with the exception of PINP, for which the reduction was significantly less in the subgroup of men with total testosterone levels of 350 ng per deciliter or less than in the subgroup with levels of more than 350 ng per deciliter (P<0.02).
Safety
No significant differences were observed between the two groups with respect to deaths or serious adverse events, with the exception of any myocardial infarction (in nine men [1.5%] in the zoledronic acid group and two [0.3%] in the placebo group, [P=0.03]; none of the events were considered by the investigator to be related to the study drug) (Table 2.). There were 31 cardiac serious adverse events in the zoledronic group (5.3%) and 30 in the placebo group (4.9%) (P=0.79). Men who received zoledronic acid reported more adverse events of pyrexia, myalgia, arthralgia, headaches, chills, pain in the extremities, and influenza-like symptoms. There were no significant differences between the groups in the incidence of atrial fibrillation, cardiac arrhythmias, or renal dysfunction. No cases of osteonecrosis of the jaw were observed. Two men in the zoledronic acid group and one man in the placebo group had hip fractures during the study, but none were atypical or subtrochanteric.
| |
| |
| |
|
|
|